Abstract
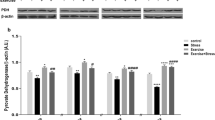
Chronic stress has been linked to many diseases resulted from dysfunction of both the nervous system and peripheral organ systems. Yet, the effects of chronic stress on the pancreas have received relatively little attention. This work aims to investigate the influence of chronic stress exposure on both the endocrine and exocrine pancreatic function and morphology and its possible mechanism of action, and also to evaluate the impact of chronic exercise with moderate intensity on ameliorating the stress-induced pancreatic changes. Forty adult male albino rats were used and divided into four groups: control group, exercised group (3 weeks of swimming exercise), stressed group (3 weeks of immobilization stress), and stressed group practicing exercise (3 weeks of exercise, concomitant with 21 daily sessions of stress). On the final day of the experiment, all rats were sacrificed. Biochemical, immunohistochemical, and histological studies were conducted. The results showed that chronic immobilization stress produced hyperglycemia, hyperinsulinemia, and increased homeostatic model assessment of insulin resistance index (HOMA-IR) with increasing exocrine pancreatic injury markers by increasing oxidative and inflammatory status of the pancreatic tissue. Histological study showed the injurious effect of stress on the morphology of pancreatic tissue. Physical exercise protected the pancreas from the negative effects of stress through its anti-inflammatory and anti-oxidative effects, evidenced by increasing pancreatic interleukin 10 and total antioxidant capacity and decreasing pancreatic tumor necrosis factor-alpha, and malondialdehyde with ameliorating most of the histological changes induced by stress exposure. Physical exercise effectively counteracts chronic stress-induced pancreatic changes through different mechanisms.






Similar content being viewed by others
References
Afifi NM (2012) Effect of mesenchymal stem cell therapy on recovery of streptozotocin-induced diabetes mellitus in adult male albino rats: a histological and immunohistochemical study. Egyptian journal of histology 35:458–469
Allen J, Sun Y, Woods JA (2015) Exercise and the regulation of inflammatory responses. In: Progress in molecular biology and translational science, vol 135. Elsevier, Amsterdam, pp 337–354
Almeida FN, Proença ARG, Chimin P, Marçal AC, Bessa-Lima F, Carvalho CRO (2012) Physical exercise and pancreatic islets: acute and chronic actions on insulin secretion. Islets 4:296–301. https://doi.org/10.4161/isl.21273
Amaral F, Lima NE, Ornelas E, Simardi L, Fonseca FLA, Maifrino LBM (2015) Effect of different exercise intensities on the pancreas of animals with metabolic syndrome. Diabetes Metab Syndr Obes 8:115
Apte M, Pirola R, Wilson J (2009) New insights into alcoholic pancreatitis and pancreatic cancer. J Gastroenterol Hepatol 24:S51–S56
Aydin C, Sonat F, Sahin SK, Cangul IT, Ozkaya G (2009) Long term dietary restriction ameliorates swimming exercise-induced oxidative stress in brain and lung of middle-aged rat. Indian J Exp Biol 47:24–31
Bello M, Basilio-Antonio L, Fragoso-Vázquez J, Avalos-Soriano A, Correa-Basurto J (2017) Molecular recognition between pancreatic lipase and natural and synthetic inhibitors. Int J Biol Macromol 98:855–868
Bielajew C, Konkle AT, Merali Z (2002) The effects of chronic mild stress on male Sprague-Dawley and Long Evans rats: I. Biochemical and physiological analyses. Behav Brain Res 136:583–592
Binker MG, Cosen-Binker LI (2014) Acute pancreatitis: the stress factor. World J Gastroenterol 20:5801–5807. https://doi.org/10.3748/wjg.v20.i19.5801
Binker MG, Binker-Cosen AA, Richards D, Gaisano HY, de Cosen RH, Cosen-Binker LI (2010a) Chronic stress sensitizes rats to pancreatitis induced by cerulein: role of TNF-alpha. World J Gastroenterol 16:5565–5581
Binker MG, Binker-Cosen AA, Richards D, Gaisano HY, de Cosen RH, Cosen-Binker LI (2010b) Chronic stress sensitizes rats to pancreatitis induced by cerulein: role of TNF-α. World J Gastroenterol: WJG 16:5565
Booth FW, Roberts CK, Laye MJ (2012) Lack of exercise is a major cause of chronic diseases. Compr Physiol 2:1143–1211. https://doi.org/10.1002/cphy.c110025
Bouzid MA, Filaire E, Matran R, Robin S, Fabre C (2018) Lifelong voluntary exercise modulates age-related changes in oxidative stress. Int J Sports Med 39:21–28. https://doi.org/10.1055/s-0043-119882
Brown GR, Köhler M, Berggren P-O (1997) Parallel changes in nuclear and cytosolic calcium in mouse pancreatic β-cells. Biochem J 325:771–778
Carroll JK, Herrick B, Gipson T, Lee SP (2007) Acute pancreatitis: diagnosis, prognosis, and treatment. Am Fam Physician:75
Contarteze RVL, Manchado FDB, Gobatto CA, De Mello MAR (2008) Stress biomarkers in rats submitted to swimming and treadmill running exercises. Comp Biochem Physiol A Mol Integr Physiol 151:415–422
Côté S (1993) Current protocol for light microscopy immunocytochemistry. Immunohistochemistry II:148–167
Dhanalakshmi S, Devi RS, Srikumar R, Manikandan S, Thangaraj R (2007) Protective effect of triphala on cold stress-induced behavioral and biochemical abnormalities in rats. Yakugaku Zasshi 127:1863–1867. https://doi.org/10.1248/yakushi.127.1863
Duric V, Clayton S, Leong ML, Yuan LL (2016) Comorbidity factors and brain mechanisms linking chronic stress and systemic illness. Neural Plast 2016:5460732–5460716. https://doi.org/10.1155/2016/5460732
Eguchi R et al (2011) Fish oil consumption prevents glucose intolerance and hypercorticosteronemy in footshock-stressed rats. Lipids Health Dis 10:71
Fonseca SG, Urano F, Burcin M, Gromada J (2010) Stress hypERactivation in the β-cell. Islets 2:1–9
Franzoni F et al (2005) Physical activity, plasma antioxidant capacity, and endothelium-dependent vasodilation in young and older men. Am J Hypertens 18:510–516. https://doi.org/10.1016/j.amjhyper.2004.11.006
Fukushima M, Taniguchi A, Sakai M, Doi K, Nagasaka S, Tanaka H, Tokuyama K, Nakai Y (1999) Homeostasis model assessment as a clinical index of insulin resistance. Diabetes Care 22:1911–1912
Geffken DF, Cushman M, Burke GL, Polak JF, Sakkinen PA, Tracy RP (2001) Association between physical activity and markers of inflammation in a healthy elderly population. Am J Epidemiol 153:242–250. https://doi.org/10.1093/aje/153.3.242
Gerber M, Puhse U (2009) Review article: do exercise and fitness protect against stress-induced health complaints? A review of the literature. Scand J Public Health 37:801–819. https://doi.org/10.1177/1403494809350522
Ghorbanzadeh V, Mohammadi M, Mohaddes G, Dariushnejad H, Chodari L (2017) Effect of Crocin and voluntary exercise on P53 protein in pancreas of type2 diabetic rats. Pharm Sci 23:182–188
Gilgun-Sherki Y, Rosenbaum Z, Melamed E, Offen D (2002) Antioxidant therapy in acute central nervous system injury: current state. Pharmacol Rev 54:271–284
Goldstein DS, McEwen B (2002) Allostasis, homeostats, and the nature of stress. Stress 5:55–58. https://doi.org/10.1080/102538902900012345
Gu H, Tang C, Peng K, Sun H, Yang Y (2009, 2009) Effects of chronic mild stress on the development of atherosclerosis and expression of toll-like receptor 4 signaling pathway in adolescent apolipoprotein E knockout mice. J Biomed Biotechnol:613879. https://doi.org/10.1155/2009/613879
Health UDo, Services H (1992) Institutional Animal Care and Use Committee Guidebook. NIH Publication
Heiskanen MA, Motiani KK, Mari A, Saunavaara V, Eskelinen JJ, Virtanen KA, Koivumäki M, Löyttyniemi E, Nuutila P, Kalliokoski KK, Hannukainen JC (2018) Exercise training decreases pancreatic fat content and improves beta cell function regardless of baseline glucose tolerance: a randomised controlled trial. Diabetologia 61(8):1817–1828
Ibrahim NA, Khaled DM (2014) Histological and immunohistochemical study on human placental tissue in normal pregnancy and preeclampsia. Cell Biol 2:72–80
Kadiiska M et al (2005) Biomarkers of oxidative stress study II: are oxidation products of lipids, proteins, and DNA markers of CCl4 poisoning? Free Radic Biol Med 38:698–710
Kim HG, Kim JY, Gim MG, Lee JM, Chung DK (2008) Mechanical stress induces tumor necrosis factor-{alpha} production through Ca2+ release-dependent TLR2 signaling. Am J Phys Cell Phys 295:C432–C439. https://doi.org/10.1152/ajpcell.00085.2008
Koo K-M, Kim C-J (2018) The effect of the type of physical activity on the perceived stress level in people with activity limitations. J Exerc Rehabil 14:361–366. https://doi.org/10.12965/jer.1836164.082
Lavie CJ, Milani RV, Artham SM, Patel DA, Ventura HO (2009) The obesity paradox, weight loss, and coronary disease. Am J Med 122:1106–1114. https://doi.org/10.1016/j.amjmed.2009.06.006
Leeuwenburgh C, Fiebig R, Chandwaney R, Ji LL (1994) Aging and exercise training in skeletal muscle: responses of glutathione and antioxidant enzyme systems. Am J Phys 267:R439–R445. https://doi.org/10.1152/ajpregu.1994.267.2.R439
Lima FD, Stamm DN, Della-Pace ID, Dobrachinski F, de Carvalho NR, Royes LFF, Soares FA, Rocha JB, González-Gallego J, Bresciani G (2013) Swimming training induces liver mitochondrial adaptations to oxidative stress in rats submitted to repeated exhaustive swimming bouts. PLoS One 8:e55668. https://doi.org/10.1371/journal.pone.0055668
Liu J, Xia Q, Zhang Q, Li H, Zhang J, Li A, Xiu R (2009) Peroxisome proliferator-activated receptor-gamma ligands 15-deoxy-delta(12,14)-prostaglandin J2 and pioglitazone inhibit hydroxyl peroxide-induced TNF-alpha and lipopolysaccharide-induced CXC chemokine expression in neonatal rat cardiac myocytes. Shock 32:317–324. https://doi.org/10.1097/SHK.0b013e31819c374c
Liu Y-Z, Wang Y-X, Jiang C-L (2017) Inflammation: the common pathway of stress-related diseases. Front Hum Neurosci 11:316. https://doi.org/10.3389/fnhum.2017.00316
Majka DS, Chang RW, Vu THT, Palmas W, Geffken DF, Ouyang P, Ni H, Liu K (2009) Physical activity and high-sensitivity C-reactive protein: the multi-ethnic study of atherosclerosis. Am J Prev Med 36:56–62
Marosi K, Bori Z, Hart N, Sárga L, Koltai E, Radák Z, Nyakas C (2012) Long-term exercise treatment reduces oxidative stress in the hippocampus of aging rats. Neuroscience 226:21–28. https://doi.org/10.1016/j.neuroscience.2012.09.001
Mazzon E, Cuzzocrea S (2008) Role of TNF-alpha in ileum tight junction alteration in mouse model of restraint stress. Am J Physiol Gastrointest Liver Physiol 294:G1268–G1280. https://doi.org/10.1152/ajpgi.00014.2008
Miller AH, Maletic V, Raison CL (2009) Inflammation and its discontents: the role of cytokines in the pathophysiology of major depression. Biol Psychiatry 65:732–741
Munhoz C et al (2004) TNF-alpha accounts for short-term persistence of oxidative status in rat brain after two weeks of repeated stress. Eur J Neurosci 20:1125–1130. https://doi.org/10.1111/j.1460-9568.2004.03560.x
Muniraj T, Dang S, Pitchumoni CS (2015) PANCREATITIS OR NOT?—elevated lipase and amylase in ICU patients. J Crit Care 30:1370–1375. https://doi.org/10.1016/j.jcrc.2015.08.020
Navarro A, Gomez C, Lopez-Cepero JM, Boveris A (2004) Beneficial effects of moderate exercise on mice aging: survival, behavior, oxidative stress, and mitochondrial electron transfer. American journal of physiology regulatory, integrative and comparative physiology. 286:R505–R511. https://doi.org/10.1152/ajpregu.00208.2003
Pavlovic S et al (2008) Further exploring the brain-skin connection: stress worsens dermatitis via substance P-dependent neurogenic inflammation in mice. J Invest Dermatol 128:434–446. https://doi.org/10.1038/sj.jid.5701079
Piroli GG, Grillo CA, Reznikov LR, Adams S, McEwen BS, Charron MJ, Reagan LP (2007) Corticosterone impairs insulin-stimulated translocation of GLUT4 in the rat hippocampus. Neuroendocrinology 85:71–80
Puterman E, Prather AA, Epel EE, Loharuka S, Adler NE, Laraia B, Tomiyama AJ (2014) Exercise mitigates chronic stress effects on BMI trajectories in girls aged 10 to 19: longitudinal findings from the NHLBI growth and health study. Am Heart Assoc, Dallas
Rafacho A, Ribeiro DL, Boschero AC, Taboga SR, Bosqueiro JR (2008) Increased pancreatic islet mass is accompanied by activation of the insulin receptor substrate-2/serine-threonine kinase pathway and augmented cyclin D2 protein levels in insulin-resistant rats. Int J Exp Pathol 89:264–275
Rafacho A, Cestari TM, Taboga SR, Boschero AC, Bosqueiro JR (2009) High doses of dexamethasone induce increased β-cell proliferation in pancreatic rat islets. Am J of Physiol Endocrinol Metab 296:E681–E689
Rafacho A et al (2010) Glucocorticoids in vivo induce both insulin hypersecretion and enhanced glucose sensitivity of stimulus-secretion coupling in isolated rat islets. Endocrinology 151:85–95
Rafacho A, Abrantes J, Ribeiro D, Paula F, Pinto M, Boschero A, Bosqueiro JR (2011) Morphofunctional alterations in endocrine pancreas of short- and long-term dexamethasone-treated rats. Horm Metab Res 43(4):275–281
Rawal S, Huang H, Novikova L, Hamedi T, Smirnova I, Stehno-Bittel L (2013) Effects of exercise on pancreatic islets in Zucker diabetic fatty rats. J Diabetes Metab 10:2
Raza H, John A, Shafarin J, Howarth FC (2016) Exercise-induced alterations in pancreatic oxidative stress and mitochondrial function in type 2 diabetic Goto-Kakizaki rats. Phys Rep 4:e12751. https://doi.org/10.14814/phy2.12751
Riccillo FL, Bracamonte M, Console G, Gómez Dumm C (2004) Histomorphological and quantitative immunohistochemical changes in the rat pancreas during aging. Biocell 28:127–134
Rohleder N (2014) Stimulation of systemic low-grade inflammation by psychosocial stress. Psychosom Med 76:181–189
Rosmond R (2005) Role of stress in the pathogenesis of the metabolic syndrome. Psychoneuroendocrinology 30:1–10
Şahin E, Gümüşlü S (2004) Cold-stress-induced modulation of antioxidant defence: role of stressed conditions in tissue injury followed by protein oxidation and lipid peroxidation. Int J Biometeorol 48:165–171
Sapolsky RM, Romero LM, Munck AU (2000) How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev 21:55–89
Seo DY, Lee SR, Kim N, Ko KS, Rhee BD, Han J (2014) Humanized animal exercise model for clinical implication. Pflugers Arch 466:1673–1687. https://doi.org/10.1007/s00424-014-1496-0
Shidham V, Chivukula M, Basir Z, Shidham G (2001) Evaluation of crystals in formalin-fixed, paraffin-embedded tissue sections for the differential diagnosis of pseudogout, gout, and tumoral calcinosis. Mod Pathol 14:806
Shiroya Y, Minato K (2015) Beneficial effects of physical exercise on the exocrine pancreas. J Phys Fitness Sports Med 4:307–313. https://doi.org/10.7600/jpfsm.4.307
Simioni C, Zauli G, Martelli AM, Vitale M, Sacchetti G, Gonelli A, Neri LM (2018) Oxidative stress: role of physical exercise and antioxidant nutraceuticals in adulthood and aging. Oncotarget 9:17181–17198. https://doi.org/10.18632/oncotarget.24729
Sloan RP, Shapiro PA, Demeersman RE, PS MK, Tracey KJ, Slavov I, Fang Y, Flood PD (2007) Aerobic exercise attenuates inducible TNF production in humans. J Appl Physiol (1985) 103:1007–1011. https://doi.org/10.1152/japplphysiol.00147.2007
Smith RC, Southwell-Keely J, Chesher D (2005) Should serum pancreatic lipase replace serum amylase as a biomarker of acute pancreatitis? ANZ J Surg 75:399–404
Soliman NBE (2012) Effect of chronic immobilization stress on the pancreatic structure and the possible protective role of testosterone administration in male albino rats. Egyptian Journal of Histology 35:448–457. https://doi.org/10.1097/01.EHX.0000418064.05379.e2
Starnes JW, Taylor RP, Park Y (2003) Exercise improves postischemic function in aging hearts. Am J Phys Heart Circ Phys 285:H347–H351. https://doi.org/10.1152/ajpheart.00952.2002
Stefanov T, Vekova A, Bonova I, Tzvetkov S, Kurktschiev D, Bluher M, Temelkova-Kurktschiev T (2013) Effects of supervised vs non-supervised combined aerobic and resistance exercise programme on cardiometabolic risk factors. Cent Eur J Public Health 21:8–16
Suvarna KS, Layton C, Bancroft JD (2018) Bancroft's theory and practice of histological techniques. Elsevier Health Sciences, Amsterdam
Suvarna S, Layton C, Bancroft J The hematoxylins and eosinBancroft's Theory and Practice of Histological Techniques, 7th edn. Churchill Livingstone, London, pp 178–179
Takács T, Czakó L, Morschl É, László F, Tiszlavicz L, Rakonczay Z Jr, Lonovics J (2002) The role of nitric oxide in edema formation in L-arginine-induced acute pancreatitis. Pancreas 25:277–282
Teixeira de Lemos E et al (2011, 2011) Differential effects of acute (extenuating) and chronic (training) exercise on inflammation and oxidative stress status in an animal model of type 2 diabetes mellitus. Mediat Inflamm:253061. https://doi.org/10.1155/2011/253061
ter Steege RW, Kolkman JJ (2012) Review article: the pathophysiology and management of gastrointestinal symptoms during physical exercise, and the role of splanchnic blood flow. Aliment Pharmacol Ther 35:516–528. https://doi.org/10.1111/j.1365-2036.2011.04980.x
Timmerman KL, Flynn MG, Coen PM, Markofski MM, Pence BD (2008) Exercise training-induced lowering of inflammatory (CD14+CD16+) monocytes: a role in the anti-inflammatory influence of exercise? J Leukoc Biol 84:1271–1278. https://doi.org/10.1189/jlb.0408244
Tsatsoulis A, Fountoulakis S (2006) The protective role of exercise on stress system dysregulation and comorbidities. Ann N Y Acad Sci 1083:196–213
Tulic MK, Sly PD, Andrews D, Crook M, Davoine F, Odemuyiwa SO, Charles A, Hodder ML, Prescott SL, Holt PG, Moqbel R (2009) Thymic indoleamine 2, 3-dioxygenase-positive eosinophils in young children: potential role in maturation of the naive immune system. Am J Pathol 175:2043–2052
Van Raalte D, Ouwens D, Diamant M (2009) Novel insights into glucocorticoid-mediated diabetogenic effects: towards expansion of therapeutic options? Eur J Clin Investig 39:81–93
Viboolvorakul S, Niimi H, Wongeak-in N, Eksakulkla S, Patumraj S (2009) Increased capillary vascularity in the femur of aged rats by exercise training. Microvasc Res 78:459–463. https://doi.org/10.1016/j.mvr.2009.07.003
Yeh SH, Chuang H, Lin LW, Hsiao CY, Wang PW, Liu RT, Yang KD (2009) Regular tai chi chuan exercise improves T cell helper function of patients with type 2 diabetes mellitus with an increase in T-bet transcription factor and IL-12 production. Br J Sports Med 43:845–850. https://doi.org/10.1136/bjsm.2007.043562
Yu Z et al (2009) Associations of physical activity with inflammatory factors, adipocytokines, and metabolic syndrome in middle-aged and older Chinese people. Circulation 119:2969–2977. https://doi.org/10.1161/circulationaha.108.833574
Zardooz H, Zahediasl S, Rostamkhani F, Farrokhi B, Nasiraei S, Kazeminezhad B, Gholampour R (2012) Effects of acute and chronic psychological stress on isolated islets' insulin release. EXCLI J 11:163
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elbassuoni, E.A., Abdel Hafez, S.M. Impact of chronic exercise on counteracting chronic stress-induced functional and morphological pancreatic changes in male albino rats. Cell Stress and Chaperones 24, 567–580 (2019). https://doi.org/10.1007/s12192-019-00988-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-019-00988-y