Abstract
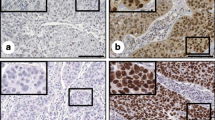
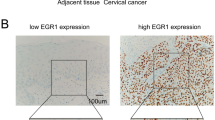
Cancer stem cells (CSCs) are considered responsible for the high recurrence rate in cervical carcinoma. It has been demonstrated that the signal transducer and activator of transcription 3 (STAT3) is involved in the oncogenesis and takes part in mediating the effects of maintaining stem cell phenotype and pluripotency by regulating the expression of stem cell-related transcription factors. However, the correlation between STAT3 and stem cell-related transcription factors in cervical cancer has not been elucidated. In this study, we established overexpressing plasmid (GV316-STAT3) and siRNA-STAT3 for transfecting Siha cells. Cells negative or positive for Nanog, Oct4, or Sox2 were selected by flow cytometry. Proliferation and differentiation rate of Siha cells was determined by detecting the efficiency of tumor sphere formation. The expression of Nanog, Oct4 and Sox2 (cancer stem cell markers) and STAT3 was detected by quantitative real-time PCR and immunoblotting for Siha cells and by immunohistochemistry (IHC) for cervical tissues, respectively. The results showed that Nanog+, Oct4+, and Sox2+ Siha-STAT3 over-expressing cells displayed the typical non-adherent spheres. The sphere formation efficiency was significantly different between Siha-STAT3 overexpressing cells and siRNA-STAT3 cells (P<0.05). Meanwhile, the expression levels of Oct4, Nanog and Sox2 mRNA and protein were significantly higher in Siha-STAT3 overexprssing cells than in siRNA-STAT3 cells (P<0.05). In addition, the positive rate of STAT3, Nanog, Oct4 and Sox2 in cervical cancer tissues was higher than that in chronic cervicitis group (P<0.05). There was a significantly positive relationship between STAT3 and Nanog or Oct4 or Sox2 expression (all P<0.001). These results suggested that Oct4+, Sox2+, and Nanog+ cell population possesses stem cell properties in cervical cancer, which may contribute to cervical carcinogenesis and be regulated by STAT3.
Similar content being viewed by others
References
Feng D, Peng C, Li CR, et al. Identification and characterization of cancer stem-like cells from primary carcinoma of the cervix uteri. Oncol Rep, 2009,22(5):1129–1134
Bortolomai I, Canevari S, Facetti I, et al. Tumor initiating cells: development and critical characterization of a model derived from the A431 carcinoma cell line forming spheres in suspension. Cell Cycle, 2010,9(6):1194–1206
Takebe N, Harris PJ, Warren RQ, et al. Targeting cancer stem cells by inhibiting Wnt, Notch, and Hedgehog pathways. Nat Rev Clin Oncol, 2011,8(2):97–106
Song L, Turkson J, Karras J, et al. Activation of Stat3 by receptor tyrosine kinases and cytokines regulates survival in human non-small cell carcinoma cells. Oncogene, 2003,22(27):4150–4165
Zhang P, Li H, Yang B, et al. Biological significance and therapeutic implication of resveratrol-inhibited Wnt, Notch and STAT3 signaling in cervical cancer cells. Genes Cancer, 2014,5(5-6):154–164
Kruczyk M, Przanowski P, Dabrowski M, et al. Integration of genome-wide of Stat3 binding and epigenetic modification mapping with transcriptome reveals novel Stat3 target genes in glioma cells. Biochim Biophys Acta, 2014,1839(11):1341–1350
Yao J, Qian CJ, Ye B, et al. Signal transducer and activator of transcription 3 signaling upregulates fascin via nuclear factor-kappaB in gastric cancer: Implications in cell invasion and migration. Oncol Lett, 2014,7(3):902–908
Bao W, Wang HH, Tian FJ, et al. A TrkB-STAT3-mir-204-5pregulatory circuitry controls proliferation and invasion of endometrial carcinoma cells. Mol Cancer, 2013,12:155
Cheng GZ, Zhang WZ, Sun M, et al. Twist is transcriptionally induced by activation of STAT3 and mediates STAT3 oncogenic function. J Biol Chem, 2008,283(21): 14 665–14 673
Qin A, Yu Q, Gao Y, et al. Inhibition of STAT3/cyclinD1 pathway promotes chemotherapeutic sensitivity of colorectal cancer. Biochem Biophys Res Commun, 2015,457(4):681–687
Takemoto S, Ushijima K, Kawano K, et al. Transcription-3 predicts poor prognosis in cervical squamous cell carcinoma. Br J Cancer, 2009,101(9):967–972
Chambers I, Colby D, Robertson M, et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell, 2003,113(5):643–655
Tsai LL, Yu CC, Chang YC, et al. Markedly increased Oct4 and Nanog expression correlates with cisplatin resistance in oral squamous cell carcinoma. J Oral Pathol Med, 2011,40(8):621–628
Wang Z, Oron E, Nelson B, et al. Distinct lineage specification roles for NANOG, 0CT4, and S0X2 in human embryonic stem cells. Cell Stem, 2012,10(4):440–454
Bourguignon LY, Peyrollier K, Xia W, et al. Hyaluronan-CD44 interaction activates stem cell marker Nanog, Stat-3-mediated MDR1 gene expression, and ankyrin-regulated multidrug efflux in breast and ovarian tumor cells. J Biol Chem, 2008,283(25):17 635–17 651
Do DV, Ueda J, Messerschmidt DM, et al. A genetic and developmental pathway from STAT3 to the OCT4-NANOG circuit is essential for maintenance of ICM lineages in vivo. Genes Dev, 2013,27(12):1378–1390
Li YQ. Master stem cell transcription factors and signaling regulation. Cell Reprogram, 2010,12(1):3–13
Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin, 2011,61(2):69–90
Devarajan E, Huang S. STAT3 as a central regulator of tumor metastases. Curr Mol Med, 2009,9(5):626–633
Cao C, Zhao GW, Yu W, et al. Activation of STAT3 stimulates AHSP expression in K562 cells. Sci China Life Sci, 2014,57(5):488–494
Yao J, Qian CJ, Ye B, et al. Signal transducer and activator of transcription 3 signaling upregulates fascin via nuclear factor-kappaB in gastric cancer: Implications in cell invasion and migration. Oncol Lett, 2014,7(3):902–908
Chiou SH, Yu CC. Positive correlations of Oct-4 and Nanog in oral cancer stem-like cells and high-grade oral squamous cell carcinoma. Clin Cancer Res, 2008, 14(13):4085–4095
Liu XF, Yang WT, Xu R, et al. Cervical cancer cells with positive Sox2 expression exhibit the properties of cancer stem cells. PLoS One, 2014,9(1):870–892
Ye F, Zhou C, Cheng Q, et al. Stem-cell-abundant proteins Nanog, Nucleostemin and Musashil are highly expressed in malignant cervical epithelial cells. BMC Cancer, 2008,18(8):108
Li SW, Wu XL, Dong CL, et al. The differential expression of OCT4 isoforms in cervical carcinoma. PLoS One, 10(3):1-15
Author information
Authors and Affiliations
Corresponding author
Additional information
This was supported by the National Natural Science Foundation of China (No. 81272866), and the Wuhan Municipal Science and Technology Bureau Foundation of China (No. 201460101010043)
Rights and permissions
About this article
Cite this article
Wang, H., Cai, Hb., Chen, Ll. et al. STAT3 correlates with stem cell-related transcription factors in cervical cancer. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 35, 891–897 (2015). https://doi.org/10.1007/s11596-015-1524-0
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-015-1524-0