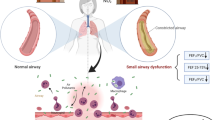
Abstract
This panel study attempted to assess the effects of ambient fine particulate matter (PM2.5) exposure on oxidative stress, inflammation, and janus kinase (JAK)/signal transducer and activator of transcription (STAT) signal molecules. Twenty-nine nonsmoking adults with asthma were followed up for three seasons. Lung function was measured four times and peripheral blood was collected one time per season. The markers of oxidative stress, inflammation, and JAK/STAT were detected. Daily concentration of ambient PM2.5 was recorded from the China National Environmental Monitoring Center. Linear mixed-effects regression models were used to investigate the relationships of ambient PM2.5 with the index of lung function, oxidative stress, inflammation, and JAK/STAT. Mediation analyses were used to explore the mediation effect of JAK. The concentration of PM2.5 was the highest in spring, with a median of 151 μg/m3. In the analyses of oxidative stress, the malonaldehyde (MDA) increased by 3.80% (95% CI: 2.43 to 5.17%; p<0.001) for each 10 μg/m3 increase in lag4 PM2.5 exposure. Furthermore, we detected a significant association in cytokines. Lag4 PM2.5 exposure was associated with an increased interleukin-4 (IL-4) by 3.00% (95% CI: 0.26 to 5.74%; p<0.05). Besides, we found mediation of JAK2 in the associations between superoxide dismutase (SOD)/IL-12 and STAT4, JAK3 in the association between MDA and STAT6. The injury of pulmonary function in asthmatic adults induced by ambient PM2.5 exposure is most likely to occur at lag 4 days. Ambient PM2.5 aggravates asthma by systemic oxidative stress and inflammation, in which JAK/STAT signal molecules may be involved.
Graphical abstract






Similar content being viewed by others
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Code availability
Not applicable.
References
Cheng Q, Yang CY, Guo BY, Wei X et al (2017) Analysis of mechanism of PM2.5 and house dust mite antigen Der p1 in attack stage of child asthma. Eur Rev Med Pharmacol Sci 21:2458–2462
Cui AH, Zhao J, Liu SX, Hao YS (2017) Associations of IL-4, IL-6, and IL-12 levels in peripheral blood with lung function, cellular immune function, and quality of life in children with moderate-to-severe asthma. Medicine (Baltimore) 96:e6265. https://doi.org/10.1097/md.0000000000006265
de Hartog JJ, Ayres JG, Karakatsani A, Analitis A et al (2010) Lung function and indicators of exposure to indoor and outdoor particulate matter among asthma and COPD patients. Occup Environ Med 67:2–10. https://doi.org/10.1136/oem.2008.040857
Delfino RJ, Quintana PJ, Floro J, Gastañaga VM et al (2004) Association of FEV1 in asthmatic children with personal and microenvironmental exposure to airborne particulate matter. Environ Health Perspect 112:932–941. https://doi.org/10.1289/ehp.6815
Ding B, Small M (2018) Disease burden of mild asthma in China. Respirology 23:369–377. https://doi.org/10.1111/resp.13189
Du PR, Du R, Ren WS, Lu ZD et al (2018) Seasonal variation characteristic of inhalable microbial communities in PM2.5 in Beijing city, China. Sci Total Environ 610:308–315. https://doi.org/10.1016/j.scitotenv.2017.07.097
Ehteshami-Afshar S, FitzGerald JM, Doyle-Waters MM, Sadatsafavi M (2016) The global economic burden of asthma and chronic obstructive pulmonary disease. Int J Tuberc Lung Dis 20:11–U38. https://doi.org/10.5588/ijtld.15.0472
Emori T, Kasahara M, Sugahara S, Hashimoto M et al (2020) Role of JAK-STAT signaling in the pathogenic behavior of fibroblast-like synoviocytes in rheumatoid arthritis: effect of the novel JAK inhibitor peficitinib. Eur J Pharmacol 882:173238. https://doi.org/10.1016/j.ejphar.2020.173238
Fan J, Li S, Fan C, Bai Z et al (2016) The impact of PM2.5 on asthma emergency department visits: a systematic review and meta-analysis. Environ Sci Pollut Res Int 23:843–850. https://doi.org/10.1007/s11356-015-5321-x
Georas SN, Donohue P, Connolly M, Wechsler ME (2021) JAK inhibitors for asthma. J Allergy Clin Immunol 148:953–963. https://doi.org/10.1016/j.jaci.2021.08.013
Hart JE, Grady ST, Laden F, Coull BA et al (2018) Effects of indoor and ambient black carbon and PM2.5 on pulmonary function among individuals with COPD. Environ Health Perspect 126:127008. https://doi.org/10.1289/ehp3668
Howell MD, Fitzsimons C, Smith PA (2018) JAK/STAT inhibitors and other small molecule cytokine antagonists for the treatment of allergic disease. Ann Allergy Asthma Immunol 120:367–375. https://doi.org/10.1016/j.anai.2018.02.012
Hsieh YY, Chang CC, Hsu CM, Wan L et al (2011) JAK-1 rs2780895 C-related genotype and allele but not JAK-1 rs10789166, rs4916008, rs2780885, rs17127114, and rs3806277 are associated with higher susceptibility to asthma. Genet Test Mol Biomarkers 15:841–847. https://doi.org/10.1089/gtmb.2011.0002
Hsu SC, Chang JH, Lee CL, Huang WC et al (2020) Differential time-lag effects of ambient PM(2.5) and PM(2.5)-bound PAHs on asthma emergency department visits. Environ Sci Pollut Res Int 27:43117–43124. https://doi.org/10.1007/s11356-020-10243-y
Jiang S, Bo L, Gong CY, Du XH et al (2016) Traffic-related air pollution is associated with cardio-metabolic biomarkers in general residents. Int Arch Occup Environ Health 89:911–921. https://doi.org/10.1007/s00420-016-1129-3
Jiang S, Bo L, Du X, Liu J et al (2017) CARD9-mediated ambient PM(2.5)-induced pulmonary injury is associated with Th17 cell. Toxicol Lett 273:36–43. https://doi.org/10.1016/j.toxlet.2017.03.015
Jiang N, Wen HY, Zhou M, Lei TT et al (2020) Low-dose combined exposure of carboxylated black carbon and heavy metal lead induced potentiation of oxidative stress, DNA damage, inflammation, and apoptosis in BEAS-2B cells. Ecotoxicol Environ Saf 206:111388. https://doi.org/10.1016/j.ecoenv.2020.111388
Juergens LJ, Worth H, Juergens UR (2020) New perspectives for mucolytic, anti-inflammatory and adjunctive therapy with 1,8-cineole in COPD and asthma: review on the new therapeutic approach. Adv Ther 37:1737–1753. https://doi.org/10.1007/s12325-020-01279-0
Kaplan MH, Sun YL, Hoey T, Grusby MJ (1996) Impaired IL-12 responses and enhanced development of Th2 cells in Stat4-deficient mice. Nature 382:174–177. https://doi.org/10.1038/382174a0
Kim Y, Seo J, Kim JY, Lee JY et al (2018) Characterization of PM2.5 and identification of transported secondary and biomass burning contribution in Seoul, Korea. Environ Sci Pollut Res 25:4330–4343. https://doi.org/10.1007/s11356-017-0772-x
Lamoureux DP, Diaz EA, Chung Y, Coull BA et al (2013) Effects of fresh and aged vehicular particulate emissions on blood pressure in normal adult male rats. Air Qual Atmos Health 6:407–418. https://doi.org/10.1007/s11869-012-0177-4
Lampinen M, Carlson M, Hakansson LD, Venge P (2004) Cytokine-regulated accumulation of eosinophils in inflammatory disease. Allergy 59:793–805. https://doi.org/10.1111/j.1398-9995.2004.00469.x
Liu L, Poon R, Chen L, Frescura AM et al (2009) Acute effects of air pollution on pulmonary function, airway inflammation, and oxidative stress in asthmatic children. Environ Health Perspect 117:668–674. https://doi.org/10.1289/ehp11813
Liu XM, Wang JH, Zhou MF, Dai QY et al (2020) Particulate matter exposure disturbs inflammatory cytokine homeostasis associated with changes in trace metal levels in mouse organs. Sci Total Environ 727. https://doi.org/10.1016/j.scitotenv.2020.138377
Mar TF, Jansen K, Shepherd K, Lumley T et al (2005) Exhaled nitric oxide in children with asthma and short-term PM2.5 exposure in Seattle. Environ Health Perspect 113:1791–1794. https://doi.org/10.1289/ehp.7883
Mordukhovich I, Lepeule J, Coull BA, Sparrow D et al (2015) The effect of oxidative stress polymorphisms on the association between long-term black carbon exposure and lung function among elderly men. Thorax 70:133–137. https://doi.org/10.1136/thoraxjnl-2014-206179
Nurmagambetov T, Kuwahara R, Garbe P (2018) The economic burden of asthma in the United States, 2008-2013. Ann Am Thorac Soc 15:348–356. https://doi.org/10.1513/AnnalsATS.201703-259OC
Ochs-Balcom HM, Grant BJ, Muti P, Sempos CT et al (2006) Antioxidants, oxidative stress, and pulmonary function in individuals diagnosed with asthma or COPD. Eur J Clin Nutr 60:991–999. https://doi.org/10.1038/sj.ejcn.1602410
Pernis AB, Rothman PB (2002) JAK-STAT signaling in asthma. J Clin Invest 109:1279–1283. https://doi.org/10.1172/jci15786
Peters JM, Avol E, Gauderman WJ, Linn WS et al (1999) A study of twelve Southern California communities with differing levels and types of air pollution. II. Effects on pulmonary function. Am J Respir Crit Care Med 159:768–775. https://doi.org/10.1164/ajrccm.159.3.9804144
Qibin L, Yacan L, Minli J, Meixi Z et al (2020) The impact of PM2.5 on lung function in adults with asthma. Int J Tuberc Lung Dis 24:570–576. https://doi.org/10.5588/ijtld.19.0394
Rice MB, Ljungman PL, Wilker EH, Dorans KS et al (2015) Long-term exposure to traffic emissions and fine particulate matter and lung function decline in the Framingham heart study. Am J Respir Crit Care Med 191:656–664. https://doi.org/10.1164/rccm.201410-1875OC
Stark GR, Cheon H, Wang YX (2018) Responses to cytokines and interferons that depend upon JAKs and STATs. Cold Spring Harb Perspect Biol 10:a028555. https://doi.org/10.1101/cshperspect.a028555
Thomas SJ, Snowden JA, Zeidler MP, Danson SJ (2015) The role of JAK/STAT signalling in the pathogenesis, prognosis and treatment of solid tumours. Br J Cancer 113:365–371. https://doi.org/10.1038/bjc.2015.233
Van Strien ME, Mercier D, Drukarch B, Brevé JJ et al (2010) Anti-inflammatory effect by lentiviral-mediated overexpression of IL-10 or IL-1 receptor antagonist in rat glial cells and macrophages. Gene Ther 17:662–671. https://doi.org/10.1038/gt.2010.8
Villarino AV, Kanno Y, O'Shea JJ (2017) Mechanisms and consequences of Jak-STAT signaling in the immune system. Nat Immunol 18:374–384. https://doi.org/10.1038/ni.3691
Wang KJ, Li B, Xie YY, Xia N et al (2020) Statin rosuvastatin inhibits apoptosis of human coronary artery endothelial cells through upregulation of the JAK2/STAT3 signaling pathway. Mol Med Rep 22:2052–2062. https://doi.org/10.3892/mmr.2020.11266
Wills-Karp M, Luyimbazi J, Xu X, Schofield B et al (1998) Interleukin-13: central mediator of allergic asthma. Science 282:2258–2261. https://doi.org/10.1126/science.282.5397.2258
Wurster AL, Tanaka T, Grusby MJ (2000) The biology of Stat4 and Stat6. Oncogene 19:2577–2584
Yue W, Chen X, He S, Li N et al (2021) Exposure interval to ambient fine particulate matter (PM2.5) collected in Southwest China induced pulmonary damage through the Janus tyrosine protein kinase-2/signal transducer and activator of transcription-3 signaling pathway both in vivo and in vitro. J Appl Toxicol 41:2042–2054. https://doi.org/10.1002/jat.4196
Zhang S, Lukacs NW, Lawless VA, Kunkel SL et al (2000) Cutting edge: differential expression of chemokines in Th1 and Th2 cells is dependent on Stat6 but not Stat4. J Immunol 165:10–14. https://doi.org/10.4049/jimmunol.165.1.10
Zhao C, Liao JP, Chu WL, Wang SX et al (2012) Involvement of TLR2 and TLR4 and Th1/Th2 shift in inflammatory responses induced by fine ambient particulate matter in mice. Inhal Toxicol 24:918–927. https://doi.org/10.3109/08958378.2012.731093
Acknowledgements
We are grateful to all staffs from three Community Healthcare Centers for their assistance in the investigation of asthmatic patients. We like to thank all the participants and investigators for their contribution to the study.
Funding
This study was supported by grants from the National Natural Science Foundation of China (81872589, 81072261), Scientific Research Foundation for Returned Scholars of Ministry of Education (2013-1792), and Research Funding Project for Returned Overseas Students in Shanxi Province (2013-057, 2017-060).
Author information
Authors and Affiliations
Contributions
NL conducted the analyses and drafted the manuscript; XM, JW, HG, BY, JH, GQ, and HL were involved in investigating the population and obtaining the data. LL, JT, XW, CW, DW, and HS revised the draft versions. ZZ designed this study and provided advice on the analyses and interpretation of the data, and revised the draft version of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The research proposal was approved by the Medical Ethics committee of Shanxi Medical University (2012033).
Consent to participate
Not applicable.
Consent to publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Exposure to PM2.5 in asthmatic patients disrupts the oxidation-antioxidant system balance and leads to increase of inflammatory cytokines.
• Mediating effect analyses show that JAK/STAT signaling molecules are involved in systemic oxidative stress and inflammation induced by PM2.5 exposure.
• The correlations between lag4 PM2.5 exposure and lung function index, systemic oxidative stress, systemic inflammation, and JAK/STAT signaling molecules are most remarkable.
Supplementary information
ESM 1
(PDF 1442 kb)
Rights and permissions
About this article
Cite this article
Liu, N., Ma, X., Wang, J. et al. Effects of ambient fine particulate matter on oxidative stress, inflammation, and janus kinase/signal transducer and activator of transcription signal molecules: a panel study of asthmatic adults in Taiyuan, China. Air Qual Atmos Health 15, 403–414 (2022). https://doi.org/10.1007/s11869-021-01139-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11869-021-01139-y