Abstract
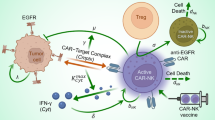
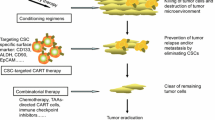
The stem cell hypothesis suggests that there is a small group of malignant cells, the cancer stem cells, that initiate the development of tumors, encourage its growth, and may even be the cause of metastases. Traditional treatments, such as chemotherapy and radiation, primarily target the tumor cells leaving the stem cells to potentially cause a recurrence. Chimeric antigen receptor (CAR) T-cell therapy is a form of immunotherapy where the immune cells are genetically modified to fight the tumor cells. Traditionally, the CAR T-cell therapy has been used to treat blood cancers and only recently has shown promising results against solid tumors. We create an ordinary differential equations model which allows for the infusion of trained CAR-T cells to specifically attack the cancer stem cells that are present in the solid tumor microenvironment. Additionally, we incorporate the influence of TGF-\(\beta \) which inhibits the CAR-T cells and thus promotes the growth of the tumor. We verify the model by comparing it to available data and then examine combinations of CAR-T cell treatment targeting both non-stem and stem cancer cells and a treatment that reduces the effectiveness of TGF-\(\beta \) to determine the scenarios that eliminate the tumor.












Similar content being viewed by others
Data Availability
Data are available upon request.
References
Arciero TL, Kirschner DE (2004) A mathematical model of tumor-immune evasion and sirna treatment. Dis Contin Dyn Syst 4:39–58. https://doi.org/10.3934/dcdsb.2004.4.39
Bashraheel SS, Domling A, Goda SK (2020) Update on targeted cancer therapies, single or in combination, and their fine tuning for precision medicine. Biomedicine & Pharmacotherapy 125:110009. https://doi.org/10.1016/j.biopha.2020.110009
Beretta E, Capasso V, Morozova N (2012) Mathematical modelling of cancer stem cells population behavior. Math Modell Nat Phenom 7(1):279–305. https://doi.org/10.1051/mmnp/20127113
Clara J, Monge C, Yang Y, Takebe N (2020) Targeting signalling pathways and the immune microenvironment of cancer stem cells - a clinical update. Nat Rev Clin Oncol 17(4):204. https://doi.org/10.1038/s41571-019-0293-2
Elliott SL, Kose E, Lewis AL, Steinfeld AE, Zollinger EA (2019) Modeling the stem cell hypothesis: investigating the effects of cancer stem cells and TGF-\(\beta \) on tumor growth. Math Biosci Eng: MBE 16(6):7177–7194. https://doi.org/10.3934/mbe.2019360
Eyvazi S, Farajnia S, Dastmalchi S, Kanipour F, Zarredar H, Bandehpour M (2018) Antibody based EpCAM targeted therapy of cancer, review and update. Curr Cancer Drug Targets 18(9):857–868. https://doi.org/10.2174/1568009618666180102102311
Fasano A, Mancini A, Primicerio M (2016) Tumours with cancer stem cells: a pde model. Math Biosci 272:76–80
Han Y, Sun B, Cai H, Xuan Y (2021) Simultaneously target of normal and stem cells-like gastric cancer cells via cisplatin and anti-CD133 CAR-T combination therapy. Cancer Immunol Immunother. https://doi.org/10.1007/s00262-021-02891-x
Hillen T, Enderling H, Hahnfeldt P (2013) The tumor growth paradox and immune system-mediated selection for cancer stem cells. Bull Math Biol 75(1):161–184
Hou AJ, Chang ZL, Lorenzini MH, Zah E, Chen YY (2018) TGF-\(\beta \)-responsive CAR-T cells promote anti-tumor immune function. Bioeng Trans Med 3(2):75–86. https://doi.org/10.1002/btm2.10097
Johnston MD, Maini PK, Chapman SJ, Edwards CM, Bodmer WF (2010) On the proportion of cancer stem cells in a tumour. J Theor Biol 266(4):708–711
Köse E, Moore S, Ofodile C, Radunskaya A, Swanson ER, Zollinger E (2017) Immuno-kinetics of immunotherapy: dosing with dcs. Lett Biomath 4(1):39–58
Lee Y, Tan Y, Oon C (2018) Molecular targeted therapy: treating cancer with specificity. Eur J Pharmacol 834:188–196. https://doi.org/10.1016/j.ejphar.2018.07.034
Liou G (2019) Cd133 as a regulator of cancer metastasis through the cancer stem cells. Int J Biochem Cell Biol 106:1–7. https://doi.org/10.1016/j.biocel.2018.10.013
Liu X, Johnson S, Liu S, Kanojia D, Yue W, Singh UP, Wang Q, Wang Q, Nie Q, Chen H (2013) Nonlinear growth kinetics of breast cancer stem cells: implications for cancer stem cell targeted therapy. Sci Rep 3(1):2473. https://doi.org/10.1038/srep02473
Louzoun Y, Xue C, Lesinski GB, Friedman A (2014) A mathematical model for pancreatic cancer growth and treatments. J Theor Biol 351:74–82. https://doi.org/10.1016/j.jtbi.2014.02.028
Marino S, Hogue IB, Ray CJ, Kirschner DE (2008) A methodology for performing global uncertainty and sensitivity analysis in systems biology. J Theor Biol 254(1):178–196. https://doi.org/10.1016/j.jtbi.2008.04.011
Marotta LLC, Polyak K (2009) Cancer stem cells: a model in the making. Curr Op Gen Dev 19:44–50. https://doi.org/10.1016/j.gde.2008.12.003
Moo-Young T, Larson J, BA B, Tan M, Hawkins W, Eberlein T, Goedegebuure P, Linehan D (2009) Tumor-derived tgf-\(\beta \) mediates conversion of cd4+foxp3+ regulatory t cells in a murine model of pancreas cancer. J Immunoth 32(1):12–21. https://doi.org/10.1097/CJI.0b013e318189f13c
Morganti S, Tarantino P, Ferraro E, D’Amico P, Duso B, Curigliano G (2019) Next generation sequencing (NGSd): a revolutionary technology in pharmacogenomics and personalized medicine in cancer. Adv Exp Med Biol 1168:9–30. https://doi.org/10.1007/978-3-030-24100-1_2
Mortezaee K (2020) Immune escape: a critical hallmark in solid tumors. Life Sci 258:118110
National Cancer Institute (2020) Cancer statistics. https://www.cancer.gov/about-cancer/understanding/statistics
Piotrowska MJ, Enderling H, van der Heiden U, Mackey MC (2008) Mathematical modeling of stem cells related to cancer. Complex Syst Biomed In: Cancer and Stem Cells, Edited by Thomas Dittmar and Kurt S. Zänker, Nova Science Publishers, Inc., ISBN 978-1-60456-478-5.
Polanczyk M, Walker E, Haley D, Guerrouahen B, Akporiaye E (2019) Blockade of TGF-\(\beta \) signaling to enhance the antitumor response is accompanied by dysregulation of the functional activity of CD4+CD25+Foxp3+ and CD4+CD25-Foxp3+ T cells. J Trans Med 16(1):1–16. https://doi.org/10.1186/s12967-019-1967-3
Radunskaya A, Kim R, Woods T II (2018) Mathematical modeling of tumor immune interactions: a closer look at the role of a PD-L1 inhibitor in cancer immunotherapy. Spora J Biomath 4(1):25–41
Renardy M, Jilkine A, Shahriyari L, Chou CS (2018) Control of cell fraction and population recovery during tissue regeneration in stem cell lineages. J Theor Biol 445:33–50. https://doi.org/10.1016/j.jtbi.2018.02.017
Rodriguez-Brenes IA, Kurtova AV, Lin C, Lee YC, Xiao J, Mims M, Chan KS, Wodarz D (2017) Cellular hierarchy as a determinant of tumor sensitivity to chemotherapy. Cancer Res 77(9):2231. https://doi.org/10.1158/0008-5472.CAN-16-2434
Sahoo P, Yang X, Abler D, Maestrini D, Adhikarla V, Frankhouser D, Cho H, Machuca V, Wang D, Barish M, Gutova M, Branciamore S, Brown CE, Rockne RC (2020) Mathematical deconvolution of CAR T-cell proliferation and exhaustion from real-time killing assay data. J R Soc Interface 17(162):20190734
Scarfò I, Maus M (2017) Current approaches to increase CAR T cell potency in solid tumors: targeting the tumor microenvironment. J Immunoth Cancer 5:28. https://doi.org/10.1186/s40425-017-0230-9
Sigal D, Przedborski M, Sivaloganathan D, Kohandel M (2019) Mathematical modelling of cancer stem cell-targeted immunotherapy. Math Biosci 318:108269. https://doi.org/10.1016/j.mbs.2019.108269
Soetaert K, Petzoldt T (2010) Inverse modelling, sensitivity and monte carlo analysis in R using package FME. J Stat Softw 33(3):1–28
Tang N, Cheng C, Zhang X, Qiao M, Li N, Mu W, Wei XF, Han W, Wang H (2020) TGF-\(\beta \) inhibition via CRISPR promotes the long-term efficacy of CAR T cells against solid tumors. JCI Insight. https://doi.org/10.1172/jci.insight.133977
Tomasetti C, Vogelstein B (2015) Variation in cancer risk among tissues can be explained by the number of stem cell divisions. Science 347(6217):78–81. https://doi.org/10.1126/science.1260825
Wang J, Lei K, Han F (2018) Tumor microenvironment: recent advances in various cancer treatments. Eur Rev Med Pharmacol Sci 22(12):3855–3864
Weekes SL, Barker B, Bober S, Cisneros K, Cline J, Thompson A, Hlatky L, Hahnfeldt P, Enderling H (2014) A multicompartment mathematical model of cancer stem cell-driven tumor growth dynamics. Bull Math Biol 76(7):1762–1782
Weiss LD, Komarova NL, Rodriguez-Brenes IA (2017) Mathematical modeling of normal and cancer stem cells. Curr Stem Cell Rep 3(3):232–239
Wilson S, Levy D (2012) A mathematical model of the enhancement of tumor vaccine efficacy by immunotherapy. Bull Math Biol 74(7):1485–1500. https://doi.org/10.1007/s11538-012-9722-4
Wodarz D (2018) Effect of cellular de-differentiation on the dynamics and evolution of tissue and tumor cells in mathematical models with feedback regulation. J Theor Biol 448:86–93. https://doi.org/10.1016/j.jtbi.2018.03.036
Youssefpour H, Li X, Lander AD, Lowengrub JS (2012) Multispecies model of cell lineages and feedback control in solid tumors. J Theor Biol 304:39–59. https://doi.org/10.1016/j.jtbi.2012.02.030
Zhang BL, Li D, Gong YL, Huang Y, Qin DY, Jiang L, Liang X, Yang X, Gou HF, Wang YS, Wei YQ, Wang W (2019) Preclinical evaluation of chimeric antigen receptor-modified t cells specific to epithelial cell adhesion molecule for treating colorectal cancer. Hum Gene Ther 30(4):402–412. https://doi.org/10.1089/hum.2018.229
Zheng Y, Chen Z, Han Y, Han L, Zou X, Zhou B, Hu R, Hao J, Bai S, Xiao H, Li W, Bueker A, Ma Y, Xie G, Yang J, Chen S, Li H, Cao J, Shen L (2020) Immune suppressive landscape in the human esophageal squamous cell carcinoma microenvironment. Nat Commun 11(1):1–17
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix: Stability Analysis
Appendix: Stability Analysis
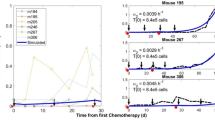
The system of Eq. (1) has four real-valued and biologically sensible equilibrium points, given in Sect. 4. These equilibrium points have been computed using the parameter values by Tang 2020, in Table 1. The Jacobian matrices for each equilibrium point, their eigenvalues and corresponding eigenvectors are given below.
-
1.
Equilibrium point \((S^*, T^*, E^*, C^*, B^*, R^*) = (10^7, 9.9\times 10^8, 0, 0, 845.3, 0).\) As stated in Sect. 4, the system has a line of equilibria on \(S^*+T^* = K\); meanwhile, we have observed in numerical simulations, a particular equilibrium on this line. Here, we present the eigenvalues and eigenvectors of this system at this particular point.
The Jacobian matrix at this equilibrium is:
$$\begin{aligned} \mathfrak {J}_1 =\left[ \begin{array}{cccccc} - 4.9\times 10^{-4}&{}-4.9\times 10^{-4}&{}0&{}-10 &{}0&{}0\\ -4.8\times 10^{-2}&{}-4.8\times 10^{-2}&{}-2.3\times 10^{-1}&{}0&{} 0&{}0\\ 0&{} 0&{} 4.7\times 10^{-1}&{}0&{}0&{}0\\ 0&{}0&{}0&{} 4.8\times 10^{-1}&{}0&{}0 \\ 0&{}{ 2.0\times 10^{-18}}&{}0&{}0&{}-7.0\times 10^{-4}&{}{ 10^{-8}}\\ 0&{}0&{}1.3\times 10^{-1}&{}0&{}0&{}-10^{-5}\end{array} \right] \end{aligned}$$The eigenvalues are: \((0, -4.9\times 10^{-2}, -7.0\times 10^{-4}, 4.8 \times 10^{-1}, -1.0 \times 10^{-5}, 4.7\times 10^{-1})\) and the corresponding eigenvectors are:
$$\begin{aligned} \mathbf {v}_1 = \left[ \begin{array}{c} 7.1 \times 10^{-1}\\ - 7.1 \times 10^{-1}\\ 0\\ 0 \\ 0\\ 0\end{array} \right] , \mathbf {v}_2 = \left[ \begin{array}{c} 1.0 \times 10^{-2}\\ 1.0\\ 0\\ 0 \\ 0\\ 0\end{array} \right] , \mathbf {v}_3 = \left[ \begin{array}{c} 0.0\\ 0.0 \\ 0.0\\ 0.0 \\ - 1.0\\ 0.0\end{array} \right] \\ \mathbf {v}_4 = \left[ \begin{array}{c} - 1.0\\ 9.0\times 10^{-2}\\ 0\\ 4.8\times 10^{-2}\\ 0\\ 0 \end{array} \right] , \mathbf {v}_5 = \left[ \begin{array}{c} 0\\ 0 \\ 0\\ 0 \\ 1.4\times 10^{-5}\\ 1.0 \end{array} \right] , \mathbf {v}_6 = \left[ \begin{array}{c}4.3\times 10^{-4}\\ - 4.1\times 10^{-1}\\ 9.1\times 10^{-1}\\ 0\\ 5.1\times 10^{-10}\\ 2.4\times 10^{-2}\end{array} \right] \end{aligned}$$ -
2.
Equilibrium point \((S^*, T^*, E^*, C^*, B^*, R^*) = (0, 0, 0, 0, 0, 0)\)
The trivial solution,
$$\begin{aligned} (S^*, T^*, E^*, C^*, B^*, R^*) = (0, 0, 0, 0, 0, 0) \end{aligned}$$is an unstable equilibrium. The four positive eigenvalues indicate population growth in the directions of the CAR T-cells, the immune cells, tumor and stem cells, all of which are to be expected since the introduction of a few of these cells would instigate growth for that population. The direction of attraction, for the two negative eigenvalues, is the \(B-\)axis and a line on the \(B-R\) plane, meaning the TGF-\(\beta \) and Tregs tend to stabilize without the presence of effector cells or the tumor. The Jacobian matrix at this equilibrium is:
$$\begin{aligned} \mathfrak {J}_2 = \left[ \begin{array}{cccccc} 4.9\times 10^{-2}&{}0&{}0&{}0&{}0&{}0 \\ 0&{} 4.7\times 10^{-1}&{}0&{}0&{}0&{}0 \\ 0&{}0&{}-7.0\times 10^{-4}&{}1.0\times 10^{-8}&{} 0 &{}0\\ 0&{}1.3\times 10^{-2}&{}0&{}-1.0\times 10^{-5}&{}0&{}0 \\ 3.1\times 10^{-1}&{}0&{}0&{}0&{}4.5\times 10^{-2}&{}0\\ 0&{}0&{}0&{}0&{}0&{}4.8\times 10^{-1}\end{array} \right] \end{aligned}$$The eigenvalues are: \((4.8\times 10^{-1}, 4.7 \times 10^{-1}, 4.9\times 10^{-2}, 4.54\times 10^{-2}, -7.0\times 10^{-4}, -1\times 10^{-5})\) and the corresponding eigenvectors are:
$$\begin{aligned} \mathbf {v}_1 = \left[ \begin{array}{c} 0\\ 0 \\ 0\\ 0 \\ 0\\ 1\end{array} \right] , \mathbf {v}_2 = \left[ \begin{array}{c} 0\\ -1.0 \\ -5.6\times 10^{-10}\\ -2.6\times 10^{-2}\\ 2.0\times 10^{-10} \\ 0\end{array} \right] , \mathbf {v}_3 = \left[ \begin{array}{c} -1.2\times 10^{-2}\\ 0 \\ 0\\ 0 \\ -1.0\\ 0 \end{array} \right] , \\ \mathbf {v}_4 = \left[ \begin{array}{c} 0\\ 0 \\ 0\\ 0 \\ -1.0\\ 0 \end{array} \right] , \mathbf {v}_5 = \left[ \begin{array}{c} 0\\ 0 \\ 1\\ 0 \\ 0\\ 0\end{array} \right] , \mathbf {v}_6 = \left[ \begin{array}{c} 0\\ 0 \\ 1.4\times 10^{-5}\\ 1 \\ 0\\ 0\end{array} \right] \end{aligned}$$ -
3.
Equilibrium point \((S^*, T^*, E^*, C^*, B^*, R^*) = (0, 0, 37.7, 0, 6.7\times 10^{-1}, 47179)\) This is a tumor and stem cell-free equilibrium. The three positive eigenvalues have eigenvectors that determine the direction of population growth for all cells, which is expected. The two complex eigenvalues with negative real parts provide a spiral sink-type behavior for the immune cells and TGF-\(\beta \); without the tumor or the cancer stem cells, the immune system will be in a nonzero equilibrium. The direction for the only attractor is the \(B-\)axis; without the tumor or Tregs, the TGF-\(\beta \) will stabilize at a low level. The Jacobian matrix at this equilibrium is:
$$\begin{aligned} \mathfrak {J}_2 = \left[ \begin{array}{cccccc}4.9\times 10^{-2}&{}0&{}0&{}0&{}0&{}0 \\ 0&{} 0&{}0&{}-3.8\times 10^{-4}&{}4.2\times 10^{-2}&{}0 \\ 0&{}0&{}-7\times 10^{-4}&{}10^{-8}&{} 0 &{}0\\ 0&{}1.3\times 10^{-2}&{}0&{}-10^{-6}&{}0&{}0 \\ 3.1\times 10^{-1}&{}0&{}0&{}0&{}4.5\times 10^{-2}&{}0 \\ 0&{}0&{}0&{}0&{}0&{}1.2\times 10^{-2}\end{array} \right] \end{aligned}$$The eigenvalues are \((4.9\times 10^{-2},4.5\times 10^{-2}, 1.2\times 10^{-2}, -5.0\times 10^{-6}+(2.2\times 10^{-3})i,\)
\(-5.0\times 10^{-6}-(2.2\times 10^{-3})i, -7.0\times 10^{-4}).\) The corresponding eigenvectors are:
$$\begin{aligned} \mathbf {v}_1 = \left[ \begin{array}{c} -8.7\times 10^{-3}\\ -6.4\times 10^{-1}\\ -3.3\times 10^{-8} \\ -1.62\times 10^{-1}\\ -7.5\times 10^{-1} \\ 0\end{array} \right] , \mathbf {v}_2 = \left[ \begin{array}{c} -2.0\times 10^{-9}\\ -6.6\times 10^{-1}\\ -4.0\times 10^{-8} \\ -1.8\times 10^{-1}\\ -7.3\times 10^{-1} \\ 0\end{array} \right] , \mathbf {v}_3 = \left[ \begin{array}{c} 0\\ 0 \\ 0\\ 0 \\ 0\\ 1\end{array} \right] ,\\ \mathbf {v}_4 = \left[ \begin{array}{c}1.8\times 10^{-10}+{ 8.8\times 10^{-11}}\,i\\ 3.9\times 10^{-4}+1.7\times 10^{-1}\,i \\ 1.3\times 10^{-6}-4.1\times 10^{-6}\,i \\ 9.9\times 10^{-1}\\ {4.8 \times 10^{-11}}-4.4\times 10^{-10}\,i\\ 0 \end{array} \right] , \mathbf {v}_5 = \mathbf {v}_4, \mathbf {v}_6 = \left[ \begin{array}{c} -{ 1\times 10^{-13}} \\ -2.0\times 10^{-10}\\ -1 \\ { 7.0\times 10^{-13}} \\ -{9.0\times 10^{-14}} \\ 0\end{array} \right] \end{aligned}$$ -
4.
Equilibrium point \((S^*, T^*, E^*, C^*, B^*, R^*) = (0, 10^9, 37.7, 0, 846, 47180)\) The fourth equilibrium is a maximal tumor case where the tumor is at its carrying capacity, which is a saddle. There are two directions of attraction; one of them is the \(T-\)axis, meaning the tumor population is converging to its carrying capacity, and the other points to a positive tumor, effector cells and TGF-\(\beta \) and an opposite behavior for Tregs. The two complex eigenvalues have negative real parts with corresponding eigenvectors that are a complex scalar multiple of \(\mathbf {e}_5\) parallel to the \(T-\)axis. This freedom is due to the equilibrium not being completely defined and rather the equilibrium of \(T^*\) and \(S^*\) just must satisfy \(T^*+S^*=K\). The Jacobian matrix at this equilibrium is:
$$\begin{aligned} \mathfrak {J}_4 = \left[ \begin{array}{cccccc}9.6\times 10^{-9}&{}0&{}0&{}0&{}0&{}0 \\ 0&{}0&{}-4.7\times 10^{-7}&{}-3.8\times 10^{-4}&{} 2.0\times 10^{-22}&{}0\\ 0&{}0&{}-7\times 10^{-4}&{}10^-8&{}{ 3.0\times 10^{-18}}&{}0\\ 0&{} 1.3\times 10^{-2}&{}0&{}-10^{-5}&{}0&{}0\\ -4.5\times 10^{-2}&{}-2.4\times 10^{-1}&{} 1.1\times 10^{-2}&{}0&{}-4.5\times 10^{-2}&{}0\\ 0&{}0&{}0&{}0&{}0&{}1.2\times 10^{-2}\end{array} \right] \end{aligned}$$The eigenvalues are \((-4.5\times 10^{-2}, 1.2\times 10^{-2}, -5.0\times 10^{-6} + (2.1\times 10^{-3})\,i, -5.0\times 10^{-6} - (2.1\times 10^{-3})\,i, -7.0\times 10^{-4}, 9.6\times 10^{-9} ).\) The corresponding eigenvectors are:
$$\begin{aligned} \mathbf {v}_1 = \left[ \begin{array}{c} 0\\ 0 \\ 0\\ 0 \\ 1\\ 0\end{array} \right] , \mathbf {v}_2 = \left[ \begin{array}{c} 0\\ 0 \\ 0\\ 0 \\ 0\\ 1\end{array} \right] , \mathbf {v}_3 = \left[ \begin{array}{c} 0\\ 2.9\times 10^{-4}- 1.3\times 10^{-1}\,i\\ 9.8\times 10^{-7}+ 3.1\times 10^{-6}\,i\\ 7.4\times 10^{-1} \\ -3.3\times 10^{-2}+6.6\times 10^{-1}\,i \\ 0\end{array} \right] ,\\ \mathbf {v}_4 = \mathbf {v}_3, \mathbf {v}_5 = \left[ \begin{array}{c} 0\\ -6.1\times 10^{-5} \\ -9.7\times 10^{-1}\\ 1.1\times 10^{-3}\\ -2.3\times 10^{-1} \\ 0\end{array} \right] , \mathbf {v}_6 = \left[ \begin{array}{c} -7.1\times 10^{-1}\\ 0 \\ 0\\ 0 \\ 7.\times 10^{-1}\\ 0 \end{array} \right] \end{aligned}$$
Rights and permissions
About this article
Cite this article
Swanson, E.R., Köse, E., Zollinger, E.A. et al. Mathematical Modeling of Tumor and Cancer Stem Cells Treated with CAR-T Therapy and Inhibition of TGF-\(\beta \). Bull Math Biol 84, 58 (2022). https://doi.org/10.1007/s11538-022-01015-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-022-01015-5