Abstract
Purpose of Review
Recent US guidelines have changed the definition of hypertension to ≥ 130/80 mmHg and recommended more intense blood pressure (BP) targets. We summarize the evidence for intense BP treatment and discuss risks that must be considered when choosing treatment goals for individual patients.
Recent Findings
The SPRINT study reported that treating to a systolic BP target of 120 mmHg reduces cardiovascular outcomes in high-risk individuals, supporting more intensive BP reduction than previously recommended. However, recent observational studies have placed emphasis on the BP J-curve phenomenon, where low BPs are associated with adverse cardiovascular outcomes, suggesting that overly aggressive BP targets may sometimes be harmful. We attempt to reconcile these apparent contradictions for the clinician. We also review other potential dangers of aggressive BP targets, including syncope, renal impairment, polypharmacy, drug interactions, subjective drug side-effects, and non-adherence.
Summary
We suggest a personalized approach to BP drug management considering individual risks, benefits, and preferences when choosing therapeutic targets, recognizing that a goal of 130/80 mmHg should always be considered. Additionally, we recommend an intense focus on lifestyle changes and medication adherence.
Similar content being viewed by others
Introduction
High blood pressure (BP) is one of the most common modifiable causes of mortality in the USA, accounting for approximately one in five deaths [1]. In addition, the global prevalence of hypertension has been steadily increasing, leading to higher attributable mortality [2]. As such, national and international efforts to control high BP are of paramount importance for public health. Based on recent clinical trials, most notably SPRINT (Systolic Blood Pressure Intervention Trial) [3••], the 2017 American College of Cardiology (ACC)/American Heart Association (AHA) guidelines for BP management have recommended more intensive BP targets, changing the definition of hypertension from ≥ 140/90 mmHg to ≥ 130/80 mmHg [4]. While SPRINT suggested benefit for treatment to BP levels of 120/80 mmHg, due in part to the technique of BP measurement in SPRINT (often unattended) - which has been extensively debated and remains controversial [5, 6] - the more conservative target BP of < 130/80 mmHg was chosen in the 2017 ACC/AHA guidelines. As a consequence, approximately 46% of American adults now meet diagnostic criteria for hypertension—an increase in prevalence from 32% with the prior definition [7]. However, concerns have been raised regarding the possibility of adverse outcomes associated with more aggressive BP control in some circumstances, specifically with pharmacologic therapy [8, 9].
Is Lower BP and More Intense BP Therapy Always Better?
The benefit of hypertension treatment has been well-established for many decades [10]. Early studies definitively established antihypertensive drug efficacy, with initial systolic BP (SBP) treatment targets < 160 mmHg showing clinical benefit [11, 12], followed by The Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial (ALLHAT) showing a benefit of SBP < 140 mmHg [13]. Similarly, early randomized placebo-controlled trials demonstrated the benefits of antihypertensive agents targeted towards patients with either Diastolic BP (DBP) ≥ 115 mmHg or DBP ≥90 mmHg. Treatment to the lower DBP target resulted in lower cardiovascular disease (CVD) events and mortality [14, 15]. These findings were then supported by much larger studies such as the Hypertension Detection and Follow-up Program (HDFP), targeting a DBP ≤ 90 mmHg and showing a significant reduction in CVD events and mortality in the intervention group compared to the usual care group [16]. Taken together, these studies firmly established a BP target of <140/90 mmHg, which had been mostly unchanged within previous BP guidelines for decades.
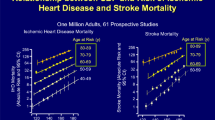
However, controversy has existed regarding the benefits of targeting even lower blood pressures. A 2002 meta-analysis of observational data from 1 million individuals participating in the Prospective Studies Collaboration showed that the risk of CVD increases above a SBP of > 115 mmHg or DBP > 75 mmHg, supporting the concept of more intense BP control to even lower targets than recommended in prior BP treatment guidelines [17]. In another meta-analysis of prospective cohort studies, SBP from 130 to 139 or DBP 85 to 89 mmHg had a higher risk of CVD events compared to individuals with SBP 120 to 129 or DBP 80 to 84 mmHg, respectively. In the same study, BP < 120/80 mmHg had better outcomes than those with higher BP [18]. These findings suggest that a target BP of < 140/90 mmHg is too liberal and could be associated with worse CVD outcomes compared to a lower BP target within the physiologically normal range. However, these are observational findings, and the risk of selection and publication bias is a serious concern. In addition, individual observational studies vary in their approach to adjusting for potential confounding risk factors.
Despite these concerns, further support for lower BP targets can also be found in the randomized trial data. For example, meta-analyses of randomized control trials reported that, compared to patients who achieved a SBP < 130 mmHg with more intensive BP therapy, those with on-treatment SBP ≥ 130 mmHg had increased stroke and all-cause mortality without a difference in cardiovascular mortality. Stroke was also reduced when DBP was <80 mmHg compared to when it was ≥80 mmHg [19••, 20]. The reduction in stroke was particularly significant in patients with high CVD risk [21]. In another meta-analysis, an on-treatment SBP of 120–124 mmHg, compared to 130–134 mmHg, was associated with a 29% decrease in CVD and a 27% decrease in all-cause mortality [22]. However, the collated randomized data are inconsistent and there are other meta-analyses supporting no benefit for stricter BP targets, including a recent one reporting no benefit to more intense SBP treatment below a target SBP of 140 mmHg [23].
Unfortunately, most of the trials included in these meta-analyses were not designed to examine the benefits of specific BP targets (rather the majority studied less versus more antihypertensive therapy). In studies supporting an intensive management approach without specific BP targets, one could argue that the lower BP achieved in the intensive arm may reflect a healthier population [24], and adults less prone to resistant hypertension (which is most commonly due to noncompliance) [25,26,27]. In addition, the study by Bundy et al. [22] included a wide variety of baseline populations including those with stroke, coronary artery disease, or heart failure all of which are different risk profiles compared to primary prevention patients who are usually healthier. In fact, each meta-analysis of randomized antihypertensive trials has included a different combination of studies and used a different approach to analyses, highlighting the inherent flaws of these types of studies and helping to explain differences in their findings.
Despite these concerns, the above data supported the need for a contemporary randomized control trial of intensive BP control. SPRINT [3••] was a trial of individuals with high CVD risk of 2.2% per year without diabetes who were randomized to an intensive target SBP of < 120 mmHg compared with the standard target of < 140 mmHg. The intensive therapy arm had a lower rate of the primary composite outcome of myocardial infarction, other acute coronary syndromes, stroke, heart failure, or CVD death. The benefit persisted regardless of baseline systolic BP (when stratified into tertiles) [28] and in patients > 75 years [29]. In addition, a recent report from a substudy of SPRINT (SPRINT Memory and cognition IN Decreased – SPRINT MIND) reported that individuals randomized to the intensive arm had a lower risk of mild cognitive impairment and dementia on follow-up. Although the full publication is still pending, this is consistent with observational study data of SBP [30].
One major concern for intensive SBP management was the adverse outcomes of lowering diastolic BP which may affect coronary perfusion [31]. However, the benefits of intensive SBP for the composite outcome in SPRINT were not attenuated among those with low baseline diastolic BP (though results for coronary outcomes specifically have not been reported according to baseline DBP) [32]. In addition, the SPRINT study excluded individuals who were < 50 years of age, estimated glomerular filtration rate < 20 ml/min/1.73m2, with diabetes, or had symptomatic heart failure within the past 6 months or left ventricular ejection fraction < 35%. Therefore, caution is required before applying the study results to these patient populations.
ACCORD was a similarly designed trial among diabetics that showed lower risk of stroke at an SBP target of 120 mmHg, though this trial did not reach statistical significance for the primary endpoint (nonfatal myocardial infarction, nonfatal stroke, or cardiovascular death) [33•]. One reason for this apparent discrepancy may be that ACCORD was underpowered with a smaller study sample than SPRINT. Looking closely at the ACCORD results shows a trend towards lower hazard for the primary outcome among those randomized to a lower BP target (hazard ratio of 0.88, 95% confidence interval 0.73–1.06). Potentially supporting this hypothesis is an analysis combining results from both ACCORD and SPRINT, which showed that intensive BP management was associated with a reduction in stroke, heart failure, and for each study’s primary outcome (which were different for each study as discussed above) [34•].
However, another reason for the discrepancy between SPRINT and ACCORD may be that the benefits of lower SBP achieved with drug therapy may not be universal and may differ according to personal clinical attributes of the patient under consideration. In the Heart Outcomes Prevention Evaluation (HOPE)-3 study of intermediate CVD risk individuals with a mean baseline SBP of 138 mmHg, aggressive SBP lowering with dual antihypertensives resulting in a mean SBP < 130 mmHg versus placebo did not result in any significant difference in CVD outcomes or mortality [35••]. Although HOPE-3 and SPRINT are often compared, the latter specifically had two BP target groups whereas the former used a more intensive antihypertensive regimen without a target BP group. Furthermore, the absolute BP lowering during the trial in HOPE-3 was far less than that seen in SPRINT. However, another reason for the difference may be that patients that are at highest risk benefit from more intensive SBP control, whereas lower risk individuals may not, as suggested by a recent meta-analysis of trials that showed that benefits from BP reduction appeared to be related to baseline estimated CVD risk [36]. As such, lower BP with drug treatment may not always be necessarily better and adults with low CVD risk may be exposed in excess to the toxicity of antihypertensive agents when treated intensively with little chance of deriving any clinical benefits, making them particularly prone to the dangers of overly aggressive BP control.
What Are the Dangers of Overly Aggressive BP Control?
With increasingly aggressive BP management, it is conceivable that SBP or DBP may become too low, resulting in more dangers than benefits. For example, at a certain SBP or DBP threshold, physiological perfusion and autoregulation of vital organs could be impaired resulting in adverse outcomes [37, 38].
J-Curve Phenomenon
Unlike SBP, targeting a more aggressive lowering of DBP appears to have thus far failed to show any benefit. In the Hypertension Optimal Treatment (HOT) trial, there was no difference in major CVD events between the groups who were allocated to a DBP target of ≤ 90, ≤ 85, or ≤ 80 mmHg, although actual reduction in DBP with treatment was lower than initially targeted [39]. Over the past three decades, data has accumulated suggesting that, not only may there not be a benefit for aggressive DBP lowering, but instead, there may be a J-curve relationship with a nadir in benefit of DBP lowering below which the risk of adverse outcomes increases. Such a J-curve could be explained by the impairment of coronary perfusion, which occurs predominantly during diastole. Supporting this idea, among patients undergoing coronary angiography and fractional flow reserve measurement, more patients had dangerously low coronary blood flow when DBP was found to be < 70 mmHg [40]. As a result, a very low DBP would be expected to increase coronary events, and have a less significant effect on other vascular beds such as cerebral or renal.
In one meta-analysis of observational studies, there was a trend for increased ischemic heart disease events among those with DBP < 70 mmHg [17], and in a second systematic review of 13 studies, CVD events were increased when patients were treated to DBP < 85 mmHg [41••]. In the Prospective Observational Longitudinal Registry of Patients with Stable Coronary Artery Disease (CLARIFY) study of > 22,000 individuals with stable coronary artery disease, after extensive adjustment, a DBP ≥ 80 mmHg or < 70 mmHg compared with 70–79 mmHg (reference) was associated with increased CVD events, but not stroke. A DBP < 60 mmHg was associated with a 2-fold increase in CVD events compared to the reference group [42••].
This increased risk of adverse outcomes has also been noted in trials. Among participants with prior ischemic heart disease enrolled in HOT, those treated to a DBP of 80 mmHg had a numerically higher rate of myocardial infarction than those treated to a DBP of 85 mmHg [43]. Further, in individuals with known CAD and hypertension enrolled in the International Verapamil-Trandolapril Study (INVEST), there was a reduction in CVD events when DBP was below < 90 mmHg but the risk increased if DBP was < 70 mmHg [44, 45•]. In addition, the recently published secondary analysis of the Ongoing Temisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET) ACEI inhibitors and Telmisartan Randomised Assessment Study of ACE Intolerant (TRANSCEND) randomized trials demonstrated that among patients greater than 54 years of age with CVD, DBP reduction to < 70 mmHg was associated with higher myocardial infarction and all-cause death but not stroke, whereas DBP ≥ 80 mmHg increased the risk of stroke and heart failure hospitalization [46•]. In this excellent study, in addition to adjustment for baseline comorbidities, analyses were also adjusted for competing events occurring during observation. In addition, the adverse outcomes of low DBP persisted in separate analyses where patients with recent nonfatal events, extremely low SBP and those not on antihypertensives were excluded to address some of the possible concerns for reverse causality as an explanation for the increased DBP outcomes [46•]. The effect of low DBP on increased CAD events is beyond what would be expected if poor health status alone was the explanation, as a more unwell patient population would also be expected to have an increase in other adverse events such as stroke.
Of note, another patient population who may also be at increased risk due to low DBP are patients with heart failure, a condition where the left ventricular end-diastolic pressure may be elevated. Lowering of the DBP would reduce the coronary perfusion pressure (related to the difference in left ventricular end-diastolic pressure and DBP) in heart failure even further [47]. In recent post hoc analyses from the Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist Trial (TOPCAT) of patients with heart failure with preserved ejection fraction, DBP was associated with increased risk CVD events when < 70 mmHg, and increased risk of heart failure hospitalization when < 60 mmHg [48, 49].
The potential dangers of low DBP may often be unrecognized. In the Atherosclerosis in Communities (ARIC) cohort in individuals without known CVD who had a DBP < 70 mmHg or DBP ≥ 100mHg had increased odds of elevated high-sensitivity troponin-T, using individuals with DBP of 80–89 mmHg as reference, suggesting asymptomatic subclinical myocardial damage in individuals with a low DBP. On a 21-year follow-up of ARIC participants, DBP < 60 mmHg was associated with increased CVD events and all-cause mortality [31]. Further, the dangers of DBP < 60 mmHg are also evident in individuals with asymptomatic subclinical coronary atherosclerosis, as evaluated by coronary artery calcium [50]. Thus, aggressive DBP lowering appears to increase the risk of CAD events particularly in individuals with underlying coronary artery disease. However, other studies have suggested that the DBP J-curve may be a non-causal phenomenon, and as such, this issue is not settled entirely [32]. Nonetheless, we still recommend that persons being treated to more intensive systolic BP goals who have CAD or LVH have close follow-up for diastolic BPs < 60 mmHg, which may in some circumstances justify consideration of de-intensification of BP therapy.
It also remains unclear if there is a SBP J-curve. In the INVEST study discussed above, patients with diabetes had increased risk of all-cause mortality if the achieved SBP was < 115 compared to SBP < 130 [51]. In the CLARIFY registry, SBP ≥ 140 mmHg and < 120 mmHg was associated with increased myocardial infarction, stroke, and CVD death [42••]. One explanation for the adverse outcomes with lower SBP may be that individuals achieving a lower SBP may be less healthy at baseline and have unadjusted confounders (such as vascular stiffness and elevated pulse pressure). In one important observational study of individuals above 85 years of age, comprehensive adjustment for health status and frailty attenuated the relationship between low SBP and all-cause mortality that was seen in models not adjusted for health status and frailty [52]. Similarly, in the Framingham Heart Study a U-shaped curve between CVD mortality and SBP was no longer significant after adjustment [53]. Although it may be true that patients with lower SBP may have other comorbidities, these findings nonetheless highlight the practical importance of identifying these high-risk patients for an individualized approach to management and avoiding excessive sudden drops in SBP where possible given their excess “real-world” levels of risk for bad outcomes.
In addition, there may also be other vulnerable populations where particular caution is required. In a small randomized trial of patients in South Korea with internal carotid artery stenosis and a subacute ischemic stroke (within the past 7–42 days), SBP < 120 mmHg compared with < 140 mmHg was associated with increased ischemic lesion volume and a trend towards higher frequency of new ischemic lesions [54•]. Of note, this study only reached a mean SBP of 124.6 mmHg in the intensive treatment arm; thus, the evaluation more closely reflects < 140 mmHg versus < 130 mmHg. Vascular stenosis related impairment of cerebral blood flow is likely exacerbated by the more aggressive BP management in this vulnerable population. In comparison, in the multicenter randomized Secondary Prevention of Small Cortical Strokes (SPS3) study of patients with a lacunar infarct, SBP target of <130 mmHg versus 130–149 mmHg resulted in a significantly lower rate of intracerebral hemorrhage (hazard ratio 0.37, 95% confidence interval 0.15–0.95) and a trend towards lower strokes [55]. In these patients, cerebral blood flow is not reduced with intensive BP lowering [56]. Therefore, the mechanism of stroke is an important consideration regarding BP management in this setting as the pathophysiology will reflect the risks.
Other Adverse Effects
Additionally, although there was no significant difference in all serious adverse events in SPRINT overall, hypotension, syncope, acute kidney injury, and electrolyte abnormalities were significantly more common in the intense arm, sometimes resulting in emergency department visits. SPRINT was also stopped early, and thus, it is unclear if there could be even more cumulative adverse effects with longer duration of intensive BP therapy [3••].
One of the long-held concerns has been the risk of falls associated with hypotensive episodes. In one study of Medicare patients, there was a transient increase in the risk of falls within the first 15 days of antihypertensive initiation or uptitration [57]. However, there was no increase in long-term risk of falls. In fact, several studies have failed to demonstrate an increase in risk of falls with the initiation or intensification of antihypertensive treatment [58,59,60, 61•]. The risk of falls with antihypertensives is associated more to individual indicators of frailty such as impaired mobility, cognitive impairment, depression, history of falls, and presence of orthostatic hypotension [58, 59, 61•]. This is not surprising, as these are the same risk factors for falls for the general population [62]. This is consistent with the ACCORD study where, although hypotension and syncope was more common in the intensive arm, there was no increase in falls or non-spine fractures [33, 63]. Further, undertreated hypertension itself is a risk for falling [59, 64].
The renal risk of severe hypotension has also been shown in metanalyses [65]. Some of these effects may persist long-term, with one analysis of SPRINT and ACCORD at 3 years showing that the incidence of chronic kidney disease was 10% in the intensive arm compared with 4.1% in the standard arm [66]. Adverse renal outcomes appeared to be more common in patients with a DBP < 70 mmHg [67]. Estimates suggest that applying the SPRINT eligibility criteria for intensive BP implementation to the 1999 to 2006 National Health and Nutrition Examination Survey (NHANES) would result in 56,100 episodes of hypotension, 34,400 episodes of syncope, and 88,700 excess cases of acute kidney injury per year. However, importantly, the study also projected a reduction in 107,500 deaths with the intensive SBP goal [68]. Thus, although the current data demonstrate a significantly higher risk of adverse outcomes, there are also clear benefits.
It is likely the dangers of the adverse outcomes of hypotension and falls affect those with multiple comorbidities disproportionately as discussed above. It is important to note that patients with a prior stroke, end-stage renal disease or eGFR < 20 ml/min/1.73m2, and symptomatic heart failure were excluded from the SPRINT study in addition to patients with diabetes. Many of these comorbidities increase the frailty of the patients and risk for adverse effects.
Polypharmacy
Intensive BP therapy inevitably results in greater polypharmacy. In the SPRINT study, the intensive arm required a mean of three medications compared with 1.9 in the standard arm [3••], and in the ACCORD trial the mean number of medications was 3.4 and 2.3 in the intensive and standard arms, respectively [33•]. These means are tied with significant variability in medication numbers at the individual level (some patients required far more medication to achieve the intensive goal). This not only increases the cost [68] but also increases the risk of medication errors, medication non-adherence, medication interactions, and medication related side-effects [69, 70]. Indicators of frailty such as psychiatric disorders, cognitive dysfunction, and audiovisual problems exacerbate these issues and remain another concern for aggressive BP treatment [71]. One potential strategy to reduce polypharmacy while also attaining more intensive BP targets is renal denervation, which is recently enjoying a revival in the literature [72, 73].
Pulse Pressure
Finally, pulse pressure is an underappreciated and infrequently discussed risk factor for adverse outcomes and may also be an important consideration in the intensive systolic BP treatment of patients. Patients with a low DBP but a high SBP (that is a large pulse pressure) may have worse outcomes [31, 46•]. In the international Reduction of Atherothrombosis for Continued Health (REACH) registry, a higher pulse pressure quartile was associated with an elevated incidence of adverse CVD outcomes [74]. However, pulse pressure remains a poorly studied parameter in randomized trials of management of BP and further thought is required in integrating pulse pressure with our understanding of the SPRINT results and the J-curve phenomenon.
Individualized Approach
What is clear from the discussion above is that there are well-demonstrated benefits to intensive BP management when applied in the right patient population, such as those with a high CVD risk. However, there are also potential risks of aggressive pharmacological BP treatment, such as an increased risk of CAD events if the DBP is lowered too severely, an increased risk of stroke if there is obstructive cerebral blood flow, or an increased risk of electrolyte and renal dysfunction in high-risk patients. These findings highlight the importance of taking an individualized approach to BP targets and management strategies as opposed to blanket guidelines for a target BP for the general population (Fig. 1). In addition, future treatment strategies may integrate a complex approach taking into account, for example, genomics, proteomics, and pharmacokinetics [75]. However, what remains consistent and clear is that BP reduction with lifestyle modifications such as smoking cessation, avoidance of excess alcohol, exercise, and/or dietary changes is always beneficial [4]. Further, as discussed above, non-adherence is common among patients being treated for hypertension [25,26,27], and instead of focusing on lower targets (130/80 mmHg), providers often need to focus on adherence to ensure even old targets (140/90 mmHg) are achieved in their patients. Only then, and after a consideration of individual risk:benefit and discussion with the patient regarding goals and preferences, should the more intensive BP target be considered.
Limitations
As the discussion above shows, there are many reports of the effects of intensive BP management (usually referred to as target < 130 mmHg or < 120 mmHg) versus standard BP management (target < 140 mmHg). However, most of these studies are meta-analysis of observational studies or nonrandomized post hoc data from previous randomized control trials where randomization was not based on an intensive versus standard BP target as defined above. The most important study for aggressive BP management is the SPRINT study among high cardiovascular risk patients without diabetes or stroke. Thus, caution is necessary when applying the SPRINT data more broadly such as in lower risk patients, as well as patients with significant obstructive cerebral blood flow as discussed above. Further, it is worth considering that the absolute benefits of intensive SBP lowering is related to the patients underlying CVD risk, for example, as shown among patients with subclinical atherosclerosis [76].
Conclusions
The current guidelines recommend a target BP of < 130/80, which appears to be safe in the majority of patients. However, an individualized approach is required to management of patients by assessing specific risks for each patient. For example, a patient with advanced renal disease may be more vulnerable to the electrolyte abnormalities associated with aggressive BP management, or a patient on triple anti-thrombotic therapy may be more vulnerable to complications of a syncopal or hypotensive episode that appears more common with intense BP management, as described above. Caution is also required when aiming for more aggressive approaches among lower risk individuals as there is a lack of randomized control trials in that patient population. Further, an abundance of data support that patients whose DBP is lowered < 60 mmHg especially if they have underlying coronary artery disease may suffer from higher CAD outcomes. In addition, in our approach to pursue guidelines recommendations, clinicians often forget to develop a doctor-patient agreement to a therapeutic goal. This likely contributes to the poor adherence of patients, and the failure to reach targets that is commonly seen; leading to a reflex addition of more agents [77]. These settings are likely to increase the dangers of aggressive blood pressure management strategies.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Danaei G, Ding EL, Mozaffarian D, Taylor B, Rehm J, Murray CJ, et al. The preventable causes of death in the United States: comparative risk assessment of dietary, lifestyle, and metabolic risk factors. PLoS Med. 2009;6(4):e1000058.
Forouzanfar MH, Liu P, Roth GA, Ng M, Biryukov S, Marczak L, et al. Global burden of hypertension and systolic blood pressure of at least 110 to 115 mm Hg, 1990–2015. JAMA. 2017;317(2):165–82.
•• Group SR, Wright JT Jr, Williamson JD, Whelton PK, Snyder JK, Sink KM, et al. A Randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373(22):2103–16. The largest randomized control trial supporting the benefit of aggressive systolic blood pressure control.
Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Dennison Himmelfarb C, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018;71(19):e127–248.
Flack JM. Method of blood pressure measurement, interpretation of SPRINT, and the Atlantic divide. Curr Hypertens Rep. 2017;19(3):19.
Johnson KC, Whelton PK, Cushman WC, Cutler JA, Evans GW, Snyder JK, et al. Blood Pressure measurement in SPRINT (Systolic Blood Pressure Intervention Trial). Hypertension. 2018;71(5):848–57.
Muntner P, Carey RM, Gidding S, Jones DW, Taler SJ, Wright JT, et al. Potential US population impact of the 2017 American College of Cardiology/American Heart Association high blood pressure guideline. J Am Coll Cardiol. 2018;71(2):109–18.
Bakris G, Sorrentino M. Redefining hypertension - assessing the new blood-pressure guidelines. N Engl J Med. 2018;378(6):497–9.
Yang JY, Chiu S, Krouss M. Overtreatment of asymptomatic hypertension—urgency is not an emergency: a teachable moment. JAMA Int Med. 2018;178(5):704–5.
Pfeffer MA, McMurray JJ. Lessons in uncertainty and humility - clinical trials involving hypertension. N Engl J Med. 2016;375(18):1756–66.
Staessen JA, Fagard R, Thijs L, Celis H, Arabidze GG, Birkenhager WH, et al. Randomised double-blind comparison of placebo and active treatment for older patients with isolated systolic hypertension. The Systolic Hypertension in Europe (Syst-Eur) Trial Investigators. Lancet. 1997;350(9080):757–64.
Beckett NS, Peters R, Fletcher AE, Staessen JA, Liu L, Dumitrascu D, et al. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358(18):1887–98.
ALLHAT Collaborative Research Group. Major cardiovascular events in hypertensive patients randomized to doxazosin vs chlorthalidone: the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). JAMA. 2000;283(15):1967–75.
Effects of treatment on morbidity in hypertension. Results in patients with diastolic blood pressures averaging 115 through 129 mm Hg. JAMA. 1967;202(11):1028–34.
Effects of treatment on morbidity in hypertension. II. Results in patients with diastolic blood pressure averaging 90 through 114 mm Hg. JAMA. 1970;213(7):1143–52.
Hypertension Detection and Follow-up Program Cooperative Group. Five-year findings of the hypertension detection and follow-up program. I. Reduction in mortality of persons with high blood pressure, including mild hypertension. JAMA. 1979;242(23):2562–71.
Lewington S, Clarke R, Qizilbash N, Peto R, Collins R, Prospective SC. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002;360(9349):1903–13.
Huang Y, Wang S, Cai X, Mai W, Hu Y, Tang H, et al. Prehypertension and incidence of cardiovascular disease: a meta-analysis. BMC Med. 2013;11:177.
•• Thomopoulos C, Parati G, Zanchetti A. Effects of blood pressure lowering on outcome incidence in hypertension: 2. Effects at different baseline and achieved blood pressure levels--overview and meta-analyses of randomized trials. J Hypertens. 2014;32(12):2296–304. Meta-analysis using randomized control trial data to compare systolic blood pressure < 130 mmHg to > 130 mmHg.
Turnbull F, Blood Pressure Lowering Treatment Trialists C. Effects of different blood-pressure-lowering regimens on major cardiovascular events: results of prospectively-designed overviews of randomised trials. Lancet. 2003;362(9395):1527–35.
Thomopoulos C, Parati G, Zanchetti A. Effects of blood-pressure-lowering treatment on outcome incidence. 12. Effects in individuals with high-normal and normal blood pressure: overview and meta-analyses of randomized trials. J Hypertens. 2017;35(11):2150–60.
Bundy JD, Li C, Stuchlik P, Bu X, Kelly TN, Mills KT, et al. Systolic blood pressure reduction and risk of cardiovascular disease and mortality: a systematic review and network meta-analysis. JAMA Cardiol. 2017;2(7):775–81.
Brunstrom M, Carlberg B. Association of blood pressure lowering with mortality and cardiovascular disease across blood pressure levels: a systematic review and meta-analysis. JAMA Intern Med. 2018;178(1):28–36.
Davis EM, Appel LJ, Wang X, Greene T, Astor BC, Rahman M, et al. Limitations of analyses based on achieved blood pressure: lessons from the African American study of kidney disease and hypertension trial. Hypertension. 2011;57(6):1061–8.
Ceral J, Habrdova V, Vorisek V, Bima M, Pelouch R, Solar M. Difficult-to-control arterial hypertension or uncooperative patients? The assessment of serum antihypertensive drug levels to differentiate non-responsiveness from non-adherence to recommended therapy. Hypertens Res. 2011;34(1):87–90.
Strauch B, Petrak O, Zelinka T, Rosa J, Somloova Z, Indra T, et al. Precise assessment of noncompliance with the antihypertensive therapy in patients with resistant hypertension using toxicological serum analysis. J Hypertens. 2013;31(12):2455–61.
Tomaszewski M, White C, Patel P, Masca N, Damani R, Hepworth J, et al. High rates of non-adherence to antihypertensive treatment revealed by high-performance liquid chromatography-tandem mass spectrometry (HP LC-MS/MS) urine analysis. Heart. 2014;100(11):855–61.
Shapiro BP, Ambrosius WT, Blackshear JL, Cushman WC, Whelton PK, Oparil S, et al. Impact of intensive versus standard blood pressure management by tertiles of blood pressure in SPRINT (Systolic Blood Pressure Intervention Trial). Hypertension. 2018;71(6):1064–74.
Williamson JD, Supiano MA, Applegate WB, Berlowitz DR, Campbell RC, Chertow GM, et al. Intensive vs standard blood pressure control and cardiovascular disease Outcomes in adults aged >/=75 Years: a randomized clinical trial. JAMA. 2016;315(24):2673–82.
Abell JG, Kivimäki M, Dugravot A, Tabak AG, Fayosse A, Shipley M et al. Association between systolic blood pressure and dementia in the Whitehall II cohort study: role of age, duration, and threshold used to define hypertension. Eur Heart J. 2018;39(33):3119–25.
McEvoy JW, Chen Y, Rawlings A, Hoogeveen RC, Ballantyne CM, Blumenthal RS, et al. Diastolic blood pressure, subclinical myocardial damage, and cardiac events: implications for blood pressure control. J Am Coll Cardiol. 2016;68(16):1713–22.
Beddhu S, Chertow GM, Cheung AK, Cushman WC, Rahman M, Greene T, et al. Influence of baseline diastolic blood pressure on effects of intensive compared with standard blood pressure control. Circulation. 2018;137(2):134–43.
• Group AS, Cushman WC, Evans GW, Byington RP, Goff DC Jr, Grimm RH Jr, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med. 2010;362(17):1575–85. ACCORD is a large trial of BP control among diabetics which, while technically negative, provides tacit support to the findings of SPRINT.
• Perkovic V, Rodgers A. Redefining Blood-Pressure Targets--SPRINT Starts the Marathon. N Engl J Med. 2015;373(22):2175–8. Pooled analysis of the SPRINT and ACCORD trials showing benefit for aggressive systolic blood pressure control.
•• Lonn EM, Bosch J, Lopez-Jaramillo P, Zhu J, Liu L, Pais P, et al. Blood-pressure lowering in intermediate-risk persons without cardiovascular disease. N Engl J Med. 2016;374(21):2009–20. Randomized control trial consistent with the suggestion that benefits of intensive blood pressure control maybe related to patient specific risk.
Blood Pressure Lowering Treatment Trialists C, Sundstrom J, Arima H, Woodward M, Jackson R, Karmali K, et al. Blood pressure-lowering treatment based on cardiovascular risk: a meta-analysis of individual patient data. Lancet. 2014;384(9943):591–8.
Ikonomidis I, Makavos G, Lekakis J. Arterial stiffness and coronary artery disease. Curr Opin Cardiol. 2015;30(4):422–31.
Lucas SJ, Tzeng YC, Galvin SD, Thomas KN, Ogoh S, Ainslie PN. Influence of changes in blood pressure on cerebral perfusion and oxygenation. Hypertension. 2010;55(3):698–705.
Hansson L, Zanchetti A, Carruthers SG, Dahlof B, Elmfeldt D, Julius S, et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT Study Group. Lancet. 1998;351(9118):1755–62.
Rabkin SW, Waheed A, Poulter RS, Wood D. Myocardial perfusion pressure in patients with hypertension and coronary artery disease: implications for DBP targets in hypertension management. J Hypertens. 2013;31(5):975–82.
•• Farnett L, Mulrow CD, Linn WD, Lucey CR, Tuley MR. The J-curve phenomenon and the treatment of hypertension. Is there a point beyond which pressure reduction is dangerous? JAMA. 1991;265(4):489–95. Landmark document on the BP J-curve phenomenon.
•• Vidal-Petiot E, Ford I, Greenlaw N, Ferrari R, Fox KM, Tardif JC, et al. Cardiovascular event rates and mortality according to achieved systolic and diastolic blood pressure in patients with stable coronary artery disease: an international cohort study. Lancet. 2016;388(10056):2142–52. A large observational study demonstrating the systolic and diastolic blood pressure J-curve phenomenon where low and high blood pressures are associated with increased cardiovascular events.
Cruickshank JM. Antihypertensive treatment and the J-curve. Cardiovasc Drugs Ther. 2000;14(4):373–9.
Pepine CJ, Handberg EM, Cooper-DeHoff RM, Marks RG, Kowey P, Messerli FH, et al. A calcium antagonist vs a non-calcium antagonist hypertension treatment strategy for patients with coronary artery disease. The International Verapamil-Trandolapril Study (INVEST): a randomized controlled trial. JAMA. 2003;290(21):2805–16.
• Messerli FH, Mancia G, Conti CR, Hewkin AC, Kupfer S, Champion A, et al. Dogma disputed: can aggressively lowering blood pressure in hypertensive patients with coronary artery disease be dangerous? Ann Int Med. 2006;144(12):884–93. Landmark document on the BP J-curve phenomenon.
• Bohm M, Schumacher H, Teo KK, Lonn E, Mahfoud F, Mann JFE et al. Achieved diastolic blood pressure and pulse pressure at target systolic blood pressure (120–140 mmHg) and cardiovascular outcomes in high-risk patients: results from ONTARGET and TRANSCEND trials. Eur Heart J. 2018;39(33):3105–14. Post hoc analysis of the ONTARGET and TRANSCEND randomized control trials showing the dangers of low diastolic blood pressure.
Duncker DJ, Bache RJ. Regulation of coronary blood flow during exercise. Physiol Rev. 2008;88(3):1009–86.
Sandesara PB, O’Neal WT, Kelli HM, Topel M, Samman-Tahhan A, Sperling LS. Diastolic Blood Pressure and Adverse Outcomes in the TOPCAT (Treatment of Preserved Cardiac Function Heart Failure With an Aldosterone Antagonist) Trial. J Am Heart Assoc. 2018;7(5).
Tsujimoto T, Kajio H. Low diastolic blood pressure and adverse outcomes in heart failure with preserved ejection fraction. Int J Cardiol. 2018;263:69–74.
Rahman F, Al Rifai M, Blaha MJ, Nasir K, Budoff MJ, Psaty BM, et al. Relation of Diastolic Blood Pressure and Coronary Artery Calcium to Coronary Events and Outcomes (From the Multi-Ethnic Study of Atherosclerosis). Am J Cardiol. 2017;120(10):1797–803.
Cooper-DeHoff RM, Gong Y, Handberg EM, Bavry AA, Denardo SJ, Bakris GL, et al. Tight blood pressure control and cardiovascular outcomes among hypertensive patients with diabetes and coronary artery disease. JAMA. 2010;304(1):61–8.
Boshuizen HC, Izaks GJ, van Buuren S, Ligthart GJ. Blood pressure and mortality in elderly people aged 85 and older: community based study. BMJ. 1998;316(7147):1780–1784.
D’Agostino RB, Belanger AJ, Kannel WB, Cruickshank JM. Relation of low diastolic blood pressure to coronary heart disease death in presence of myocardial infarction: the Framingham Study. BMJ. 1991;303(6799):385–9.
• Park J-M, Kim BJ, Kwon SU, Hwang Y-H, Heo SH, Rha J-H et al. Intensive blood pressure control may not be safe in subacute ischemic stroke by intracranial atherosclerosis: a result of randomized trial. J Hypertens. 2018;36(9):1936–41. Small randomized trial showing risks of aggressive blood pressure control in patients with subacute ischemic stroke with carotid stenosis.
Group SPSS, Benavente OR, Coffey CS, Conwit R, Hart RG, McClure LA, et al. Blood-pressure targets in patients with recent lacunar stroke: the SPS3 randomised trial. Lancet. 2013;382(9891):507–15.
Croall ID, Tozer DJ, Moynihan B, Khan U, O’Brien JT, Morris RG, et al. Effect of Standard vs Intensive Blood Pressure Control on Cerebral Blood Flow in Small Vessel Disease: The PRESERVE Randomized Clinical Trial. In: JAMA Neurol; 2018.
Shimbo D, Barrett Bowling C, Levitan EB, Deng L, Sim JJ, Huang L, et al. Short-Term Risk of Serious Fall Injuries in Older Adults Initiating and Intensifying Treatment With Antihypertensive Medication. Circ Cardiovasc Qual Outcomes. 2016;9(3):222–9.
Tinetti ME, Han L, Lee DS, McAvay GJ, Peduzzi P, Gross CP, et al. Antihypertensive medications and serious fall injuries in a nationally representative sample of older adults. JAMA Intern Med. 2014;174(4):588–95.
Gangavati A, Hajjar I, Quach L, Jones RN, Kiely DK, Gagnon P, et al. Hypertension, orthostatic hypotension, and the risk of falls in a community-dwelling elderly population: the maintenance of balance, independent living, intellect, and zest in the elderly of Boston study. J Am Geriatr Soc. 2011;59(3):383–9.
Lipsitz LA, Habtemariam D, Gagnon M, Iloputaife I, Sorond F, Tchalla AE, et al. Reexamining the Effect of Antihypertensive Medications on Falls in Old Age. Hypertension. 2015;66(1):183–9.
• Bromfield SG, Ngameni CA, Colantonio LD, Bowling CB, Shimbo D, Reynolds K, et al. Blood pressure, antihypertensive polypharmacy, frailty, and risk for serious fall injuries among older treated adults with hypertension. Hypertension. 2017;70(2):259–66. Analysis evaluating the risks of falls among older patients showing that frailty indicators main predictor of fall risk.
Deandrea S, Lucenteforte E, Bravi F, Foschi R, La Vecchia C, Negri E. Risk factors for falls in community-dwelling older people: a systematic review and meta-analysis. Epidemiology 2010;21(5):658–668.
Margolis KL, Palermo L, Vittinghoff E, Evans GW, Atkinson HH, Hamilton BP, et al. Intensive blood pressure control, falls, and fractures in patients with type 2 diabetes: the ACCORD trial. J Gen Intern Med. 2014;29(12):1599–606.
Stubbs B, Mueller C, Gaughran F, Lally J, Vancampfort D, Lamb SE, et al. Predictors of falls and fractures leading to hospitalization in people with schizophrenia spectrum disorder: A large representative cohort study. Schizophr Res. 2018. https://doi.org/10.1016/j.schres.2018.05.010.
Xie X, Atkins E, Lv J, Bennett A, Neal B, Ninomiya T, et al. Effects of intensive blood pressure lowering on cardiovascular and renal outcomes: updated systematic review and meta-analysis. Lancet. 2016;387(10017):435–43.
Beddhu S, Greene T, Boucher R, Cushman WC, Wei G, Stoddard G et al. Intensive systolic blood pressure control and incident chronic kidney disease in people with and without diabetes mellitus: secondary analyses of two randomised controlled trials. Lancet Diabetes Endocrinol. 2018;6(7):555–63.
Del Pinto R, Pietropaoli D, Ferri C. Diastolic blood pressure and risk profile in renal and cardiovascular diseases. Results from the SPRINT trial. J Am Soc Hypertens. 2018;12(7)513–23.
Bress AP, Bellows BK, King JB, Hess R, Beddhu S, Zhang Z, et al. Cost-Effectiveness of Intensive versus Standard Blood-Pressure Control. N Engl J Med. 2017;377(8):745–55.
Cooney D, Pascuzzi K. Polypharmacy in the elderly: focus on drug interactions and adherence in hypertension. Clin Geriatr Med. 2009;25(2):221–33.
Mukete BN, Ferdinand KC. Polypharmacy in Older Adults With Hypertension: A Comprehensive Review. J Clin Hypertens (Greenwich). 2016;18(1):10–8.
Herr M, Sirven N, Grondin H, Pichetti S, Sermet C. Frailty, polypharmacy, and potentially inappropriate medications in old people: findings in a representative sample of the French population. Eur J Clin Pharmacol. 2017;73(9):1165–72.
Kandzari DE, Bohm M, Mahfoud F, Townsend RR, Weber MA, Pocock S, et al. Effect of renal denervation on blood pressure in the presence of antihypertensive drugs: 6-month efficacy and safety results from the SPYRAL HTN-ON MED proof-of-concept randomized trial. Lancet. 2018;391(10137):2346–55.
Azizi M, Schmieder RE, Mahfoud F, Weber MA, Daemen J, Davies J, et al. Endovascular ultrasound renal denervation to treat hypertension (RADIANCE-HTN SOLO): a multicentre, international, single-blind, randomized, sham-controlled trial. Lancet. 2018;391(10137):2335–45.
Selvaraj S, Steg PG, Elbez Y, Sorbets E, Feldman LJ, Eagle KA, et al. Pulse Pressure and Risk for Cardiovascular Events in Patients With Atherothrombosis: From the REACH Registry. J Am Coll Cardiol. 2016;67(4):392–403.
Savoia C, Volpe M, Grassi G, Borghi C, Agabiti Rosei E, Touyz RM. Personalized medicine-a modern approach for the diagnosis and management of hypertension. Clin Sci (Lond). 2017;131(22):2671–85.
McEvoy JW, Martin SS, Dardari ZA, Miedema MD, Sandfort V, Yeboah J, et al. Coronary Artery Calcium to Guide a Personalized Risk-Based Approach to Initiation and Intensification of Antihypertensive Therapy. In: Circulation. 2017;135(2):153–65.
Tamblyn R, Abrahamowicz M, Dauphinee D, Wenghofer E, Jacques A, Klass D et al. Influence of physicians’ management and communication ability on patients’ persistence with antihypertensive medication. Arch Intern Med 2010;170(12):1064–1072.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Faisal Rahman and John W. McEvoy declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Hypertension
Rights and permissions
About this article
Cite this article
Rahman, F., McEvoy, J.W. Dangers of Overly Aggressive Blood Pressure Control. Curr Cardiol Rep 20, 108 (2018). https://doi.org/10.1007/s11886-018-1063-y
Published:
DOI: https://doi.org/10.1007/s11886-018-1063-y