Abstract
Background
Modern hip implants typically feature modular heads, which allow for easy exchange and removal from the femoral stem at the time of revision. However, owing to fretting, corrosion, or cold welding, the modular head may be difficult or impossible to separate from the underlying trunnion, especially if the implant has titanium interfaces between the head and the stem. We have repeatedly encountered difficulty removing the titanium sleeve adapter in the M2a-MagnumTM implant. Although the manufacturer warns about this complication and cases with these difficulties have been reported to the United States FDA, we believed this topic is important to study, because the frequency of difficulties in head removal is unknown and the complications related to this event have not been characterized.
Questions/purposes
We asked: (1) Do revisions of M2a-MagnumTM implants differ from those of M2a-38TM implants in terms of ease of removal of the femoral head? (2) In cases where difficulty with M2a-MagnumTM head removal occurred, was the operative time, bleeding, risk of periprosthetic fracture, or joint infection increased compared with cases where the M2a-MagnumTM head was removed without difficulties?
Methods
Between 2004 and 2014, we revised 296 THAs with metal-on-metal implants that involved M2a-MagnumTM (123) or M2a-38TM heads (88); of those, 84 were planned to include a femoral stem revision and insufficient data were available for three operations, so they were excluded from this analysis, leaving 124 THAs in the current retrospective study (70 THAs with M2a-MagnumTM and 54 THAs with M2a-38TM heads).The method of modular head removal, any difficulties removing the femoral head from the trunnion, operation time, and complications were recorded based on chart review.
Results
All the observed problems of detaching the head or taper adapter were among M2a-MagnumTM heads; there were no problems detaching the head in revisions of the M2a-38TM implant. In 29% (20 of 70) of revisions of the M2a-MagnumTM implant, the modular head could not be detached by knocking it with a punch and a mallet. Seventeen percent (12 of 70) of hips needed an unplanned stem revision owing to difficulties with head removal. In revisions of the M2a-MagnumTM implant that experienced head-removal problems, the median operative time was longer (144 minutes; range, 75–274 minutes) and bleeding was greater (725 mL; range, 300–2200 mL) compared with revisions of the M2a-MagnumTM implant without head removal problems (77 minutes, range, 33–197 minutes, p < 0.001; 475 mL, range, 50–1500 mL, p = 0.004). With the numbers available, we did not see differences in terms of the proportion of patients experiencing major complications (periprosthetic fracture or postoperative infections) between the groups (difficult versus easy; 25% [five of 20] versus 8% [four of 50]; odds ratio, 3.8 [95% CI, 0.9–16.2], p = 0.067).
Conclusions
The titanium–titanium taper junction can be very difficult to separate during revision THAs, and if not anticipated, this problem can result in larger and more complicated revision procedures in patients who have the M2a-MagnumTM implant. Although the global use of metal-on-metal implants in THAs has decreased dramatically during the last several years, many thousands remain in service and therefore still might require revision. It is crucial to be prepared with special tools, including a femoral head extraction tool and diamond saw. The patient has to be informed of the possibility of a more extensive operation than preoperatively planned.
Level of Evidence
Level III, therapeutic study.
Similar content being viewed by others
Introduction
Adverse local tissue reaction, which may lead to early failure of an implant, has been a concern for THAs with metal-on-metal (MoM) implants [1, 8, 9]. The survival rate of THAs with large-diameter MoM implants can be as low as 51% at 6 years followup [11]. Although the national guidelines in several countries recommend considering carefully further use of MoM implants [6, 7, 12, 16, 19], a large number of these THAs will be revised not only as a result of adverse local tissue reaction, but also because of dislocations, periprosthetic fractures, and infections. Although THAs with MoM implants no longer are as common as they once were [14], many of these implants remain in service and may yet need revision surgery [2].
One MoM implant that has been in common use is the M2a-MagnumTM (Biomet, Warsaw, IL, USA), which consists of a monoblock, press-fit cup articulating with a femoral head, both made of a high carbon-cobalt-chrome molybdenum head. The femoral head is connected to a neck with a modular taper adapter, which provides the option to adjust the neck length. The stem and taper adapter are made of a titanium, aluminum, and vanadium alloy (Ti-6Al-4V) [4]. The M2a-38TM (Biomet), however, has a solid, fixed 38-mm cobalt-chromium head and does not contain a separate titanium taper adapter as the M2a-MagnumTM does [5]. The M2a-38TM may be attached to the same stems and tapers as the M2a-MagnumTM. In revision THAs of MoM implants, exchange of the modular liner or revision of the whole acetabular component combined with exchange of the modular femoral head without removing the stem is usually sufficient. Generally, the modular head can be removed without problems by using a punch and a mallet. However, owing to fretting, corrosion, and cold welding, the modular head may be difficult or even impossible to remove [13]. In this regard, the titanium-on-titanium head-trunnion couple has been identified as a source of difficulty by the manufacturer [3] and by the FDA [17, 18].
In our practice, however, we repeatedly encountered difficulty removing the titanium sleeve adapter with the M2a-MagnumTM. This was not seen in other THA revisions with MoM implants, including a similar design, the M2a-38TM, from the same manufacturer. Although the manufacturer warns about this complication and cases with these difficulties have been reported to the FDA, we believe this topic is important to study because the frequency of difficulties in head removal is unknown and the complications related to this event have not been characterized.
Because the difficulties we have encountered with removal of the femoral head and adapter sleeve appeared to be unique to the M2a-MagnumTM, we asked the following: (1) Do revisions of the M2a-MagnumTM implant differ from those of the M2a-38TM in terms of ease of removal of the femoral head? (2) In cases where difficulty with M2a-MagnumTM head removal occurred, was the operation time, bleeding, risk of periprosthetic fracture, or joint infection increased compared with cases where the M2a-MagnumTM head was removed without difficulties?
Materials and Methods
Study Design and Setting
Between April 2004 and January 2012, we performed 2326 revision THAs in two tertiary-level hospitals. Of those 296 (13%) were performed in THAs with MoM implants. During that period, the most common indications for revision were aseptic loosening, infection, dislocation, periprosthetic fracture, and adverse local tissue reaction. Of this group, we looked specifically at primary THAs with MoM implants performed with a stem provided from one manufacturer with a similar type of taper (Biomet, Type 1), and either a M2a-MagnumTM or M2a-38TM modular head. After excluding three patients who had insufficient operation data and patients for whom an exchange of the femoral stem or of both components was planned, 124 remained (70 who had the M2a-MagnumTM head and 54 who had the M2a-38TM femoral head) for followup at a minimum of 1 year (median, 1.7 years; range, 1–8.7 years) (Table 1). None of the patients had died or were lost to followup less than 1 year after the revision surgery. The two groups (M2a-MagnumTM and M2a-38TM) were essentially the same in terms of age, sex, and BMI (Table 1). Information regarding the preoperative plan of the revision, performed revision, implants, operation date, perioperative bleeding, and complications were recorded retrospectively from the patient records. Preoperative serum chromium and cobalt ion levels were available for 68% (84 of 124) of the patients.
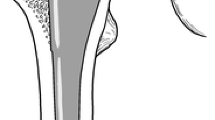
The primary outcome measure was our ability to remove the femoral head. The method of modular head or adapter removal was categorized into four groups: (1) a punch and a mallet; (2) a special femoral head extractor tool (Fig. 1); (3) a diamond saw cut; and (4) not removable. With a diamond saw the head and adapter sleeve were cut almost through (without damaging the trunnion), after which the head could be removed by levering out with an osteotome and punching the adapter sleeve with a mallet (Fig. 2).
The secondary outcome measures were operation time, blood loss, and complications during the first year after revision. Our secondary outcome variable on complications evaluated the proportion of patients who experienced periprosthetic infection or fracture. We considered infection potentially relevant, as the risk of infection may increase as the surgical procedures get longer, and periprosthetic fracture was considered because these revisions may have entailed imparting more force to the proximal femur while attempting to remove the femoral head, or extended trochanteric osteotomy had to be performed owing to an unremovable femoral head or severely damaged trunnion. The infections diagnosis was based on clinical suspicion of infection, and patients underwent débridement during which several bacteria specimen were taken from around the implant (deep specimens). The periprosthetic infection diagnosis was confirmed if the same bacteria were cultured in two or more specimens. We believe that none of the measured outcome complications was missed, because, at our institutions, we treat all periprosthetic fractures, infections, or suspicion of infection in the region.
We compared the M2a-38TM group with 54 patients versus the M2a-MagnumTM group with 70 patients. We also compared THAs that had the M2a-MagnumTM implant with easy-to-remove heads (50 patients) versus hard-to-remove heads (20 patients). If the head was easy-to-remove, it was defined as “head removal, punch” and if special instruments were used or the head could not be removed, it was defined as “head removal, other”.
Statistical Analysis
A chi-square test was used to compare sex distribution between groups with the M2a-38TM and M2a-MagnumTM femoral heads. The differences in age and BMI between groups were analyzed with a two-sample t-test. The Mann-Whitney U test was used to test the differences in operation time, blood loss, and chromium and cobalt ion levels between groups. Median differences with 95% CI were calculated using Hodges-Lehmann estimates. Logistic regression was used to compare complications between groups. The results are expressed using odd ratios (OR) with 95 CIs. A p value less than 0.05 was considered statistically significant. Statistical analyses were performed using SAS® System for Windows, Version 9.4 (SAS Institute Inc, Cary, NC, USA).
Results
No difficulty was noted with head extraction in the M2a-38TM group; all problems with detaching the head or taper adapter from the stem were among M2a-MagnumTM heads. In 71% (50 of 70) of revisions of the M2a-MagnumTM implant there were no problems with head or adapter removal. In 29% (20 of 70) of revisions of the M2a-MagnumTM implant, the modular head could not be detached by knocking it with a punch and a mallet. Seventeen percent (12 of 70) of the hips needed unplanned stem revision owing to difficulties with the head removal (Table 2). Stem revisions included one case in which the taper adapter was successfully removed with an extraction tool, but the taper was severely damaged, requiring a stem revision (Fig. 3). The median preoperative chromium and cobalt ion level of the cases with difficult head removal (ion levels available in 80% [16 of 20] of cases) was 8.0 μg/L (range, 0.8–116.5 μg/L), and cases without difficulties (ion levels available in 65% [68 of 104] of cases) was 12.6 μg/L (range, 0.5–144.6 μg/L), respectively. The preoperative chromium and cobalt ion levels were not associated with the method of head removal (difficult versus easy; median difference, 1.6 [95% CI, -7.9 to 9.9 μg/L], p = 0.53) (Fig 4).
The association of preoperative chromium and cobalt ion levels, followup time, and method of head removal are shown. The preoperative chromium and cobalt ion levels were not associated with the method of head removal (difficult versus easy; median difference, 1.6, [95% CI, −7.9 to 9.9], p = 0.53). *Other = head removal with a special extraction tool or a diamond saw cut, or the head was not removable.
In the M2a-MagnumTM group, the operation time and amount of bleeding were greater for patients whose revisions involved difficulty with head removal (Table 3). For patients with the M2a-MagnumTM (n = 20) implant in which head removal was difficult, the median operative time was longer (144 minutes; range, 75–274 minutes) and amount of bleeding was greater (725 mL; range, 300–2200 mL) than in patients with M2a-MagnumTM heads that were more easily removed (operative time: 77 minutes, range, 33–197 minutes, p < 0.001; bleeding amount: 475 mL, range, 50–1500 mL, p = 0.004; respectively) (Table 3). A total of nine measured complications occurred in M2a-MagnumTM groups with easy and hard removals during the first year after revision, including six infections and three periprosthetic fractures. With the numbers available, we did not see differences in terms of the proportion of patients experiencing major complications (periprosthetic fracture or postoperative infections) between the groups (difficult versus easy; 25% [five of 20] versus 8% [four of 50]; OR, 3.8, [95% CI, 0.9-16.2], p = 0.067).
Discussion
THAs with MoM implants have been used commonly, but owing to concerns regarding premature failure, the number of these implantations has decreased dramatically. However, many MoM implants remain in service, and the Biomet MoM hip implant may have particular problems with difficulties of removal of the modular head. Despite the manufacturer’s concerns and the FDA cases, we thought this topic was important to study because the frequency of this event has not been characterized. We found that the problem of head removal was identified only among M2a-MagnumTM heads. In 29% (20 of 70) of hips with M2a-MagnumTM implants, we had difficulties with head removal, which increased operation time and amount bleeding, and in 17% (12 of 70) of cases difficulties led to unplanned stem revision.
A weakness of the current study was that evaluation of removal of the heads and taper adapters was made retrospectively based on operative reports. However, the cold-welding and the method of head extraction were clearly described in the operative reports in general. For the first head removals, we did not have a specific extraction tool, and our knowledge of the risk of cold welding among M2a-MagnumTM heads was limited. Therefore, it is possible that some of the stem revisions could have been avoided in the M2a-MagnumTM group by using an extraction tool or diamond saw. Nevertheless, if the difficulties with head removal occur, the operation time, and apparently bleeding, will increase, even if the femoral head is able to be detached with an extraction tool or diamond saw. In addition, we had to revise one stem with a severely damaged taper, even though we were able to remove a cold-welded femoral head. A final limitation is that we had preoperative ion levels measured in only 68% of patients. Therefore, we cannot reach a firm conclusion regarding the role of ion levels, but the median ion levels were higher among patients without head removal problems, which suggests that the head-detaching problem is not associated with high ion levels.
All the difficulties with head removal were among M2a-MagnumTM heads. The size of the femoral head is different between M2a-MagnumTM and M2a-38TM heads; the latter has a fixed head size of 38 mm. The role of head size in cold welding is unclear. To our knowledge, this specific topic has not been considered by others, therefore we cannot compare this finding with those of other studies. Cold welding seems to be typical in titanium devices, such as titanium plates and screws, but in THAs with MoM implants, it remains unclear how much cold welding is related to corrosion and chromium and cobalt ions release from the bearing surfaces or the mechanical properties of the taper. In the current study, the ion levels were not associated with the problems of head removal. The problems of detaching the head were seen in the first revisions, but we presume that the longer the time from implantation, the greater the risk for difficulties in detaching the head.
The M2a-MagnumTM revisions in which we experienced difficulties extracting the femoral head were longer and involved more blood loss than the M2a-MagnumTM revisions in which this problem was not experienced. As there are no other published studies reporting on difficulties of head removal except FDA case reports, we cannot compare our results with those of other studies. In general, revisions of MoM implants have a high risk for complications [15, 20]. In the current study, we compared revisions of MoM implants (M2a-MagnumTM groups) in which the head could be removed with or without problems. The risk for periprosthetic fracture or joint infection was similar in both groups, but the trend for complications was greater in patients with difficult head removal (25%; five of 20) compared with patients with no difficulties (8%; four of 50).
The titanium–titanium taper junction can be difficult to separate during revision THA, and if not anticipated, this problem can result in larger and more complicated revision procedures in patients with the M2a-MagnumTM implant. These unexpected difficulties may arise even if the primary operation was performed recently (within 2 years) and even if serum ion levels are low. The problems with head removal increase the operation time and amount of bleeding, and the surgeon should inform the patient of the possibility of a more extensive operation than preoperatively planned, including extended trochanteric osteotomy for stem revision. These technical problems also may arise suddenly in emergency THA revisions for septic infections and periprosthetic fractures. In revision procedures with the M2a-MagnumTM implant, it is crucial to be prepared with special tools, including a femoral head extraction tool provided by the stem manufacturer and a diamond saw. We also note that the use of a titanium sleeve over an existing titanium trunnion is increasing [10]. Based on the results of our study, we raise the concern that a titanium sleeve may be cold-welded over a titanium stem, and removal of the sleeve may be difficult in the case of rerevision. We recommend additional studies with sufficient followup before the extensive use of titanium sleeves.
References
Australian Orthopaedic Association National Joint Replacement Registry. Annual Report 2008. Available at: https://aoanjrr.sahmri.com/documents/10180/42662/Annual%20Report%202008?version=1.1&t=1349406277970. Accessed January 28, 2016.
Australian Orthopaedic Association National Joint Replacement Registry. Metal on Metal Bearing Surface Total Conventional Hip Arthroplasty. Supplementary Report 2015. Available at: https://aoanjrr.sahmri.com/documents/10180/217647/Metal%20on%20Metal%20Bearing%20Surface%20Total%20Conventional%20Hip%20%20Arthroplasty. Accessed January 28, 2016.
Biomet Orthopedics. Biomet M2a-Magnum and M2a-Magnum Tri-Spike Acetabular Shells & Biomet Selex Modular Head Components. Attention Operating Surgeon. Available at: http://www.biomet.com/wps/wcm/connect/internet/94d7e61d-393c-4b0f-b15c-92b05cb87545/0958+Biomet+Metal+on+Metal+Hip+Devices+Rev+O.pdf?MOD=AJPERES. Accessed January 28, 2016.
Biomet Orthopedics. M2a-MagnumTM Large Metal Articulation: Design Rationale. Available at: http://www.biomet.com/campaign/trueAlternativeBearings/BOI03400MagnumDesignRationale.pdf. Accessed January 28, 2016.
Biomet Orthopedics Inc. M2a-38TM Metal-on-Metal. Available at: http://www.biomet.com/wps/wcm/connect/internet/acb6d5c6-e3e9-42e2-b3e6-83fd38a567f1/Y-BMT-735_021502_K.pdf?MOD=AJPERES. Accessed January 28, 2016.
European Commission, Health and Food Safety Scientific Committees. Final opinion on Metal-on-Metal joint replacements. Available at: http://ec.europa.eu/health/scientific_committees/consultations/public_consultations/scenihr_consultation_20_en.htm. Accessed January 28, 2016.
Finnish Arthroplasty Association. [Recommendation of metal-on-metal total hip arthroplasty use and follow-up. The recommendation for the update 2015.][in Finnish] May 15, 2012. Available at: http://www.suomenartroplastiayhdistys.fi/index.php?page=1074&lang=1. Accessed January 28, 2016.
Graves SE, Rothwell A, Tucker K, Jacobs JJ, Sedrakyan A. A multinational assessment of metal-on-metal bearings in hip replacement. J Bone Joint Surg Am. 2011;93(suppl 3):43-47.
Heneghan C, Langton D, Thompson M. Ongoing problems with metal-on-metal hip implants. BMJ. 2012;344:e1349.
Jack CM, Molloy DO, Walter WL, Zicat BA, Walter WK. The use of ceramic-on-ceramic bearings in isolated revision of the acetabular component. Bone Joint J. 2013;95:333-338.
Langton DJ, Jameson SS, Joyce TJ, Gandhi JN, Sidaginamale R, Mereddy P, Lord J, Nargol AV. Accelerating failure rate of the ASR total hip replacement. J Bone Joint Surg Br. 2011;93:1011-1016.
MHRA (Medicines and Healthcare products Regulatory Agency). Medical Device Alert: All metal-on-metal (MoM) hip replacements (MDA/2012/036). Available at: https://assets.digital.cabinet-office.gov.uk/media/5485abf6ed915d4c10000273/con155767.pdf. Accessed January 28, 2016.
Mokka J, Junnila M, Seppänen M, Virolainen P, Pölönen T, Vahlberg T, Mattila K, Tuominen EK, Rantakokko J, Äärimaa V, Kukkonen J, Mäkelä KT. Adverse reaction to metal debris after ReCap-M2A-Magnum large-diameter-head metal-on-metal total hip arthroplasty. Acta Orthop. 2013;84:549-554.
NJR National Joint Registry. 12th Annual Report 2015. Available at: http://www.njrcentre.org.uk/njrcentre/Portals/0/Documents/England/Reports/12th%20annual%20report/NJR%20Online%20Annual%20Report%202015.pdf. Accessed January 28, 2016.
Stryker LS, Odum SM, Fehring TK, Springer BD. Revisions of monoblock metal-on-metal THAs have high early complication rates. Clin Orthop Relat Res. 2015;473:469-474.
US Food and Drug Administration. General Recommendations for Orthopaedic Surgeons BEFORE Metal-on-Metal Hip Replacement Surgery. Available at: http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/MetalonMetalHipImplants/ucm241667.htm#1. Accessed January 28, 2016.
US Food and Drug Administration. MAUDE Adverse Event Report: Biomet Orthopedics M2A Magnum Taper Adapter 42-50 mm Head Size +3 mm Prosthesis, Hip. Available at: http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfMAUDE/detail.cfm?mdrfoi__id=2266338. Accessed January 28, 2016.
US Food and Drug Administration. MAUDE Adverse Event Report: Biomet Inc M2A Magnum Taper Adaptor Prosthesis, Hip Component. Available at: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfmaude/detail.cfm?mdrfoi__id=1183524. Accessed January 28, 2016.
US Food and Drug Administration. Recalls Specific to Metal-on-Metal Hip Implants. Available at: http://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/MetalonMetalHipImplants/ucm241770.htm. Accessed January 28, 2016.
Wyles CC, Van Demark RE 3rd, Sierra RJ, Trousdale RT. High rate of infection after aseptic revision of failed metal-on-metal total hip arthroplasty. Clin Orthop Relat Res. 2014; 472:509-516.
Author information
Authors and Affiliations
Corresponding author
Additional information
Each author certifies that he or she, or a member of his or her immediate family, has no funding or commercial associations (eg, consultancies, stock ownership, equity interest, patent/licensing arrangements, etc) that might pose a conflict of interest in connection with the submitted article.
All ICMJE Conflict of Interest Forms for authors and Clinical Orthopaedics and Related Research ® editors and board members are on file with the publication and can be viewed on request.
Clinical Orthopaedics and Related Research ® neither advocates nor endorses the use of any treatment, drug, or device. Readers are encouraged to always seek additional information, including FDA-approval status, of any drug or device prior to clinical use.
This work was performed in Turku University Hospital, Turku, Finland, and Oulu University Hospital, Oulu, Finland.
About this article
Cite this article
Mäntymäki, H., Mäkelä, K.T., Vahlberg, T. et al. Modular to Monoblock: Difficulties of Detaching the M2a-MagnumTM Head Are Common in Metal-on-metal Revisions. Clin Orthop Relat Res 474, 1999–2005 (2016). https://doi.org/10.1007/s11999-016-4774-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11999-016-4774-7