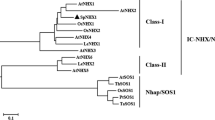
Abstract
d-Sorbitol-6-phosphate 2-dehydrogenase (S6PDH, E.C. 1.1.1.140) catalyzes the NADH-dependent conversion of d-fructose 6-phosphate (F6P) to d-sorbitol 6-phosphate (S6P). In this work, recombination and characterization of Haloarcula marismortui d-sorbitol-6-phosphate 2-dehydrogenase are reported. Haloarcula marismortui d-sorbitol-6-phosphate 2-dehydrogenase was expressed in P. pastoris and Arabidopsis thaliana. Enzyme assay indicated that HmS6PDH catalyzes the reduction of d-fructose 6-phosphate to d-sorbitol 6-phosphate and HmS6PDH activity was enhanced by NaCl. Furthermore, transgenic A. thaliana ectopic expressing HmS6PDH accumulate more sorbitol under salt stress. These results suggest that the ectopic expression of HmS6PDH in plants can facilitate future studies regarding the engineering and breeding of salt-tolerant crops.






Similar content being viewed by others
Abbreviations
- S6PDH:
-
Sorbitol-6-phosphate 2-dehydrogenase
- HmS6PDH:
-
Haloarcula marismortui sorbitol-6-phosphate 2-dehydrogenase
- SDH:
-
Sorbitol dehydrogenase
- F6P:
-
d-Fructose 6-phosphate
- S6P:
-
d-Sorbitol 6-phosphate
- MDA:
-
Malondialdehyde
References
Bianco RL, Rieger M, Sung SS (2000) Effect of drought on sorbitol and sucrose metabolism in sinks and sources of peach. Physiol Plant 108(1):71–78
Bohnert HJ, Nelson DE, Jensen RG (1995) Adaptations to environmental stresses. Plant Cell 7(7):1099–1111
Chen GC, Ren L, Zhang J, Reed BM, Zhang D, Shen XH (2015) Cryopreservation affects ROS-induced oxidative stress and antioxidant response in Arabidopsis seedlings. Cryobiology 70(1):38–47
Cheng Y, Tian Q, Zhang WH (2016) Glutamate receptors are involved in mitigating effects of amino acids on seed germination of Arabidopsis thaliana under salt stress. Environ Exp Bot 130:68–78
Choudhury FK, Rivero RM, Blumwald E, Mittler R (2017) Reactive oxygen species, abiotic stress and stress combination. Plant J 90(5):856–867
Christian JH, Waltho JA (1962) Solute concentrations within cells of halophilic and non-halophilic bacteria. Biochem Biophys Acta 65:506–508
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16(6):735–743
Gao M, Tao R, Miura K, Dandekar AM, Sugiura A (2001) Transformation of Japanese persimmon (Diospyros kaki Thunb.) with apple cDNA encoding NADP-dependent sorbitol-6-phosphate dehydrogenase. Plant Sci 160(5):837–845
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Kanamaru N, Ito Y, Komori S, Saito M, Kato H, Takahashi S, Omura M, Soejima J, Shiratake K, Yamada K, Yamaki S (2004) Transgenic apple transformed by sorbitol-6-phosphate dehydrogenase cDNA Switch between sorbitol and sucrose supply due to its gene expression. Plant Sci 167:55–61
Kanayama Y, Mori H, Imaseki H, Yamaki S (1992) Nucleotide sequence of a cDNA encoding NADP-sorbitol-6-phosphate dehydrogenase from apple. Plant Physiol 100(3):1607–1608
Kim HY, Ahn JC, Choi JH, Hwang B, Choi DW (2007) Expression and cloning of the full-length cDNA for sorbitol-6-phosphate dehydrogenase and NAD-dependent sorbitol dehydrogenase from pear (Pyrus pyrifolia N.). Sci Hortic 112:406–412
Kobashi K, Gemma H, Iwahori S (2000) Abscisic acid content and sugar metabolism of peaches grown under water stress. J Am Soc Hortic Sci 125(4):425–428
Kurth E, Cramer GR, Läuchli A, Epstein E (1986) Effects of NaCl and CaCl2 on cell enlargement and cell production in cotton roots. Plant Physiol 82(4):1102–1106
Lengeler J, Lin EC (1972) Reversal of the mannitol-sorbitol diauxie in Escherichia coli. J Bacteriol 112:840–848
Liss M, Horwitz SB, Kaplan NO (1962) d-Mannitol-l-phosphate dehydrogenase and d-sorbitol-6-phosphate dehydrogenase in Aerobacter aerogenes. J Biol Chem 237:1342–1350
Liu W, Li RJ, Han TT, Cai W, Fu ZW, Lu YT (2015) Salt stress reduces root meristem size by nitric oxide-mediated modulation of auxin accumulation and signaling in Arabidopsis. Plant Physiol 168(1):343–356
Mevarech M, Frolow F, Gloss LM (2000) Halophilic enzymes: proteins with a grain of salt. Biophys Chem 86:155–164
Mundree SG, Farrant JM (2000) Some physiological and molecular insights into the mechanisms of desiccation tolerance in the resurrection plant Xerophyta viscosa Baker. Plant Toler Abiotic Stresses Agric Role Genet Eng 83:201–222
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nosarzewski M, Downie AB, Wu B, Archbold DD (2012) The role of sorbitol dehydrogenase in Arabidopsis thaliana. Funct Plant Biol 39:462–470
Oura Y, Yamada K, Shiratake K, Yamaki S (2000) Purification and characterization of a NAD+-dependent sorbitol dehydrogenase from Japanese pear fruit. Phytochemistry 54(6):567–572
Parihar P, Singh S, Singh R, Singh VP, Prasad SM (2015) Effect of salinity stress on plants and its tolerance strategies: a review. Environ Sci Pollut Res 22(6):4056–4075
Pharr DM, Stoop JMH, Williamson JD, Feusi MES, Massel MO, Conkling MA (1995) The dual role of mannitol as osmoprotectant and photoassimilate in celery. Am Soc Hortic Sci 30(6):1151–1192
Qiu ZB, Wang YF, Zhu AJ, Peng FL, Wang LS (2014) Exogenous sucrose can enhance tolerance of Arabidopsis thaliana seedlings to salt stress. Biol Plant 58(4):611–617
Saleki R, Young P, Lefebvre DD (1993) Mutants of Arabidopsis thaliana capable of germination under saline conditions. Plant Physiol 101:839–845
Sheveleva E, Marquez S, Chmara W, Wegeer A, Jensen RG, Bohnert HJ (1998) Sorbitol-6-phosphate dehydrogenase expression in transgenic tobacco. Plant Physiol 117:831–839
Shimada TL, Shimada T, Takahashi H, Fukao Y, Hara-Nishimura I (2008) A novel role for oleosins in freezing tolerance of oilseeds in Arabidopsis thaliana. Plant J 55(5):798–809
Singh M, Kumar J, Singh S, Singh VP, Prasad SM (2015) Roles of osmoprotectants in improving salinity and drought tolerance in plants: a review. Rev Environ Sci Bio/Technol 14(3):407–426
Umezawa T, Mizuno K, Fujimura T (2002) Discrimination of genes expressed in response to the ionic or osmotic effect of salt stress in soybean with cDNA-AFLP. Plant Cell Environ 25(12):1617–1625
West G, Inze D, Beemster GTS (2004) Cell cycle modulation in the response of the primary root of Arabidopsis to salt stress. Plant Physiol 135:1050–1058
Wu S, Letchworth GJ (2004) High efficiency transformation by electroporation of Pichia pastoris pretreated with lithium acetate and dithiothreitol. Biotechniques 36:152–154
Xiong AS, Yao QH, Peng RH, Li X, Fan HQ, Cheng ZM, Li Y (2004) A simple, rapid, high-fidelity and cost-effective PCR-based two-step DNA synthesis method for long gene sequence. Nucleic Acids Res 32:e98
Xiong AS, Yao QH, Peng RH, Duan H, Li X, Fan HQ, Cheng ZM, Li Y (2006) PCR-based accurate synthesis of long DNA sequences. Nat Protoc 1:791–797
Xu ZS, Xue W, Xiong AS, Lin YQ, Xu J, Zhu B, Zhao W, Peng RH, Yao QH (2013) Characterization of a bifunctional O- and N-Glucosyltransferase from Vitis vinifera in glucosylating phenolic compounds and 3,4-dichloroaniline in Pichia pastoris and Arabidopsis thaliana. PLoS One 8(11):e80449
Yamaki S, Ishikawa K (1986) Roles of four sorbitol related enzymes and invertase in the seasonal alteration of sugar metabolism in apple tissue. J Am Soc Hortic Sci 111:134–137
Zhou R, Cheng LL, Wayne R (2003) Purification and characterization of sorbitol-6-phosphate phosphatase from apple leaves. Plant Sci 165:227–232
Zhou S, Tang QJ, Zhang Z, Li CH, Cao H, Yang Y, Zhang JS (2015) Nutritional composition of three domesticated culinary-medicinal mushrooms: Oudemansiella sudmusida, Lentinus squarrosulus, and Tremella aurantialba. Int J Med Mushrooms 17(1):43–49
Acknowledgements
This research was sponsored by Shanghai Sailing Program(15YF1410400), the Young Growing Project Fund of the Shanghai Municipal Committee of Agriculture (2015, 1-27), the run-up project of the Shanghai Academy of Agricultural Sciences (ZP18), the Key Project Fund of the Shanghai Municipal Committee of Agriculture (zhongzi2014-2, zhongzi2014 7-1-3, zhongzi2016 1-2), the Agriculture Science Technology Achievement Transformation Fund (143919N0300), the National Natural Science Foundation (31200212, 31200075, 31200076, and 31672439), and the Basic Research in the Field of Science and Technology Project of Science and Technology Commission of the Shanghai Municipality (14JC1403602).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Q. Wang.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11738_2018_2668_MOESM2_ESM.jpg
Analysis of transgenic HmS6PDH lines. (A) Schematic of the vector used in this study. The vector contains a double CaMV 35S (DCaMV35S) promoter and the tobacco mosaic virus (TMV) sequence fused to the HmSorDI gene. For steady transcription of the HmSorDI gene, two scaffold attachment regions (SAR) were fused upstream of the DCaMV35S promoter and downstream of the Nos-terminator (Nos-T). (B) Analysis by PCR of HmSorDI expression in Arabidopsis transgenic lines. Wild-type (lane 5) and transgenic lines (line 3, line 4, and lines 6–8) of A. thaliana were used as PCR templates. Line 3 represents HT-1, and lines 6 and 7 represent HT-8 and HT-9, respectively. Line 2 represents a water-only control. Specific primer pairs for PCR amplification of HmSorDI were used. (JPEG 60 KB)
Rights and permissions
About this article
Cite this article
Gao, Jj., Sun, Yr., Zhu, B. et al. Ectopic expression of sorbitol-6-phosphate 2-dehydrogenase gene from Haloarcula marismortui enhances salt tolerance in transgenic Arabidopsis thaliana. Acta Physiol Plant 40, 108 (2018). https://doi.org/10.1007/s11738-018-2668-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-018-2668-x