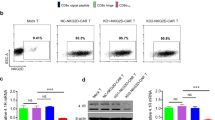
Abstract
Chimeric antigen receptor-modified T cell (CAR-T) therapy is a newly developed adoptive treatment for cancer in recent years that has shown potential for curative therapeutic efficacy in patients with hematological malignancies. However, the efficacy against solid tumors was very limited, the reasons are complex and have not been fully understood. In this study, CAR-T cells with 4-1BB or CD28 co-stimulatory molecules targeting carbonic anhydrase IX (CAIX) in the presence of 0, 10, 50, and 100 µM Docosahexaenoic acid (C22:6, DHA) has been used to treat renal cancer cells to illustrate the anti-tumor efficacy to solid tumor cells. The cytotoxicity of CAIX-CAR-T cells against cancer cells in the presence of DHA was determined by real-time cell assay (RTCA). Cytokines released by CAR-T cells were analyzed by Enzyme linked immunosorbent assay (ELISA). CAR-T cell apoptosis and exhaustion induced by DHA were determined by flow cytometry. Furthermore, signaling molecules involved in cell apoptosis and exhaustion were examined by Western blot analysis. This study showed that both 4-1BB and CD28-derived CAIX-CAR-T cells displayed efficient anti-tumor ability against renal carcinoma OSRC-2 cells. DHA attenuated cytotoxicity of CAR-T cells by inducing cell apoptosis and exhaustion in an opposite regulatory manner of AKT/PKB (protein kinase B) activation, which may provide a strategy to ameliorate CAR-T efficacy against solid tumors.




Similar content being viewed by others
Data availability
All raw data are obtained from devices and presented in GraphPad prism software.
Code availability
Not applicable.
References
Abramson HN (2020) B-Cell Maturation Antigen (BCMA) as a target for New Drug Development in Relapsed and/or refractory multiple myeloma. Int J Mol Sci 21. https://doi.org/10.3390/ijms21155192
Abramson JS, Palomba ML, Gordon LI, Lunning MA, Wang M, Arnason J, Mehta A, Purev E, Maloney DG, Andreadis C, Sehgal A, Solomon SR, Ghosh N, Albertson TM, Garcia J, Kostic A, Mallaney M, Ogasawara K, Newhall K, Kim Y, Li D, Siddiqi T (2020) Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet 396:839–852. https://doi.org/10.1016/S0140-6736(20)31366-0
Blank CU, Haining WN, Held W, Hogan PG, Kallies A, Lugli E, Lynn RC, Philip M, Rao A, Restifo NP, Schietinger A, Schumacher TN, Schwartzberg PL, Sharpe AH, Speiser DE, Wherry EJ, Youngblood BA, Zehn D (2019) Defining ‘T cell exhaustion’. Nat Rev Immunol 19:665–674. https://doi.org/10.1038/s41577-019-0221-9
Bonifant CL, Jackson HJ, Brentjens RJ, Curran KJ (2016) Toxicity and management in CAR T-cell therapy. Mol Ther Oncolytics 3:16011. https://doi.org/10.1038/mto.2016.11
Brown M, Bellon M, Nicot C (2007) Emodin and DHA potently increase arsenic trioxide interferon-alpha-induced cell death of HTLV-I-transformed cells by generation of reactive oxygen species and inhibition of akt and AP-1. Blood 109:1653–1659. https://doi.org/10.1182/blood-2006-04-015537
Cantrell DA (2002) T-cell antigen receptor signal transduction. Immunology 105:369–374. https://doi.org/10.1046/j.1365-2567.2002.01391.x
Endres S, Meydani SN, Ghorbani R, Schindler R, Dinarello CA (1993) Dietary supplementation with n-3 fatty acids suppresses interleukin-2 production and mononuclear cell proliferation. J Leukoc Biol 54:599–603
Fan YY, Ly LH, Barhoumi R, McMurray DN, Chapkin RS (2004) Dietary docosahexaenoic acid suppresses T cell protein kinase C theta lipid raft recruitment and IL-2 production. J Immunol 173:6151–6160. https://doi.org/10.4049/jimmunol.173.10.6151
Fraietta JA, Lacey SF, Orlando EJ, Pruteanu-Malinici I, Gohil M, Lundh S, Boesteanu AC, Wang Y, O’Connor RS, Hwang WT, Pequignot E, Ambrose DE, Zhang C, Wilcox N, Bedoya F, Dorfmeier C, Chen F, Tian L, Parakandi H, Gupta M, Young RM, Johnson FB, Kulikovskaya I, Liu L, Xu J, Kassim SH, Davis MM, Levine BL, Frey NV, Siegel DL, Huang AC, Wherry EJ, Bitter H, Brogdon JL, Porter DL, June CH, Melenhorst JJ (2018) Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med 24:563–571. https://doi.org/10.1038/s41591-018-0010-1
Fritsche KL, Anderson M, Feng C (2000) Consumption of eicosapentaenoic acid and docosahexaenoic acid impair murine interleukin-12 and interferon-gamma production in vivo. J Infect Dis 182(Suppl 1):S54–61. https://doi.org/10.1086/315925
Gao J, Fan M, Xiang G, Wang J, Zhang X, Guo W, Wu X, Sun Y, Gu Y, Ge H, Tan R, Qiu H, Shen Y, Xu Q (2017) Diptoindonesin G promotes ERK-mediated nuclear translocation of p-STAT1 (Ser727) and cell differentiation in AML cells. Cell Death Dis 8:e2765. https://doi.org/10.1038/cddis.2017.159
Goedert JJ, Sampson JN, Moore SC, Xiao Q, Xiong X, Hayes RB, Ahn J, Shi J, Sinha R (2014) Fecal metabolomics: assay performance and association with colorectal cancer. Carcinogenesis 35:2089–2096. https://doi.org/10.1093/carcin/bgu131
Gorjao R, Verlengia R, Lima TM, Soriano FG, Boaventura MF, Kanunfre CC, Peres CM, Sampaio SC, Otton R, Folador A, Martins EF, Curi TC, Portiolli EP, Newsholme P, Curi R (2006) Effect of docosahexaenoic acid-rich fish oil supplementation on human leukocyte function. Clin Nutr 25:923–938. https://doi.org/10.1016/j.clnu.2006.03.004
Guedan S, Alemany R (2018) CAR-T cells and oncolytic viruses: joining forces to overcome the solid Tumor Challenge. Front Immunol 9:2460. https://doi.org/10.3389/fimmu.2018.02460
Gutierrez S, Svahn SL, Johansson ME (2019) Effects of Omega-3 fatty acids on Immune cells. Int J Mol Sci 20. https://doi.org/10.3390/ijms20205028
Han D, Xu Z, Zhuang Y, Ye Z, Qian Q (2021) Current progress in CAR-T cell therapy for hematological malignancies. J Cancer 12:326–334. https://doi.org/10.7150/jca.48976
Kagi D, Vignaux F, Ledermann B, Burki K, Depraetere V, Nagata S, Hengartner H, Golstein P (1994) Fas and perforin pathways as major mechanisms of T cell-mediated cytotoxicity. Science 265:528–530. https://doi.org/10.1126/science.7518614
Kane LP, Weiss A (2003) The PI-3 kinase/Akt pathway and T cell activation: pleiotropic pathways downstream of PIP3. Immunol Rev 192:7–20. https://doi.org/10.1034/j.1600-065x.2003.00008.x
Kato T, Kolenic N, Pardini RS (2007) Docosahexaenoic acid (DHA), a primary tumor suppressive omega-3 fatty acid, inhibits growth of colorectal cancer independent of p53 mutational status. Nutr Cancer 58:178–187. https://doi.org/10.1080/01635580701328362
Kawalekar OU, O’Connor RS, Fraietta JA, Guo L, McGettigan SE, Posey AD Jr, Patel PR, Guedan S, Scholler J, Keith B, Snyder NW, Blair IA, Milone MC, June CH (2016) Distinct signaling of Coreceptors regulates specific metabolism pathways and impacts Memory Development in CAR T cells. Immunity 44:380–390. https://doi.org/10.1016/j.immuni.2016.01.021
Kim EH, Sullivan JA, Plisch EH, Tejera MM, Jatzek A, Choi KY, Suresh M (2012) Signal integration by akt regulates CD8 T cell effector and memory differentiation. J Immunol 188:4305–4314. https://doi.org/10.4049/jimmunol.1103568
Kim EH, Suresh M (2013) Role of PI3K/Akt signaling in memory CD8 T cell differentiation. Front Immunol 4:20. https://doi.org/10.3389/fimmu.2013.00020
Kochenderfer JN, Dudley ME, Feldman SA, Wilson WH, Spaner DE, Maric I, Stetler-Stevenson M, Phan GQ, Hughes MS, Sherry RM, Yang JC, Kammula US, Devillier L, Carpenter R, Nathan DA, Morgan RA, Laurencot C, Rosenberg SA (2012) B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood 119:2709–2720. https://doi.org/10.1182/blood-2011-10-384388
Labanieh L, Majzner RG, Mackall CL (2018) Programming CAR-T cells to kill cancer. Nat Biomed Eng 2:377–391. https://doi.org/10.1038/s41551-018-0235-9
Li W, Wu L, Huang C, Liu R, Li Z, Liu L, Shan B (2020) Challenges and strategies of clinical application of CAR-T therapy in the treatment of tumors-a narrative review. Ann Transl Med 8:1093. https://doi.org/10.21037/atm-20-4502
Long AH, Haso WM, Shern JF, Wanhainen KM, Murgai M, Ingaramo M, Smith JP, Walker AJ, Kohler ME, Venkateshwara VR, Kaplan RN, Patterson GH, Fry TJ, Orentas RJ, Mackall CL (2015) 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med 21:581–590. https://doi.org/10.1038/nm.3838
Malireddi RKS, Karki R, Sundaram B, Kancharana B, Lee S, Samir P, Kanneganti TD (2021) Inflammatory cell death, PANoptosis, mediated by Cytokines in Diverse Cancer Lineages inhibits Tumor Growth. Immunohorizons 5:568–580. https://doi.org/10.4049/immunohorizons.2100059
Monk JM, Hou TY, Turk HF, McMurray DN, Chapkin RS (2013) n3 PUFAs reduce mouse CD4 + T-cell ex vivo polarization into Th17 cells. J Nutr 143:1501–1508. https://doi.org/10.3945/jn.113.178178
Poorebrahim M, Melief J, Pico de Coana Y, Cid-Arregui SLW A and, Kiessling R (2021) Counteracting CAR T cell dysfunction. Oncogene 40:421–435. https://doi.org/10.1038/s41388-020-01501-x
Saka D, Gokalp M, Piyade B, Cevik NC, Arik Sever E, Unutmaz D, Ceyhan GO, Demir IE, Asimgil H (2020) Mechanisms of T-Cell exhaustion in pancreatic Cancer. Cancers (Basel) 12. https://doi.org/10.3390/cancers12082274
Sasaki T, Kanke Y, Kudoh K, Misawa Y, Shimizu J, Takita T (1999) Effects of dietary docosahexaenoic acid on surface molecules involved in T cell proliferation. Biochim Biophys Acta 1436:519–530. https://doi.org/10.1016/s0005-2760(98)00162-3
Schietinger A, Philip M, Krisnawan VE, Chiu EY, Delrow JJ, Basom RS, Lauer P, Brockstedt DG, Knoblaugh SE, Hammerling GJ, Schell TD, Garbi N, Greenberg PD (2016) Tumor-specific T cell dysfunction is a dynamic Antigen-Driven differentiation program initiated early during tumorigenesis. Immunity 45:389–401. https://doi.org/10.1016/j.immuni.2016.07.011
Shariati M, Meric-Bernstam F (2019) Targeting AKT for cancer therapy. Expert Opin Investig Drugs 28:977–988. https://doi.org/10.1080/13543784.2019.1676726
Shin YH, Tian X, Park JJ, Kim GYG, Aboujaoude E, Sturgill MG (2021) Management of chimeric antigen receptor T-cell induced cytokine release syndrome: current and emerging approaches. J Oncol Pharm Pract 10781552211039238. https://doi.org/10.1177/10781552211039238
Spencer L, Mann C, Metcalfe M, Webb M, Pollard C, Spencer D, Berry D, Steward W, Dennison A (2009) The effect of omega-3 FAs on tumour angiogenesis and their therapeutic potential. Eur J Cancer 45:2077–2086. https://doi.org/10.1016/j.ejca.2009.04.026
Sterner RC, Sterner RM (2021) CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J 11:69. https://doi.org/10.1038/s41408-021-00459-7
Switzer KC, McMurray DN, Chapkin RS (2004) Effects of dietary n-3 polyunsaturated fatty acids on T-cell membrane composition and function. Lipids 39:1163–1170. https://doi.org/10.1007/s11745-004-1343-y
Vermeulen K, Berneman ZN, Van Bockstaele DR (2003) Cell cycle and apoptosis. Cell Prolif 36:165–175. https://doi.org/10.1046/j.1365-2184.2003.00267.x
Wang S, Ma A, Song S, Quan Q, Zhao X, Zheng X (2008) Fasting serum free fatty acid composition, waist/hip ratio and insulin activity in essential hypertensive patients. Hypertens Res 31:623–632. https://doi.org/10.1291/hypres.31.623
Wang Z, Chen W, Zhang X, Cai Z, Huang W (2019) A long way to the battlefront: CAR T cell therapy against solid cancers. J Cancer 10:3112–3123. https://doi.org/10.7150/jca.30406
Xue M, Wang Q, Zhao J, Dong L, Ge Y, Hou L, Liu Y, Zheng Z (2014) Docosahexaenoic acid inhibited the Wnt/beta-catenin pathway and suppressed breast cancer cells in vitro and in vivo. J Nutr Biochem 25:104–110. https://doi.org/10.1016/j.jnutbio.2013.09.008
Yamagami T, Porada CD, Pardini RS, Zanjani ED, Almeida-Porada G (2009) Docosahexaenoic acid induces dose dependent cell death in an early undifferentiated subtype of acute myeloid leukemia cell line. Cancer Biol Ther 8:331–337. https://doi.org/10.4161/cbt.8.4.7334
Yan L, Lu L, Hu F, Shetti D, Wei K (2019) Piceatannol attenuates RANKL-induced osteoclast differentiation and bone resorption by suppressing MAPK, NF-kappaB and AKT signalling pathways and promotes Caspase3-mediated apoptosis of mature osteoclasts. R Soc Open Sci 6:190360. https://doi.org/10.1098/rsos.190360
Yoo JK, Cho JH, Lee SW, Sung YC (2002) IL-12 provides proliferation and survival signals to murine CD4 + T cells through phosphatidylinositol 3-kinase/Akt signaling pathway. J Immunol 169:3637–3643. https://doi.org/10.4049/jimmunol.169.7.3637
Zeyda M, Szekeres AB, Saemann MD, Geyeregger R, Stockinger H, Zlabinger GJ, Waldhausl W, Stulnig TM (2003) Suppression of T cell signaling by polyunsaturated fatty acids: selectivity in inhibition of mitogen-activated protein kinase and nuclear factor activation. J Immunol 170:6033–6039. https://doi.org/10.4049/jimmunol.170.12.6033
Zhang Q, Xu J, Ding J, Liu H, Li H, Li H, Lu M, Miao Y, Wang Z, Fu Q, Zheng J (2018) Bortezomib improves adoptive carbonic anhydrase IXspecific chimeric antigen receptormodified NK92 cell therapy in mouse models of human renal cell carcinoma. Oncol Rep 40:3714–3724. https://doi.org/10.3892/or.2018.6731
Acknowledgements
The authors thank Qianqian Dai, Cancer Institute, Xuzhou Medical University, for helping with flow cytometry operation, and data acquisition.
Funding
This work was supported by the National Natural Science Foundation of China [81773253, 81972242]; the Jiangsu Province Social Development Projects [BE2020641]; the Natural Science Foundation of Jiangsu Province [BK20211057]; the Natural Science Project of Jiangsu Provincial Education Department [19KJB310018]; the Research Foundation of Xuzhou Medical University [D2019023]; the Xuzhou Science and Technology Bureau projects [KC19058]; Youth Technology Innovation Team of Xuzhou Medical University [TD202003]; the Project of Invigorating Health Care through Science, Technology and Education [ZDXKA2016014]; National Demonstration Center for Experimental Basic Medical Science Education (Xuzhou Medical University) [202010313049Y].
Author information
Authors and Affiliations
Contributions
Conceptualization, SS.S., and JN.Z.; methodology, XG.G. and Q.Z.; software, XG.G.; validation, YF.W., YX.Z. and YZ.Z.; formal analysis, Q.Z.; investigation, YF.W., YX.Z., YZ.Z., H.X., WX.Z. and C.H., BX.W.; resources, YF.W., SS.S., JN.Z. and Q.Z.; data curation, YF.W.; writing—original draft preparation, YF.W.; writing—review and editing, YF.W., and SS.S.; visualization, JN.Z.; supervision, JN.Z. and SS.S.; project administration, JN.Z. and Q.Z.; funding acquisition, SS.S., YF.W., JN.Z. and Q.Z. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All procedures performed in studies involving human peripheral blood mononuclear cells were in accordance with the ethical standards of the Xuzhou Medical University, Jiangsu, P.R. China.
Consent to participate
All authors have confirmed their participation in this study.
Consent to publish
All authors have agreed to publish this manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yifei Wang, Yuxiao Zhao, Yizhang Zhu thease authors are shared first authorship.
Electronic supplementary material
Below is the link to the electronic supplementary material.


Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Zhao, Y., Zhu, Y. et al. DHA attenuates CAR-T cell efficacy through aggravating exhaustion and inducing apoptosis. Biologia 78, 1141–1152 (2023). https://doi.org/10.1007/s11756-023-01326-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-023-01326-5