Abstract
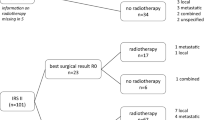
Metastatic synovial sarcoma (SS) is associated with poor prognosis. Nevertheless, data addressing prognostic factors for patients with metastatic disease are very limited. We identified patients with SS who presented with or developed metastases at our institution from January 2000 to October 2012. Potential demographic and disease-related factors were analyzed for possible influence on survival. A second analysis for patients who received chemotherapy was undertaken to assess efficacy of first-line regimens. Thirty-three patients with metastatic SS were included in this analysis. The following factors were associated with inferior overall survival (OS); age >30 years, presence of extra-pulmonary metastases, lymph node (LN) involvement, presence of uncontrolled primary site, and treatment not including pulmonary metastasectomy. Multivariate analysis identified LN metastases (HR 6.06, 95 % CI 1.18–31) and extra-pulmonary metastases (HR 4.06, 95 % CI 1.22–13.57) as the only independent factors associated with inferior OS. Assessment of efficacy of first-line chemotherapy showed superiority in progression-free survival (PFS) for ifosfamide-containing regimens versus non-ifosfamide-containing regimens (median PFS of 8.3 and 2.5 months, respectively, p = 0.002). No such significant difference in PFS was detected for comparison between doxorubicin- and non-doxorubicin-containing regimens (p = 0.45). The current study highlights that the pattern of metastases at first detection of metastatic disease is an important determinant of survival. Future studies evaluating therapeutic strategies for metastatic SS should address the comparability of those factors among study arms. In addition, our results suggest that high-dose ifosfamide should be an integral component of first-line chemotherapy regimen.


Similar content being viewed by others

References
Siegel HJ, Sessions W, Casillas MA Jr, Said-Al-Naief N, Lander PH, Lopez-Ben R. Synovial sarcoma: clinicopathologic features, treatment, and prognosis. Orthopedics. 2007;30:1020–7.
Weitz J, Antonescu CR, Brennan MF. Localized extremity soft tissue sarcoma: improved knowledge with unchanged survival over time. J Clin Oncol. 2003;21:2719–25.
Ryan JR, Baker LH, Benjamin RS. The natural history of metastatic synovial sarcoma: experience of the Southwest Oncology group. Clin Orthop Relat Res. 1982;164:257–60.
Spurrell EL, Fisher C, Thomas JM, Judson IR. Prognostic factors in advanced synovial sarcoma: an analysis of 104 patients treated at the Royal Marsden Hospital. Ann Oncol. 2005;16(3):437–44.
Paulino AC. Synovial sarcoma prognostic factors and patterns of failure. Am J Clin Oncol. 2004;27(2):122–7.
Palmerini E, Staals EL, Alberghini M, et al. Synovial sarcoma: retrospective analysis of 250 patients treated at a single institution. Cancer. 2009;115(13):2988–98.
Ferrari A, De Salvo GL, Dall’Igna P, et al. Salvage rates and prognostic factors after relapse in children and adolescents with initially localised synovial sarcoma. Eur J Cancer. 2012;48(18):3448–55.
Buck P, Mickelson MR, Bonfiglio M. Synovial sarcoma: a review of 33 cases. Clin Orthop Relat Res. 1981;156:211–5.
Bergh P, Meis-Kindblom JM, Gherlinzoni F, et al. Synovial sarcoma: identification of low and high risk groups. Cancer. 1999;85:2596–607.
Hajdu SI, Shiu MH, Fortner JG. Tendosynovial sarcoma: a clinicopathological study of 136 cases. Cancer. 1977;39(3):1201–17.
Sleijfer S, Ouali M, van Glabbeke M, et al. Prognostic and predictive factors for outcome to first-line ifosfamide-containing chemotherapy for adult patients with advanced soft tissue sarcomas: an exploratory, retrospective analysis on large series from the European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group (EORTC-STBSG). Eur J Cancer. 2010;46(1):72–83.
Bokemeyer C, Franzke A, Hartmann JT, et al. A phase I/II study of sequential, dose-escalated, high dose ifosfamide plus doxorubicin with peripheral blood stem cell support for the treatment of patients with advanced soft tissue sarcomas. Cancer. 1997;80(7):1221–7.
Rosen G, Forscher C, Lowenbraun S, et al. Synovial sarcoma. Uniform response of metastases to high dose ifosfamide. Cancer. 1994;73(10):2506–11.
Przybyl J, Jurkowska M, Rutkowski P, Debiec-Rychter M, Siedlecki JA. Downstream and intermediate interactions of synovial sarcoma-associated fusion oncoproteins and their implication for targeted therapy. Sarcoma. 2012;2012:249219. Doi:10.1155/2012/249219. Epub 2012 Mar 25.
Kekeeva TV, Riazantseva AA, Zavalishina LÉ, et al. Analysis of SYT/SSX1 and SYT/SSX2 fusion genes from synovial sarcoma. [Article in Russian]. Mol Biol (Mosk). 2011;45(5):840–4.
Srinivasan R, Gautam U, Gupta R, Rajwanshi A, Vasistha RK. Synovial sarcoma: diagnosis on fine-needle aspiration by morphology and molecular analysis. Cancer. 2009;117(2):128–36.
Eilber FC, Dry SM. Diagnosis and management of synovial sarcoma. J Surg Oncol. 2008;97(4):314–20.
Thomas DG, Giordano TJ, Sanders D, et al. Expression of receptor tyrosine kinases epidermal growth factor receptor and HER-2/neu in synovial sarcoma. Cancer. 2005;103(4):830–8.
Tawbi H, Thomas D, Lucas DR, et al. Epidermal growth factor receptor expression and mutational analysis in synovial sarcomas and malignant peripheral nerve sheath tumors. Oncologist. 2008;13(4):459–66. doi:10.1634/theoncologist.2007-0166.
Hosaka S, Horiuchi K, Yoda M, et al. A novel multi-kinase inhibitor pazopanib suppresses growth of synovial sarcoma cells through inhibition of the PI3 K-AKT pathway. J Orthop Res. 2012;30(9):1493–8. Doi:10.1002/jor.22091. Epub 2012 Feb 22.
Peng CL, Guo W, Ji T, et al. Sorafenib induces growth inhibition and apoptosis in human synovial sarcoma cells via inhibiting the RAF/MEK/ERK signaling pathway. Cancer Biol Ther. 2009;8(18):1729–36.
Schöffski P. Pazopanib in the treatment of soft tissue sarcoma. Expert Rev Anticancer Ther. 2012;12(6):711–23. doi:10.1586/era.12.41.
Sleijfer S, Ray-Coquard I, Papai Z, et al. Pazopanib, a multikinase angiogenesis inhibitor, in patients with relapsed or refractory advanced soft tissue sarcoma: a phase II study from the European organisation for research and treatment of cancer-soft tissue and bone sarcoma group (EORTC study 62043). J Clin Oncol. 2009;27(19):3126–32.
Deeks ED. Pazopanib: in advanced soft tissue sarcoma. Drugs. 2012;72(16):2129–40.
van der Graaf WT, Blay JY, Chawla SP, et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2012;379(9829):1879–86.
Fukukawa C, Hanaoka H, Nagayama S, et al. Radioimmunotherapy of human synovial sarcoma using a monoclonal antibody against FZD10. Cancer Sci. 2008;99(2):432–40.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salah, S., Yaser, S., Salem, A. et al. Factors influencing survival in metastatic synovial sarcoma: importance of patterns of metastases and the first-line chemotherapy regimen. Med Oncol 30, 639 (2013). https://doi.org/10.1007/s12032-013-0639-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0639-z