Abstract
Introduction
Chronic kidney disease (CKD) is a major public health challenge given its high global prevalence and associated risks of cardiovascular disease and progression to end stage renal disease. Although it is known that numerous metabolic changes occur in CKD patients, identifying novel metabolite associations with kidney function may enhance our understanding of the physiologic pathways relating to CKD.
Objectives
The objective of this study was to elucidate novel metabolite associations with kidney function among participants of two community-based cohorts with carefully ascertained metabolomics, kidney function, and covariate data.
Methods
Untargeted ultrahigh-performance liquid chromatography–tandem mass spectrometry was used to detect and quantify blood metabolites. We used multivariate adjusted linear regression to examine associations between single metabolites and creatinine-based estimated glomerular filtration rate (eGFRcr) among 1243 Bogalusa Heart Study (BHS) participants (median eGFRcr: 94.4, 5th–95th percentile: 66.0–119.6 mL/min/1.73 m2). Replication, determined by statistical significance and consistent effect direction, was tested using gold standard measured glomerular filtration rate (mGFR) among 260 Multi-Ethnic Study of Atherosclerosis (MESA) participants (median mGFR: 72.0, 5th–95th percentile: 43.5–105.0 mL/min/1.73 m2). All analyses used Bonferroni-corrected alpha thresholds.
Results
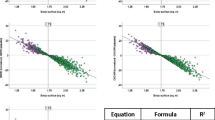
Fifty-one novel metabolite associations with kidney function were identified, including 12 from previously unrelated sub-pathways: N6-carboxymethyllysine, gulonate, quinolinate, gamma-CEHC-glucuronide, retinol, methylmalonate, 3-hydroxy-3-methylglutarate, 3-aminoisobutyrate, N-methylpipecolate, hydroquinone sulfate, and glycine conjugates of C10H12O2 and C10H14O2(1). Significant metabolites were generally inversely associated with kidney function and smaller in mass-to-charge ratio than non-significant metabolites.
Conclusion
The 51 novel metabolites identified may serve as early, clinically relevant, kidney function biomarkers.


Similar content being viewed by others
Data availability
The metabolomic data sets generated and analyzed during this study are available as supplementary data.
Abbreviations
- AGE:
-
Advanced glycation end-product
- BAIBA:
-
Β-aminoisobutyric acid
- BCAA:
-
Branched-chain amino acids
- BHS:
-
Bogalusa Heart Study
- BMI:
-
Body mass index
- CEHC:
-
Carboxyethyl hydroxychroman
- CKD:
-
Chronic kidney disease
- CML:
-
N6-carboxymethyllysine
- CVD:
-
Cardiovascular disease
- eGFR:
-
Estimated glomerular filtration rate
- eGFRcr:
-
Creatinine based eGFR
- GFR:
-
Glomerular filtration rate
- MESA:
-
Multi-Ethnic Study of Atherosclerosis
- mGFR:
-
Measured glomerular filtration rate
- MMA:
-
Methylmalonate
- SD:
-
Standard deviation
- UPLC–MS/MS:
-
Ultrahigh performance liquid chromatography–tandem mass spectrometry
References
Barski, O. A., Papusha, V. Z., Ivanova, M. M., Rudman, D. M., & Finegold, M. J. (2005). Developmental expression and function of aldehyde reductase in proximal tubules of the kidney. American Journal of Physiology. Renal Physiology, 289(1), F200–F207. https://doi.org/10.1152/ajprenal.00411.2004.
Berenson, G. S., Wattigney, W. A., Bao, W., Srinivasan, S. R., & Radhakrishnamurthy, B. (1995). Rationale to study the early natural history of heart disease: The Bogalusa Heart Study. The American journal of the medical sciences, 310(Supplement 1), S22–S28. https://doi.org/10.1097/00000441-199512000-00005.
Bild, D. E., Bluemke, D. A., Burke, G. L., Detrano, R., Diez Roux, A. V., Folsom, A. R., et al. (2002). Multi-ethnic study of atherosclerosis: Objectives and design. American Journal of Epidemiology, 156(9), 871–881. https://doi.org/10.1093/aje/kwf113.
Breier, M., Wahl, S., Prehn, C., Fugmann, M., Ferrari, U., Weise, M., et al. (2014). Targeted metabolomics identifies reliable and stable metabolites in human serum and plasma samples. PLoS ONE, 9(2), 1–11. https://doi.org/10.1371/journal.pone.0089728.
Brenner, B. M., Hostetter, T. H., & Humes, H. D. (1978). Glomerular permselectivity: Barrier function based on discrimination of molecular size and charge. The American Journal of Physiology, 234(6), F455–F460. https://doi.org/10.1152/ajprenal.1978.234.6.F455.
Coresh, J., Inker, L. A., Sang, Y., Chen, J., Shafi, T., Post, W. S., et al. (2018). Metabolomic profiling to improve glomerular filtration rate estimation: A proof-of-concept study. Nephrology, Dialysis, Transplantation, 34(5), 1–9. https://doi.org/10.1093/ndt/gfy094.
Evans, A. M., DeHaven, C. D., Barrett, T., Mitchell, M., & Milgram, E. (2009). Integrated, nontargeted ultrahigh performance liquid chromatography/electrospray ionization tandem mass spectrometry platform for the identification and relative quantification of the small-molecule complement of biological systems. Analytical Chemistry, 81(16), 6656–6667. https://doi.org/10.1021/ac901536h.
Foster, T. A., & Berenson, G. S. (1987). Measurement error and reliability in four pediatric cross-sectional surveys of cardiovascular disease risk factor variables—The Bogalusa Heart Study. Journal of Chronic Diseases, 40(1), 13–21. https://doi.org/10.1016/0021-9681(87)90092-0.
Gejyo, F., Kinoshita, Y., & Ikenaka, T. (1976). Identification of beta-aminoisobutyric acid in uremic serum. Clinica chimica Acta; International Journal of Clinical Chemistry, 70(3), 407–415. https://doi.org/10.1016/0009-8981(76)90354-5.
Goek, O.-N., Prehn, C., Sekula, P., Römisch-Margl, W., Döring, A., Gieger, C., et al. (2013). Metabolites associate with kidney function decline and incident chronic kidney disease in the general population. Nephrology, Dialysis, Transplantation, 28(8), 2131–2138. https://doi.org/10.1093/ndt/gft217.
Hamamura, K., Matsunaga, N., Ikeda, E., Kondo, H., Ikeyama, H., Tokushige, K., et al. (2016). Alterations of hepatic metabolism in chronic kidney disease via D-box-binding protein aggravate the renal dysfunction. The Journal of biological chemistry, 291(10), 4913–4927. https://doi.org/10.1074/jbc.M115.696930.
Handelman, G. J., & Levin, N. W. (2011). Guidelines for vitamin supplements in chronic kidney disease patients: What is the evidence? Journal of Renal Nutrition, 21(1), 117–119. https://doi.org/10.1053/j.jrn.2010.11.004.
Haraldsson, B., Nystrom, J., & Deen, W. M. (2008). Properties of the glomerular barrier and mechanisms of proteinuria. Physiological Reviews, 88(2), 451–487. https://doi.org/10.1152/physrev.00055.2006.
Hard, G. C., Whysner, J., English, J. C., Zang, E., & Williams, G. M. (1997). Relationship of hydroquinone-associated rat renal tumors with spontaneous chronic progressive nephropathy. Toxicologic Pathology, 25(2), 132–143. https://doi.org/10.1177/019262339702500202.
Inker, L. A., Shafi, T., Okparavero, A., Tighiouart, H., Eckfeldt, J. H., Katz, R., et al. (2016). Effects of race and sex on measured GFR: The multi-ethnic study of atherosclerosis. American Journal of Kidney Diseases, 68(5), 743–751. https://doi.org/10.1053/j.ajkd.2016.06.021.
Jha, V., Garcia-Garcia, G., Iseki, K., Li, Z., Naicker, S., Plattner, B., et al. (2013). Chronic kidney disease: Global dimension and perspectives. The Lancet, 382(9888), 260–272. https://doi.org/10.1016/S0140-6736(13)60687-X.
Kaluarachchi, M., Boulangé, C. L., Karaman, I., Lindon, J. C., Ebbels, T. M. D., Elliott, P., et al. (2018). A comparison of human serum and plasma metabolites using untargeted 1H NMR spectroscopy and UPLC-MS. Metabolomics, 14(3), 1–12. https://doi.org/10.1007/s11306-018-1332-1.
Katakami, N., Shimomura, I., Yamamoto, Y., Ninomiya, H., Omori, K., Matsuoka, T., et al. (2019). Identification of metabolites associated with onset of CAD in diabetic patients using CE-MS analysis: A pilot study. Journal of Atherosclerosis and Thrombosis, 26(3), 233–245. https://doi.org/10.5551/jat.42945.
Levey, A. S., & Coresh, J. (2012). Chronic kidney disease. The Lancet, 379(9811), 165–180. https://doi.org/10.1016/S0140-6736(11)60178-5.
Levey, A. S., Inker, L. A., & Coresh, J. (2014). GFR estimation: From physiology to public health. American Journal of Kidney Diseases, 63(5), 820–834. https://doi.org/10.1053/j.ajkd.2013.12.006.
Levey, A. S., Stevens, L. A., Schmid, C. H., Zhang, Y. L., Iii, A. F. C., Feldman, H. I., et al. (2009). A new equation to estimate glomerular filtration rate. Annals of Internal Medicine, 150(9), 604–612. https://doi.org/10.7326/0003-4819-150-9-200905050-00006.
Lisowska-Myjak, B. (2014). Uremic toxins and their effects on multiple organ systems. Nephron Clinical Practice, 128(3–4), 303–311. https://doi.org/10.1159/000369817.
Mallipattu, S. K., & Uribarri, J. (2014). Advanced glycation end product accumulation: A new enemy to target in chronic kidney disease? Current Opinion in Nephrology and Hypertension, 23(6), 547–554. https://doi.org/10.1097/MNH.0000000000000062.
Mills, K. T., Xu, Y., Zhang, W., Bundy, J. D., Chen, C.-S., Kelly, T. N., et al. (2015). A systematic analysis of worldwide population-based data on the global burden of chronic kidney disease in 2010. Kidney International, 88(5), 950–957. https://doi.org/10.1038/ki.2015.230.
Nicholson, J. K., & Lindon, J. C. (2008). Systems biology: Metabonomics. Nature, 455(7216), 1054–1056. https://doi.org/10.1038/4551054a.
Niewczas, M. A., Mathew, A. V., Croall, S., Byun, J., Major, M., Sabisetti, V. S., et al. (2017). Circulating modified metabolites and a risk of ESRD in patients with type 1 diabetes and chronic kidney disease. Diabetes Care, 40(3), 383–390. https://doi.org/10.2337/dc16-0173.
Niewczas, M. A., Sirich, T. L., Mathew, A. V., Skupien, J., Mohney, R. P., Warram, J. H., et al. (2014). Uremic solutes and risk of end-stage renal disease in type 2 diabetes: Metabolomic study. Kidney International, 85(5), 1214–1224. https://doi.org/10.1038/ki.2013.497.
Niwa, T., Yoshizumi, H., Emoto, Y., Miyazaki, T., Hashimoto, N., Takeda, N., et al. (1991). Accumulation of quinolinic acid in uremic serum and its removal by hemodialysis. Clinical Chemistry, 37(2), 159–161.
Pawlak, K., Brzosko, S., Mysliwiec, M., & Pawlak, D. (2009). Kynurenine, quinolinic acid—The new factors linked to carotid atherosclerosis in patients with end-stage renal disease. Atherosclerosis, 204(2), 561–566. https://doi.org/10.1016/j.atherosclerosis.2008.10.002.
Rhee, E. P., Clish, C. B., Ghorbani, A., Larson, M. G., Elmariah, S., McCabe, E., et al. (2013). A combined epidemiologic and metabolomic approach improves CKD prediction. Journal of the American Society of Nephrology, 24(8), 1330–1338. https://doi.org/10.1681/ASN.2012101006.
Rhee, E. P., Clish, C. B., Wenger, J., Roy, J., Elmariah, S., Pierce, K. A., et al. (2016). Metabolomics of chronic kidney disease progression: A case-control analysis in the chronic renal insufficiency cohort study. American Journal of Nephrology, 43(5), 366–374. https://doi.org/10.1159/000446484.
Rhee, E. P., Souza, A., Farrell, L., Pollak, M. R., Lewis, G. D., Steele, D. J. R., et al. (2010). Metabolite profiling identifies markers of uremia. Journal of the American Society of Nephrology: JASN, 21(6), 1041–1051. https://doi.org/10.1681/ASN.2009111132.
Rietman, A., Stanley, T. L., Clish, C., Mootha, V., Mensink, M., Grinspoon, S. K., et al. (2016). Associations between plasma branched-chain amino acids, β-aminoisobutyric acid and body composition. Journal of Nutritional Science, 5, e6. https://doi.org/10.1017/jns.2015.37.
Roberts, L. D., Boström, P., O’Sullivan, J. F., Schinzel, R. T., Lewis, G. D., Dejam, A., et al. (2014). β-Aminoisobutyric acid induces browning of white fat and hepatic β-oxidation and is inversely correlated with cardiometabolic risk factors. Cell Metabolism, 19(1), 96–108. https://doi.org/10.1016/j.cmet.2013.12.003.
Sarnak, M. J., Levey, A. S., Schoolwerth, A. C., Coresh, J., Culleton, B., Hamm, L. L., et al. (2003). Kidney disease as a risk factor for development of cardiovascular disease: A statement from the American Heart Association Councils on kidney in cardiovascular disease, high blood pressure research, clinical cardiology, and epidemiology and prevention. Circulation, 108(17), 2154–2169. https://doi.org/10.1161/01.CIR.0000095676.90936.80.
Saulnier, P.-J., Wheelock, K. M., Howell, S., Weil, E. J., Tanamas, S. K., Knowler, W. C., et al. (2016). Advanced glycation end products predict loss of renal function and correlate with lesions of diabetic kidney disease in American Indians with type 2 diabetes. Diabetes, 65(12), 3744–3753. https://doi.org/10.2337/db16-0310.
Schefold, J. C., Zeden, J.-P., Fotopoulou, C., von Haehling, S., Pschowski, R., Hasper, D., et al. (2009). Increased indoleamine 2,3-dioxygenase (IDO) activity and elevated serum levels of tryptophan catabolites in patients with chronic kidney disease: A possible link between chronic inflammation and uraemic symptoms. Nephrology, Dialysis, Transplantation: Official Publication of the European Dialysis and Transplant Association—European Renal Association, 24(6), 1901–1908. https://doi.org/10.1093/ndt/gfn739.
Schuck, P. F., Alves, L., Pettenuzzo, L. F., Felisberto, F., Rodrigues, L. B., Freitas, B. W., et al. (2013). Acute renal failure potentiates methylmalonate-induced oxidative stress in brain and kidney of rats. Free Radical Research, 47(3), 233–240. https://doi.org/10.3109/10715762.2012.762771.
Scoppola, A., De Paolis, P., Menzinger, G., Lala, A., & Di Giulio, S. (1997). Plasma mevalonate concentrations in uremic patients. Kidney International, 51(3), 908–912. https://doi.org/10.1038/ki.1997.128.
Sekula, P., Goek, O.-N., Quaye, L., Barrios, C., Levey, A. S., Romisch-Margl, W., et al. (2016). A Metabolome-wide association study of kidney function and disease in the general population. Journal of the American Society of Nephrology, 27(4), 1175–1188. https://doi.org/10.1681/ASN.2014111099.
Semba, R. D., Fink, J. C., Sun, K., Bandinelli, S., Guralnik, J. M., & Ferrucci, L. (2009). Carboxymethyl-lysine, an advanced glycation end product, and decline of renal function in older community-dwelling adults. European Journal of Nutrition, 48(1), 38–44. https://doi.org/10.1007/s00394-008-0757-0.
Semba, R. D., Fink, J. C., Sun, K., Windham, B. G., & Ferrucci, L. (2010a). Serum carboxymethyl-lysine, a dominant advanced glycation end product, is associated with chronic kidney disease: The Baltimore longitudinal study of aging. Journal of renal nutrition, 20(2), 74–81. https://doi.org/10.1053/j.jrn.2009.08.001.
Semba, R. D., Nicklett, E. J., & Ferrucci, L. (2010b). Does accumulation of advanced glycation end products contribute to the aging phenotype? The Journals of Gerontology Series A, Biological sciences and medical sciences, 65(9), 963–975. https://doi.org/10.1093/gerona/glq074.
Shah, V. O., Townsend, R. R., Feldman, H. I., Pappan, K. L., Kensicki, E., & Vander Jagt, D. L. (2013). Plasma metabolomic profiles in different stages of CKD. Clinical Journal of the American Society of Nephrology, 8(3), 363–370. https://doi.org/10.2215/CJN.05540512.
Solini, A., Manca, M. L., Penno, G., Pugliese, G., Cobb, J. E., & Ferrannini, E. (2016). Prediction of declining renal function and albuminuria in patients with type 2 diabetes by metabolomics. Journal of Clinical Endocrinology and Metabolism, 101(2), 696–704. https://doi.org/10.1210/jc.2015-3345.
Sumner, L. W., Amberg, A., Barrett, D., Beale, M. H., Beger, R., Daykin, C. A., et al. (2007). Proposed minimum reporting standards for chemical analysis: Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics, 3(3), 211–221. https://doi.org/10.1007/s11306-007-0082-2.
Tanaka, H., Sirich, T. L., Plummer, N. S., Weaver, D. S., & Meyer, T. W. (2015). An enlarged profile of uremic solutes. PLoS ONE, 10(8), e0135657. https://doi.org/10.1371/journal.pone.0135657.
Toyohara, T., Akiyama, Y., Suzuki, T., Takeuchi, Y., Mishima, E., Tanemoto, M., et al. (2010). Metabolomic profiling of uremic solutes in CKD patients. Hypertension Research, 33(9), 944–952. https://doi.org/10.1038/hr.2010.113.
Wiley, M. H., Howton, M. M., & Siperstein, M. D. (1977). The quantitative role of the kidneys in the in vivo metabolism of mevalonate. The Journal of Biological Chemistry, 252(2), 548–554.
Yu, B., Zheng, Y., Nettleton, J. A., Alexander, D., Coresh, J., & Boerwinkle, E. (2014). Serum metabolomic profiling and incident CKD among African Americans. Clinical Journal of the American Society of Nephrology, 9(8), 1410–1417. https://doi.org/10.2215/CJN.11971113.
Zheng, Y., Yu, B., Alexander, D., Mosley, T. H., Heiss, G., Nettleton, J. A., et al. (2013). Metabolomics and incident hypertension among blacks: The atherosclerosis risk in communities study. Hypertension, 62(2), 398–403. https://doi.org/10.1161/HYPERTENSIONAHA.113.01166.
Zsengellér, Z. K., Aljinovic, N., Teot, L. A., Korson, M., Rodig, N., Sloan, J. L., et al. (2014). Methylmalonic acidemia: a megamitochondrial disorder affecting the kidney. Pediatric Nephrology (Berlin, Germany), 29(11), 2139–2146. https://doi.org/10.1007/s00467-014-2847-y.
Acknowledgements
This study was funded by the National Institute on Aging of the NIH under award numbers R01AG041200 and R21AG051914. Research reported in this publication was partially supported by the National Institute of General Medical Sciences of the NIH under award number P20GM109036. We are grateful for the contribution of all staff members who were involved in conducting BHS and MESA. We extend our gratitude to the participants of BHS (many of whom have diligently participated since they were children) and MESA.
Funding
This study was funded by the National Institute on Aging of the NIH under Award Numbers R01AG041200 and R21AG051914. Research reported in this publication was partially supported by the National Institute of General Medical Sciences of the NIH under Award Number P20GM109036.
Author information
Authors and Affiliations
Contributions
JLN, TNK, JH, SL, LB, HH, AHA, and WC designed the study; JLN, CL, XG, MS, ACR, and XM analyzed the data; JMK contributed to the logistics and optimization of the untargeted metabolomics; CMR, JC, ASL, LAI, and MS performed replication of the study results; JLN made the figures; JLN and TNK drafted and revised the manuscript; all authors read and approved the final article.
Corresponding author
Ethics declarations
Conflict of interest
JLN, TNK, JH, SL, LB, HH, AHA, WC, CL, XG, MS, ACR, XM, CMR, JC, ASL, LAI, and MS declare that they have no conflict of interest. JMK is employed by Metabolon, Inc. He contributed to the logistics, optimization, and interpretation of the untargeted metabolomics. Metabolon, Inc. was not involved in the study design, statistical analysis, or interpretation of the results.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional review boards at participating institutions and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nierenberg, J.L., He, J., Li, C. et al. Novel associations between blood metabolites and kidney function among Bogalusa Heart Study and Multi-Ethnic Study of Atherosclerosis participants. Metabolomics 15, 149 (2019). https://doi.org/10.1007/s11306-019-1613-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-019-1613-3