Abstract
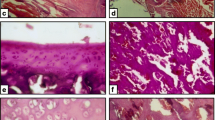
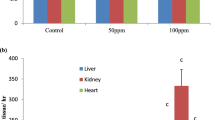
Excessive exposure to fluoride poses several detrimental effects to human health particularly the kidney which is a major organ involved in its elimination from the body. The influence of taurine on fluoride-induced renal toxicity was investigated in a co-exposure paradigm for 45 days using five groups of eight rats each. Group I rats received normal drinking water alone, group II rats were exposed to sodium fluoride (NaF) in drinking water at 15 mg/L alone, group III received taurine alone at a dose of 200 mg/kg group IV rats were co-administered with NaF and taurine (100 mg/kg), while group V rats were co-administered with NaF and taurine (200 mg/kg). Administration of taurine significantly reversed the fluoride-mediated decrease in absolute weight and organo-somatic index of the kidney in the exposed rats. Taurine significantly prevented fluoride-induced elevation in plasma urea and creatinine levels in the exposed rats. Moreover, taurine restored fluoride-mediated decrease in the circulatory concentrations of triiodothyronine, thyroxine, and the ratio of triiodothyronine to thyroxine. Taurine ameliorated fluoride-mediated decrease in renal antioxidant status by significantly enhancing the antioxidant enzyme activities as well as glutathione level in the exposed rats. Additionally, taurine inhibited fluoride-induced renal oxidative damage by markedly decreasing the hydrogen peroxide and malondialdehyde levels as well as improved the kidney architecture in the treated rats. Collectively, taurine protected against fluoride-induced renal toxicity via enhancement of thyroid gland function, renal antioxidant status, and histology in rats.







Similar content being viewed by others
Reference
Ayoob S, Gupta AK (2006) Fluoride in drinking water: a review on the status and stress effects. Crit Rev Environ Sci Technol 36:433–487
USNRC (1993) Health effects of ingested fluoride. National Research Council, National Academy Press, Washington D.C
World Health Organization (2004). Guidelines for Drinking-water Quality (GDWQ). WHO/SDE/WSH/03.04/96. Pp 1–17.
Cao SR (1992) Study on preventive and control measures on coal-combustion type endemic fluorosis in the three gorges area in China. (proceedings of the fourth National Academic Conference on endemic fluorosis. Chin J Endemic Die II(Suppl):6–21
World Health Organization (2002) Fluorides. World Health Organization, Geneva
Ding JP, Mao JQ, Gu SY (1998) Fluoride contamination and its control of lava groundwater in Guiyang dam area. Environ Protect Sci Technol 3:14–15
Wu GJ, Xu BF, Lu L (2006) Comment on fluorine pollution and prevention of Shilongba hydro-electric power station. J Kunming Univ Sci Technol 31:55–58
Saralakumari D, Ramakrishna RP (1993) Endemic fluorosis in the village Ralla Anantapuram in Andhra Pradesh: an epidemiological study. Fluoride 26:177
Atmaca N, Atmaca HT, Kanici A, Anteplioglu T (2014) Protective effect of resveratrol on sodium fluoride-induced oxidative stress, hepatotoxicity and neurotoxicity in rats. Food Chem Toxicol 70:191–197
Shashi A, Singh JP, Thapar SP (2002) Toxic effects of fluoride on rabbit kidney. Fluoride 35:38–50
Liu GY, Chai CHY, Kang SHL (2002) Effects of fluoride on the ultrastructure of thyroid in chicks. Chin J Vet Sci 22:512–514
VV P, VT D (2015) Exposure to sodium fluoride affects thyroid follicular cells in albino rats. Int J Plant, Animal and Environ Sci 5l:56–61
Karaoz E, Oncu M, Gulle K, Kanter M, Gultekin F, Karaoz S, Mumcu E (2004) Effect of chronic fluorosis on lipid peroxidation and histology of kidney tissues in first-and second-generation rats. Biol Trace Elem Res 102:199–208
Ibarra-Santana C, Ruiz-Rodríguez Mdel S, Fonseca-Leal Mdel P, Gutiérrez-Cantú FJ, Pozos-Guillén AJ (2007) Enamel hypoplasia in children with renal disease in a fluoridated area. J Clin Pediatr Dent 31:274–278
Barbier O, Arreola-Mendoza L, Del RL (2010) Molecular mechanisms of fluoride toxicity. Chem Biol Interact 188:319–333
Nabavi SF, Moghaddam AH, Eslami S, Nabavi SM (2012) Protective effects of curcumin against sodium fluoride-induced toxicity in rat kidneys. Biol Trace Elem Res 145:369–374
Wright CE, Tallan HH, Lin YY (1986) Taurine: biological update. Annu Rev Biochem 55:427–453
Huxtable RJ (1992) Physiological actions of taurine. Physiol Rev 72:101–163
Das J, Roy A, Sil PC (2012) Mechanism of the protective action of taurine in toxin and drug induced organ pathophysiology and diabetic complications: a review. Food Funct 3:1251–1264
Das J, Sil PC (2012) Taurine ameliorates alloxan-induced diabetic renal injury, oxidative stress-related signaling pathways and apoptosis in rats. Amino Acids 43:1509–1523
Chattopadhyay A, Podder S, Agarwal S, Bhattacharya S (2011) Fluoride-induced histopathology and synthesis of stress protein in liver and kidney of mice. Arch Toxicol 85:327–335
Das J, Ghosh J, Manna P, Sil PC (2012) Taurine protects rat testes against doxorubicin-induced oxidative stress as well as p53, Fas and caspase 12-mediated apoptosis. Amino Acids 42:1839–1855
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Jollow DJ, Mitchell JR, Zampaglione N, Gillette JR (1974) Bromobenzene induced liver necrosis: protective role of glutathione and evidence for 3,4 bromobenzene oxide as the hepatotoxic metabolite. Pharmacology 11:151–169
Wolff SP (1994) Ferrous ion oxidation in the presence of ferric ion indicator xylenol orange for measurement of hydroperoxides. Methods Enzymol 233:182–189
Farombi EO, Tahnteng JG, Agboola AO, Nwankwo JO, Emerole GO (2000) Chemoprevention of 2-acetylaminofluorene-induced hepatotoxicity and lipid peroxidation in rats by kolaviron-a Garcinia kola seed extract. Food Chem Toxicol 38:535–541
Misra HP, Fridovich I (1972) The role of superoxide anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Clairborne A (1995) Catalase activity. In: Greewald AR (ed) Handbook of methods for oxygen radical research. CRC Press, Boca Raton, FL, pp. 237–242
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferase. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590
Bancroft JD. Gamble M. (2008). Theory and practice of histology techniques, 6th edition. Churchill Livingstone Elsevier, Pp 83–134.
Adedara IA, Abolaji AO, Odion BE, Okwudi IJ, Omoloja AA, Farombi EO (2014) Impairment of hepatic and renal functions by 2, 5-hexanedione is accompanied by oxidative stress in rats. J Toxicol 239240:1–9
Nabavi SM, Habtemariam S, Nabavi SF, Sureda A, Daglia M, Moghaddam AH, Amani MA (2013) Protective effect of gallic acid isolated from Peltiphyllum peltatum against sodium fluoride-induced oxidative stress in rat’s kidney. Mol Cell Biochem 372:233–239
Den Hollander JG, Wulkan RW, Mantel MJ, Berghout A (2005) Correlation between severity of thyroid dysfunction and renal function. Clin Endocrinol 62:423–427
Gopinath B, Harris DC, Wall JR, Kifley A, Mitchell P (2013) Relationship between thyroid dysfunction and chronic kidney disease in community-dwelling older adults. Maturitas 75:159–164
Montenegro J, Gonzalez O, Saracho R, Aguirre R, Gonzalez O, Martinez I (1996) Changes in renal function in primary hypothyroidism. Am J Kidney Dis 27:195–198
Wang H, Yang Z, Zhou B, Gao H, Yan X, Wang J (2009) Fluoride-induced thyroid dysfunction in rats: roles of dietary protein and calcium level. Toxicol Ind Health 25:49–57
Zhan XA, Wang M, Xu ZR, Li JX (2006) Toxic effects of fluoride on kidney function and histology in young pigs. Fluoride 39:22–26
Podder S, Chattopadhyay A, Bhattacharya S (2008) In vivo suppression by fluoride of chromosome aberrations induced by mitomycin-c in mouse bone marrow cells. Fluoride 41:40–43
Podder S, Chattopadhyay A, Bhattacharya S, Ray MR (2008) Differential in vivo genotoxic effect of lower and higher concentrations of fluoride in mouse bone marrow cells. Fluoride 41:301–307
Acknowledgment
This research was done without specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that there are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Adedara, I.A., Ojuade, T.J.D., Olabiyi, B.F. et al. Taurine Ameliorates Renal Oxidative Damage and Thyroid Dysfunction in Rats Chronically Exposed to Fluoride. Biol Trace Elem Res 175, 388–395 (2017). https://doi.org/10.1007/s12011-016-0784-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-016-0784-2