Abstract
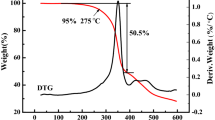
A novel PEO-based blends solid polymer electrolytes doping liquid crystalline ionomers (LCI), PEO/PMMA/LiClO4/LCI, and PEO/LiClO4/LCI were prepared by solution casting technology. Scanning electron microscope (SEM) and energy-dispersive spectroscopy (EDS) analysis proved that LCI uniformly dispersed into the solid electrolytes and restrained phase separation of PEO and PMMA. Differential scanning calorimetry (DSC) results showed that LCI decreases the crystallinity of blends solid polymer electrolytes. Thermogravimetric analysis (TGA) proved LCI not only improved thermal stability of PEO/PMMA/LiClO4 blends but also prevent PEO/PMMA from phase separation. Infrared spectra results illustrated that there exists interaction among Li+ and O, and LCI that promotes the synergistic effects between PEO and PMMA. The EIS result revealed that the conductivity of the electrolytes increases with LiClO4 concentration in PEO/PMMA blends, but it increases at first and reaches maximum value of 2.53 × 10−4 S/cm at 1.0 % of LCI. The addition of 1.0 % LCI increases the conductivity of the electrolytes due to that LCl promoting compatibility and interaction of PEO and PMMA. Under the combined action of rigidity induced crystal unit, soft segment and the terminal ionic groups in LCI, PEO/PMMA interfacial interaction are improved, the reduction of crystallinity degree of PEO leads Li+ migration more freely.
















Similar content being viewed by others
References
Nazri GA, Pistoia G (2008) Lithium batteries: science and technology. Springer Science & Business
Scrosati B, Garche J (2010) J Power Sources 195(9):2419
Scrosati B, Croce F, Persi L (2000) J Electrochem Soc 147(5):1718
Fenton D, Parker J, Wright P (1973) Polymer 14(11):589
Xu X, Wang Q (1991) Chem J Chin Univ 12(3):413
Cai F, Zuo X, Liu XM (2013) Electrochim Acta 106:209
Radhakrishnan S, Venkatachalapathy P (1996) Polymer 37(16):3749
Imrie CT, Ingram MD, Mchattie GS (1999) Adv Mater 11(10):832
Nayak GC, Das CK (2016) Liquid Crystalline Polymers: LCP Based Polymer Blend Nanocomposites. Springer International Publishing, p251
Li Y, Zhang BY, Feng Z (2002) J Appl Polym Sci 83(13):2749
Qu WZ, Xu XY, Chu HZ, Zhang BY (2011) Polym Mater Sci Eng 27(2):107
Xu XY, Zhou ZL (2015) Appl Mech Mater 751:21–25
Li YM, Zhang BY, Wang J (2002) J Funct Polym 15(1):1
Stoeva Z, Lu Z, Ingram MD (2013) Electrochim Acta 93:p279
Xia Y, Wang S, Ma N (2014) China Synth Resin Plast 31(2):25
Donald AM, Windle AH, Hanna S (2006) Liquid Crystalline Polymers: Liquid crystalline polymers in blends and composites (Cambridge University Press), p483
Zhang AL (2002) Main-Chain Liquid Crystalline Ionomer and Composite Materials. Ph.D., Northeastern University, China
Liu Q, Pan C, Shen S (2006) Chin J Nonferrous Met 16(2):377
Wang Y, Li M, Rong J (2013) Colloid Polym Sci 291(6):1541
Su YL, Liu HZ, Guo C (2003) Mol Simul 29(12):803
Ramesh S, Yuen TF, Shen CJ (2008) Spectrochim Acta A Mol Biomol Spectrosc 69(2):670
Ghelichi M, Qazvini NT, Jafari SH (2013) J Appl Polym Sci 129(4):1868
Chen N, Yan LT, Xie XM (2013) Macromolecules 46(9):3544
Schwahn D, Pipich V, Richter D (2012) Macromolecules 45(4):2035
Shi W, Yang J, Zhang Y (2012) Macromolecules 45(2):941
Angulakshmi N, Thomas S, Nahm KS (2011) Ionics 17(5):407
Toshimi H, Takashi K, James EB (1985) Macromolecules 18:1410
Acknowledgments
Liaoning Provincial Key Laboratory for Polymer Catalytic Synthesis Technology (Document No.36 by DST, Liaoning Province [2010].); Advanced Polymer Materials Engineering Laboratory in Liaoning province (2012.5); and Shenyang Science and Technology plan project (F14-231-1-28) are acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Al., Cao, Fy., Na, Gz. et al. A novel PEO-based blends solid polymer electrolytes doping liquid crystalline ionomers. Ionics 22, 2103–2112 (2016). https://doi.org/10.1007/s11581-016-1732-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-016-1732-z