Abstract
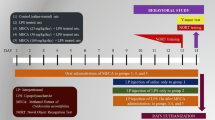
Mainly found in brussels sprouts, broccoli, and black mustard seeds, sinigrin (2-propenyl glucosinolate) has enjoyed some attention currently for its effects on health and disease prevention. The present research design is aimed at investigating the effects of sinigrin on inflammation, oxidative stress (OS) and memory. Randomly, six groups of male Wistar rats were categorized into the control and experimental groups. The experimental groups were treated with sinigrin (10 and 20 mg/kg, orally). The control positive group was given the pentylenetetrazole (PTZ) treatment and the control negative one was given normal saline. All groups were kindled by the sub-threshold dose (35 mg/kg, i.p.) of PTZ for 12 times in one month. When the kindling procedure was done, the seizure behaviors and the behavioral function were evaluated. For cognitive parameters, the shuttle box test was employed. When the experiment was terminated, the rats were euthanized and their blood serum as well as brain samples were isolated for respective measuring of OS and gene expression parameters. The treatment with sinigrin significantly delayed the appearance of the seizure symptoms in comparison to that of the PTZ group. It also significantly increased the memory parameters like retention latency and the total time having been spent in the light compartment in the epileptic rats. In addition, sinigrin increased the superoxide dismutase and catalase levels. Treatment with sinigrin suppressed the Il1b and Nlrp3 gene expression at hippocampal level. In sum, sinigrin prevents inflammation, OS and memory impairment against the PTZ-kindling epilepsy in rats.





Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ahmadian, S. R., Ghasemi-Kasman, M., Pouramir, M., & Sadeghi, F. (2019). Arbutin attenuates cognitive impairment and inflammatory response in pentylenetetrazol-induced kindling model of epilepsy. Neuropharmacology, 146, 117–127.
Alyu, F., & Dikmen, M. (2017). Inflammatory aspects of epileptogenesis: contribution of molecular inflammatory mechanisms. Acta Neuropsychiatrica. Officieel Wetenschappelijk Orgaan van Het IGBP (Interdisciplinair Genootschap voor Biologische Psychiatrie), 29(1), 1–16.
Azizi, V., Allahyari, F., & Hosseini, A. (2018). The antiepileptic and neuroprotective effect of the Buxus hyrcana Pojark hydroethanolic extract against the pentylentetrazol induced model of the seizures in the male rats. International Journal of Neuroscience, 128(10), 913–919. https://doi.org/10.1080/00207454.2018.1443925
Berg, A. T., Altalib, H. H., & Devinsky, O. (2017). Psychiatric and behavioral comorbidities in epilepsy: A critical reappraisal. Epilepsia, 58(7), 1123–1130.
Bonda, D. J., Lee, H.-P., Lee, H.-G., Friedlich, A. L., Perry, G., Zhu, X., et al. (2010). Novel therapeutics for Alzheimer’s disease: An update. Current Opinion in Drug Discovery & Development, 13(2), 235.
Cerri, C., Caleo, M., & Bozzi, Y. (2017). Chemokines as new inflammatory players in the pathogenesis of epilepsy. Epilepsy Research, 136, 77–83.
Church, L. D., Savic, S., & McDermott, M. F. (2008). Long term management of patients with cryopyrin-associated periodic syndromes (CAPS): Focus on rilonacept (IL-1 Trap). Biologics, 2(4), 733.
Dong, Y., Li, S., Lu, Y., Li, X., Liao, Y., Peng, Z., et al. (2020). Stress-induced NLRP3 inflammasome activation negatively regulates fear memory in mice. Journal of Neuroinflammation, 17(1), 1–16.
Ghosh, S., Sinha, J. K., Khan, T., Devaraju, K. S., Singh, P., Vaibhav, K., et al. (2021). Pharmacological and therapeutic approaches in the treatment of epilepsy. Biomedicines. https://doi.org/10.3390/biomedicines9050470
Gomar, A., Hosseini, A., & Mirazi, N. (2014). Preventive effect of Rubus fruticosus on learning and memory impairment in an experimental model of diabetic neuropathy in male rats. PharmaNutrition, 2(4), 155–160.
Han, T., Qin, Y., Mou, C., Wang, M., Jiang, M., & Liu, B. (2016). Seizure induced synaptic plasticity alteration in hippocampus is mediated by IL-1β receptor through PI3K/Akt pathway. American Journal of Translational Research, 8(10), 4499.
Helmstaedter, C., & Witt, J.-A. (2017). Epilepsy and cognition–a bidirectional relationship? Seizure, 49, 83–89.
Hoppe, C., Elger, C. E., & Helmstaedter, C. (2007). Long-term memory impairment in patients with focal epilepsy. Epilepsia, 48, 26–29.
Ippoushi, K., Takeuchi, A., & Azuma, K. (2010). Sinigrin suppresses nitric oxide production in rats administered intraperitoneally with lipopolysaccharide. Food Chemistry, 120(4), 1119–1121.
Jie, M., Cheung, W. M., Yu, V., Zhou, Y., Tong, P. H., & Ho, J. W. (2014). Anti-proliferative activities of sinigrin on carcinogen-induced hepatotoxicity in rats. PLoS ONE, 9(10), e110145. https://doi.org/10.1371/journal.pone.0110145
Jo, E.-K., Kim, J. K., Shin, D.-M., & Sasakawa, C. (2016). Molecular mechanisms regulating NLRP3 inflammasome activation. Cellular & Molecular Immunology, 13(2), 148–159.
Josephson, C. B., & Jetté, N. (2017). Psychiatric comorbidities in epilepsy. International Review of Psychiatry, 29(5), 409–424.
Kalayci, R., Kaya, M., Ahishali, B., Arican, N., Elmas, I., & Kucuk, M. (2006). Long-term L-NAME treatment potentiates the blood–brain barrier disruption during pentylenetetrazole-induced seizures in rats. Life Sciences, 79(1), 16–20.
Kelley, N., Jeltema, D., Duan, Y., & He, Y. (2019). The NLRP3 inflammasome: An overview of mechanisms of activation and regulation. International Journal of Molecular Sciences, 20(13), 3328.
Kuhlmann, L., Lehnertz, K., Richardson, M. P., Schelter, B., & Zaveri, H. P. (2018). Seizure prediction—ready for a new era. Nature Reviews: Neurology, 14(10), 618–630.
Kumar, R., Arora, R., Agarwal, A., & Gupta, Y. K. (2018). Protective effect of Terminalia chebula against seizures, seizure-induced cognitive impairment and oxidative stress in experimental models of seizures in rats. Journal of Ethnopharmacology, 215, 124–131. https://doi.org/10.1016/j.jep.2017.12.008
Lee, H., Lee, C., Kim, J., & Pyo, S. (2014). The inhibitory effect of sinigrin on the production of inflammatory mediators induced by lipopolysaccharide in RAW 264.7 macrophages (1056.5). FASEB Journal, 28(1_supplement), 1056.1055.
Lorenz, G., Darisipudi, M. N., & Anders, H.-J. (2014). Canonical and non-canonical effects of the NLRP3 inflammasome in kidney inflammation and fibrosis. Nephrology, Dialysis, Transplantation : Official Publication of the European Dialysis and Transplant Association - European Renal Association, 29(1), 41–48.
Mazumder, A., Dwivedi, A., & Du Plessis, J. (2016). Sinigrin and its therapeutic benefits. Molecules, 21(4), 416.
Meng, X.-F., Tan, L., Tan, M.-S., Jiang, T., Tan, C.-C., Li, M.-M., et al. (2014). Inhibition of the NLRP3 inflammasome provides neuroprotection in rats following amygdala kindling-induced status epilepticus. Journal of Neuroinflammation, 11(1), 212.
Migliore, L., Fontana, I., Colognato, R., Coppede, F., Siciliano, G., & Murri, L. (2005). Searching for the role and the most suitable biomarkers of oxidative stress in Alzheimer’s disease and in other neurodegenerative diseases. Neurobiology of Aging, 26(5), 587–595.
Mishra, P., Mittal, A. K., Rajput, S. K., & Sinha, J. K. (2021a). Cognition and memory impairment attenuation via reduction of oxidative stress in acute and chronic mice models of epilepsy using antiepileptogenic Nux vomica. Journal of Ethnopharmacology, 267, 113509. https://doi.org/10.1016/j.jep.2020.113509
Mishra, P., Sinha, J. K., & Rajput, S. K. (2021b). Efficacy of Cicuta virosa medicinal preparations against pentylenetetrazole-induced seizures. Epilepsy & Behavior, 115, 107653. https://doi.org/10.1016/j.yebeh.2020.107653
Nigar, S., Pottoo, F. H., Tabassum, N., Verma, S. K., & Javed, M. N. (2016). Molecular insights into the role of inflammation and oxidative stress in epilepsy. Journal of Advances in Medical and Pharmaceutical Sciences, 1–9.
Pahuja, M., Mehla, J., Reeta, K. H., Tripathi, M., & Gupta, Y. K. (2013). Effect of Anacyclus pyrethrum on pentylenetetrazole-induced kindling, spatial memory, oxidative stress and rho-kinase II expression in mice. Neurochemical Research, 38(3), 547–556. https://doi.org/10.1007/s11064-012-0947-2
Saxena, M., Saxena, J., Nema, R., Singh, D., & Gupta, A. (2013). Phytochemistry of medicinal plants. Journal of Pharmacognosy and Phytochemistry, 1(6), 168–182.
Shen, K., Mao, Q., Yin, X., Zhang, C., Jin, Y., Deng, A., et al. (2018). NLRP3 inflammasome activation leads to epileptic neurona l apoptosis. Current Neurovascular Research, 15(4), 276–281.
Shin, E.-J., Jeong, J. H., Chung, Y. H., Kim, W.-K., Ko, K.-H., Bach, J.-H., et al. (2011). Role of oxidative stress in epileptic seizures. Neurochemistry International, 59(2), 122–137.
Vezzani, A., Balosso, S., & Ravizza, T. (2019). Neuroinflammatory pathways as treatment targets and biomarkers in epilepsy. Nature Reviews: Neurology, 15(8), 459–472.
Winyard, P. G., Moody, C. J., & Jacob, C. (2005). Oxidative activation of antioxidant defence. Trends in Biochemical Sciences, 30(8), 453–461.
Wu, C., Zhang, G., Chen, L., Kim, S., Yu, J., Hu, G., et al. (2019). The role of NLRP3 and IL-1β in refractory epilepsy brain injury. Frontiers in Neurology, 10, 1418. https://doi.org/10.3389/fneur.2019.01418
Zhou, K., Shi, L., Wang, Y., Chen, S., & Zhang, J. (2016). Recent advances of the NLRP3 inflammasome in central nervous system disorders. Journal of Immunology Research, 2016(2016), 9238290. https://doi.org/10.1155/2016/9238290
Funding
None received.
Author information
Authors and Affiliations
Contributions
Conception and design of the work: FA, MR, AH, FM, SK, AH; data collection: FA, MR, AH, FM, SK, AH; analysis and interpretation of the data: FA, MR, AH, FM, SK, AH; statistical analysis: FA, MR, AH, FM, SK, AH; drafting the manuscript: FA, MR, AH, FM, SK, AH; critical revision of the manuscript: FA, MR, AH, FM, SK, AH.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
All experiments implemented in the present study observed standard ethical guidelines stipulated by European Communities Directive (2010/63/EU); they also had the approval of Shahid Beheshti University’s Local Ethics Committee (# IR.SBU.REC.1399.085).
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aghaie, F., Rajabi, M., Hosseini, A. et al. Preventive Effects of Sinigrin Against the Memory Deterioration in the Pentylenetetrazole-Kindled Male Wistar Rats: Possible Modulation of NLRP3 Pathway. Neuromol Med 24, 311–319 (2022). https://doi.org/10.1007/s12017-021-08690-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-021-08690-y