Abstract
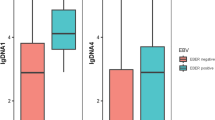
T/natural killer-cell lymphoma (T/NKCL) is a heterogeneous group of lymphoma and has a higher incidence in Asia than in Western countries. T/NKCL is presented with various clinicopathologic features, and in general, associated with a poor clinical outcome. This study aims to analyze the clinical prognostic factors in patients with T/NKCL. From January 1999 to December 2009, a total of 170 patients with T/NKCL, except mycosis fungoides, were included in this retrospective study. The diagnosis was established according to World Health Organization classification. The clinical characteristics and prognostic factors were evaluated. Of the 170 patients, mainly peripheral T-cell lymphoma-unspecified (65 cases), precursor T-lymphoblastic lymphoma/leukemia (31 cases) and nasal NK/T-cell lymphoma (NKTCL, 19 cases), advanced disease (Ann Arbor stages III–IV) was presented in 68.8% and extranodal involvement was in 71.2% of the patients. According to the international prognostic index (IPI), 77 cases were categorized as high/intermediate or high-risk group. Using the prognostic index for peripheral T-cell lymphoma-unspecified (PIT), 87 cases were classified as group 3 or 4. Most of the initial regimens were CHOP (cyclophosphamide, doxorubicin, vincristine and prednisone)-based chemotherapy (87.6%). Cumulative probability of overall survival at 5 years was 43%, and the median survival time was 44.5 months. Univariate analysis revealed that factors associated with a poor outcome were poor performance status (ECOG > 1) (P = 0.001), advanced disease (P = 0.009), the presence of B symptom (P = 0.001), multiple extranodal involvement (P = 0.005), bone marrow involvement (P = 0.003), elevated lactic dehydrogenase level (P = 0.019), IPI (P < 0.001), PIT (P < 0.001), abnormal white blood cell count (P = 0.016), decreased platelet count (P = 0.005) and serum Epstein–Barr virus (EBV) IgA positivity (P = 0.016). In the multivariate analysis, PIT (P < 0.001; relative risk, 3.221; 95% CI = 2.115–4.907) and EBV serum IgA (P = 0.049; relative risk, 1.901; 95% CI = 1.002–3.606) remained independent factors predictive for overall survival. The PIT may therefore be a useful index for risk stratification in patients with T/NKCL. The serum EBV antibody test could be a simple and quick marker to predict the outcome of the patients.


Similar content being viewed by others
References
A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin’s lymphoma. The Non-Hodgkin’s Lymphoma Classification Project. Blood. 1997;89:3909–18.
Anderson JR, Armitage JO, Weisenburger DD. Epidemiology of the non-Hodgkin’s lymphomas: distributions of the major subtypes differ by geographic locations. Non-Hodgkin’s Lymphoma Classification Project. Ann Oncol. 1998;9:717–20.
Ng CS, Chan JK. Malignant lymphomas in Chinese: what is the east–west difference? Hum Pathol. 1988;19:614–5.
Rudiger T, Weisenburger DD, Anderson JR, Armitage JO, Diebold J, MacLennan KA, Nathwani BN, Ullrich F, Muller-Hermelink HK. Peripheral T-cell lymphoma (excluding anaplastic large-cell lymphoma): results from the Non-Hodgkin’s Lymphoma Classification Project. Ann Oncol. 2002;13:140–9.
Coiffier B, Brousse N, Peuchmaur M, Berger F, Gisselbrecht C, Bryon PA, Diebold J. Peripheral T-cell lymphomas have a worse prognosis than B-cell lymphomas: a prospective study of 361 immunophenotyped patients treated with the LNH-84 regimen. The GELA (Groupe d’Etude des Lymphomes Agressives). Ann Oncol. 1990;1:45–50.
Gisselbrecht C, Gaulard P, Lepage E, Coiffier B, Briere J, Haioun C, Cazals-Hatem D, Bosly A, Xerri L, Tilly H, et al. Prognostic significance of T-cell phenotype in aggressive non-Hodgkin’s lymphomas. Groupe d’Etudes des Lymphomes de l’Adulte (GELA). Blood. 1998;92:76–82.
Melnyk A, Rodriguez A, Pugh WC, Cabannillas F. Evaluation of the Revised European-American Lymphoma classification confirms the clinical relevance of immunophenotype in 560 cases of aggressive non-Hodgkin’s lymphoma. Blood. 1997;89:4514–20.
Armitage JO, Vose JM, Linder J, Weisenburger D, Harrington D, Casey J, Bierman P, Sorensen S, Hutchins M, Moravec DF, et al. Clinical significance of immunophenotype in diffuse aggressive non-Hodgkin’s lymphoma. J Clin Oncol. 1989;7:1783–90.
Grogan TM, Fielder K, Rangel C, Jolley CJ, Wirt DP, Hicks MJ, Miller TP, Brooks R, Greenberg B, Jones S. Peripheral T-cell lymphoma: aggressive disease with heterogeneous immunotypes. Am J Clin Pathol. 1985;83:279–88.
A predictive model for aggressive non-Hodgkin’s lymphoma. The International Non-Hodgkin’s Lymphoma Prognostic Factors Project. N Engl J Med. 1993;329:987–94.
Gallamini A, Stelitano C, Calvi R, Bellei M, Mattei D, Vitolo U, Morabito F, Martelli M, Brusamolino E, Iannitto E, et al. Peripheral T-cell lymphoma unspecified (PTCL-U): a new prognostic model from a retrospective multicentric clinical study. Blood. 2004;103:2474–9.
Escalon MP, Liu NS, Yang Y, Hess M, Walker PL, Smith TL, Dang NH. Prognostic factors and treatment of patients with T-cell non-Hodgkin lymphoma: the M. D. Anderson Cancer Center experience. Cancer. 2005;103:2091–8.
Park BB, Ryoo BY, Lee JH, Kwon HC, Yang SH, Kang HJ, Kim HJ, Oh SY, Ko YH, Huh JR, et al. Clinical features and treatment outcomes of angioimmunoblastic T-cell lymphoma. Leuk Lymphoma. 2007;48:716–22.
Rodriguez J, Conde E, Gutierrez A, Arranz R, Leon A, Marin J, Bendandi M, Albo C, Caballero MD. The results of consolidation with autologous stem-cell transplantation in patients with peripheral T-cell lymphoma (PTCL) in first complete remission: the Spanish Lymphoma and Autologous Transplantation Group experience. Ann Oncol. 2007;18:652–7.
Beltran B, Quinones P, Morales D, Cotrina E, Castillo JJ. Different prognostic factors for survival in acute and lymphomatous adult T-cell leukemia/lymphoma. Leuk Res. 2011;35:334–9.
Engels EA. Infectious agents as causes of non-Hodgkin lymphoma. Cancer Epidemiol Biomarkers Prev. 2007;16:401–4.
Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer. 2006;118:3030–44.
Young LS, Rickinson AB. Epstein-Barr virus: 40 years on. Nat Rev Cancer. 2004;4:757–68.
Anagnostopoulos I, Hummel M, Finn T, Tiemann M, Korbjuhn P, Dimmler C, Gatter K, Dallenbach F, Parwaresch MR, Stein H. Heterogeneous Epstein-Barr virus infection patterns in peripheral T-cell lymphoma of angioimmunoblastic lymphadenopathy type. Blood. 1992;80:1804–12.
Dupuis J, Emile JF, Mounier N, Gisselbrecht C, Martin-Garcia N, Petrella T, Bouabdallah R, Berger F, Delmer A, Coiffier B, et al. Prognostic significance of Epstein-Barr virus in nodal peripheral T-cell lymphoma, unspecified: A Groupe d’Etude des Lymphomes de l’Adulte (GELA) study. Blood. 2006;108:4163–9.
Kanavaros P, Lescs MC, Briere J, Divine M, Galateau F, Joab I, Bosq J, Farcet JP, Reyes F, Gaulard P. Nasal T-cell lymphoma: a clinicopathologic entity associated with peculiar phenotype and with Epstein-Barr virus. Blood. 1993;81:2688–95.
Manns A, Hisada M, La Grenade L. Human T-lymphotropic virus type I infection. Lancet. 1999;353:1951–8.
Vose J, Armitage J, Weisenburger D. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes. J Clin Oncol. 2008;26:4124–30.
Liang X, Bi S, Yang W, Wang L, Cui G, Cui F, Zhang Y, Liu J, Gong X, Chen Y, et al. Epidemiological serosurvey of hepatitis B in China–declining HBV prevalence due to hepatitis B vaccination. Vaccine. 2009;27:6550–7.
Marcucci F, Mele A, Spada E, Candido A, Bianco E, Pulsoni A, Chionne P, Madonna E, Cotichini R, Barbui A, et al. High prevalence of hepatitis B virus infection in B-cell non-Hodgkin’s lymphoma. Haematologica. 2006;91:554–7.
Nath A, Agarwal R, Malhotra P, Varma S. Prevalence of hepatitis B virus infection in non-Hodgkin lymphoma: a systematic review and meta-analysis. Intern Med J. 2010;40:633–41.
Lee Y, Uhm JE, Lee HY, Park MJ, Kim H, Oh SJ, Jang JH, Kim K, Jung CW, Ahn YC, et al. Clinical features and prognostic factors of patients with “peripheral T cell lymphoma, unspecified”. Ann Hematol. 2009;88:111–9.
Shi YX, Peng RJ, Lin SX, Wu QL, Lin TY, Sun XF, Huang HQ, Xia ZJ, Li YH, Xu RH, et al. A survival of 103 cases of T-cell non-Hodgkin lymphoma. Zhonghua Nei Ke Za Zhi. 2007;46:1000–2.
Sonnen R, Schmidt WP, Muller-Hermelink HK, Schmitz N. The international prognostic index determines the outcome of patients with nodal mature T-cell lymphomas. Br J Haematol. 2005;129:366–72.
Rudiger T, Weisenburger DD, Anderson JR, Armitage JO, Diebold J, MacLennan KA, Nathwani BN, Ullrich F, Muller-Hermelink HK. Peripheral T-cell lymphoma (excluding anaplastic large-cell lymphoma): results from the Non-Hodgkin’s Lymphoma Classification Project. Ann Oncol. 2002;13:140.
Niitsu N, Okamoto M, Nakamine H, Aoki S, Motomura S, Hirano M. Clinico-pathologic features and outcome of Japanese patients with peripheral T-cell lymphomas. Hematol Oncol. 2008;26:152–8.
d’Amore F, Johansen P, Houmand A, Weisenburger DD, Mortensen LS. Epstein-Barr virus genome in non-Hodgkin’s lymphomas occurring in immunocompetent patients: highest prevalence in nonlymphoblastic T-cell lymphoma and correlation with a poor prognosis. Danish Lymphoma Study Group, LYFO. Blood. 1996;87:1045–55.
Went P, Agostinelli C, Gallamini A, Piccaluga PP, Ascani S, Sabattini E, Bacci F, Falini B, Motta T, Paulli M, et al. Marker expression in peripheral T-cell lymphoma: a proposed clinical-pathologic prognostic score. J Clin Oncol. 2006;24:2472–9.
Hirose Y, Masaki Y, Sawaki T, Shimoyama K, Karasawa H, Kawabata H, Fukushima T, Ogawa N, Wano Y, Umehara H. Association of Epstein-Barr virus with human immunodeficiency virus-negative peripheral T-cell lymphomas in Japan. Eur J Haematol. 2006;76:109–18.
Liang R. Diagnosis and management of primary nasal lymphoma of T-cell or NK-cell origin. Clin Lymphoma. 2000;1:33–7. discussion 38.
Mitarnun W, Suwiwat S, Pradutkanchana J, Saechan V, Ishida T, Takao S, Mori A. Epstein-Barr virus-associated peripheral T-cell and NK-cell proliferative disease/lymphoma: clinicopathologic, serologic, and molecular analysis. Am J Hematol. 2002;70:31–8.
Nikoskelainen J, Neel EU, Stevens DA. Epstein-Barr virus-specific serum immunoglobulin A as an acute-phase antibody in infectious mononucleosis. J Clin Microbiol. 1979;10:75–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Peng-Peng Xu and Yan Wang equally contributed to this work.
Rights and permissions
About this article
Cite this article
Xu, PP., Wang, Y., Shen, Y. et al. Prognostic factors of Chinese patients with T/NK-cell lymphoma: a single institution study of 170 patients. Med Oncol 29, 2176–2182 (2012). https://doi.org/10.1007/s12032-011-0011-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-0011-0