Abstract
Langerhans cell histiocytosis (LCH) is a disease with varied clinic manifestations. The oral symptoms and signs of LCH localized to the jaws are nonspecific, which may lead to misdiagnosis of this disease. The purpose of this paper was to present the case of a 2-year, 4-month-old LCH patient with progressive destruction of jaws caused by the delayed treatment due to the global outbreak of COVID-19. The cone beam CT analysis after an interval of 6 months reminded us the great significance of early diagnosis and treatment of LCH.
Similar content being viewed by others
Introduction
Langerhans cell histiocytosis (LCH) is a rare disease whose clinical manifestations varies extremely since the affected organs differ a lot in patients [1]. The bone is commonly encroached in patients with LCH. In pediatric patients, the bone lesions mainly happen to the skull, the spine, the extremities, pelvic bone and ribs. LCH localized to the jaws is relatively rare and the oral symptoms and signs are nonspecific [2, 3].
This paper presents a case of 2-year-old LCH patient with progressive destruction of jaws caused by the delayed treatment due to the global outbreak of COVID-19. The cone beam CT (CBCT) performed to define the margins and characteristics of the lesion before surgery illustrated the progressive lytic destruction of the jaws. Early diagnosis and treatment of LCH is of great significance for reducing the rapid resorption of the jaws.
Case report
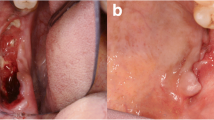
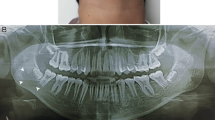
A 2-year-and-4-month-old boy was referred to our hospital on December 16, 2019, whose chief complaint was the recurrent swelling of the left face. Asymmetric face was observed clinically, assembling the swelling caused by the maxillofacial space inflammation of acute attack of chronic periapical periodontitis. Intraoral examination (Fig. 1) showed the dental caries, gingival necrosis, irregular ulcer and hypermobility of the teeth (55, 65, 74, 75, and 85). Panoramic radiograph (Fig. 2a) revealed radiolucency zone around the periapical area of 74 and 84. Increased radiotracer uptake within the mandible was elucidated by bone scintigraphy (Fig. 2b), and the remainder of the skeleton was within normal limit. None abnormal observation was found by the complete physical examination and other laboratory evaluation, such as full blood count, blood chemistry, electrolytes, total protein, albumin, ferritin, and urinalysis. Systemic disease was suspected and surgery was planned to get the biopsy specimen to make the correct diagnosis. CBCT before surgery was performed on January 7, 2020. However, the surgery was delayed due to the global outbreak of COVID-19. The patient returned to the hospital in July, 2020, when the situation of COVID-19 was not so serious. CBCT was taken again to define the margins and characteristics of the lesion (Figs. 3 and 4). Sagittal images around the teeth on January 7, 2020 showed that bone destruction of the jaws was restrained to 85, 84, 74 and 75. Periosteal reaction was detected at the mandibular bone in correspondence to 75. CBCT scan performed on July, 2020 revealed an enlargement of the lesion. Bone around the deciduous teeth were almost affected except the tooth 83. Focal interruption of lingual and buccal cortical bone was obvious. Teeth 85, 84, 81, 71, 72, 73, 74 and 75 seemed to be floating in the mandible (Fig. 3). Sagittal imaging of CBCT scan in Fig. 4 showed abnormal enlargement of the left mandibular ramus. An osteolytic lesion on January 7, 2020 occupied half of the left mandibular ramus, while the bone destruction in July, 2020 almost involved the whole left mandibular ramus, reaching the sigmoid notch area. The coronal section of CBCT scan illustrated the left part of palatal plate was encroached on January 7, 2020. The unaffected right palatal plate during the first examination was also destroyed when the second CBCT examination was taken. It also confirmed the progressive destructive radiolucent lesion of the left mandibular ramus. Axial section revealed the broadening of the bone destruction around the maxillary sinus and mandible (Fig. 4). Biopsy specimen was obtained under general anesthesia. Standard hematoxylin and eosin (H&E) and immunohistochemical (IHC) staining was further adopted to facilitate the diagnosis. Postoperative H&E staining (Fig. 5) showed infiltration of typical Langerhans cells, accompanied by eosinophils, lymphocytes, neutrophils, multinucleated giant cells and some other atypical cells. Positive staining of Langerin, CD-1a and S-100 was revealed by IHC staining, confirming the diagnosis of LCH. Genotyping study demonstrated the oncogenic BRAF V600E mutation. The patient was transferred to the pediatric department for further assessment. A combination of prednisone and vinblastine was used as systemic treatment. The genotyping test of BRAF V600E turned into negative result and CBCT analysis showed lytic destruction of the jaws reduced at the follow-up visit after 27 weeks’ treatment, indicating a good prognosis of this patient.
Discussion
The incidence of LCH is about 4–5 cases per million per year for children under 15 years old [1]. The clinical symptoms of LCH vary a lot since the affected organs differ in patients. The lesions often occur in the bone and the skin in pediatric patients. Skull is the most often involved bone lesion. Only 7–10% of cases exhibit oral abnormality [4]. The oral manifestations of LCH include palatal mucosa with reddish or strawberry appearance, irregular ulcerated lesions on oral mucosa with periodontal involvement, and floating teeth observed by panoramic radiograph [2, 5, 6]. The rarity of LCH and the nonspecific oral lesions make the diagnosis of LCH patients only with oral involvement difficulty. The CBCT results of this patient elucidated that the bone damage progresses rapidly after 6-month interval, implying the great significance of early diagnosis and timely treatment.
The diagnosis of LCH depends on the clinical presentations, radiographic evaluation and histopathological features. The finding of Birbeck granules in LCH cells by electron microscopy was required to get the definitive diagnosis [7]. However, the detection of Birbeck granules by electron microscopy needs special instruments. The positive rate is low and the cost is high. Its diagnostic value has been replaced by a cell-surface receptor–langerin, which induces the formation of Birbeck granule [1].
The etiology and pathogenesis of LCH remain controversial, with two main arguments, one being the reactive disorders caused by the aberrant immune system and the other being the neoplastic dysfunction [8]. An overall BRAF mutation frequency of 48.5% indicated that LCH is a neoplasm in nature [9]. Patients with BRAF V600E mutation may have a higher recurrence rate.
Currently, LCH is classified into two categories according to the organs and systems affected, namely single-system LCH and multisystem LCH. Single-system LCH is further divided into unifocal lesion and multifocal lesions. Multisystem LCH is defined as risk organ positive multisystem LCH when the liver, lung, spleen or bone marrow is involved. Otherwise, it is diagnosed as risk organ negative multisystem LCH [10].
Appropriate treatment for LCH patients depends on the site and the dispersion status of affected organs, the phase of lesions and the healing procedure. Surgical procedures can be used for biopsy and curettage for the accessible maxillofacial LCH lesions. It is not suggested to remove all the lesions and the involved teeth with enough bone support may be maintained without influencing the prognosis of LCH [11]. Low-dose radiation therapy can be adopted in maxillofacial LCH cases in the management of unavailable, multifocal or recurrent lesions [12, 13]. The side effects of radiation on the growth of teeth and bone should be concerned in the following oral health management. For maxillofacial LCH patients, chemotherapy is also recommended protocol [8]. A combination of prednisone and vinblastine, employed in this case, is considered as the standard initial therapy for patients requiring systemic treatment. Treatment duration of 12 months is better than 6 months in decreasing the reactivation of the disease [14]. LCH patients with BRAF mutation may showed resistance and poor short-term response to chemotherapy [15]. Targeted therapy of BRAF inhibitor, such as Vemurafenib, is considered as an additional tool [10].
Oral health management for children during the entire growth and development is advocated [16]. Oral hygiene instructions, prevention and treatment of dental caries, regular fluoride application and follow-up should be commonly implemented for LCH children in pediatric clinic. The promotion of soft tissue health, such as topical application of chlorhexidine, should also be taken into consideration in the case of mucosal ulcer, gingival necrosis and periodontal inflammation. Space management and occlusal recovery is necessary for LCH patients with tooth loss. Monitoring the growth of teeth and bone should be highlighted during the follow-up sessions for children with LCH.
In summary, the case report presents the case of a 2-year, 4-month-old LCH patient with progressive destruction of jaws caused by the delayed treatment due to the global outbreak of COVID-19. The CBCT analysis after an interval of 6 months reminded us the great significance of early diagnosis and treatment of LCH. The diagnosis of LCH requires histological and immunophenotypic examination of lesional tissue. Bridges should be built with pediatric specialists to provide an appropriate and sufficient treatment. Comprehensive oral health management will facilitate the growth and development of the teeth, as well as the occlusion and the jaws for children with LCH.
References
Morimoto A, Oh Y, Shioda Y, Kudo K, Imamura T. Recent advances in Langerhans cell histiocytosis. Pediatr Int. 2014;56:451–61.
Hammouri EH, Sweidan HA, Ashokaibi O, Al Omari L. Langerhans cell histiocytosis: a case report with oral manifestations and the role of pediatric dentists in the diagnosis. Clin Case Rep. 2020;8:545–9.
Wu JS, Zhang WL, Li ZF, Wang HF, Yang X, Zhang M, et al. Mandibular Langerhans cell histiocytosis: a case report. Hua Xi Kou Qiang Yi Xue Za Zhi. 2020;38:224–7.
Capodiferro S, Tempesta A, Limongelli L, Ingravallo G, Maiorano E, Sfasciotti GL, et al. Primary oro-facial manifestations of langerhans cell histiocytosis in pediatric age: a bi-institutional retrospective study on 45 cases. Children. 2020;7:104.
Neves-Silva R, Fernandes DT, Fonseca FP, Rebelo P, Helder A, Brasileiro BF, et al. Oral manifestations of Langerhans cell histiocytosis: a case series. Spec Care Dentist. 2018;38:426–33.
Chugh A, Kaur A, Kumar Patnana A, Kumar P, Chugh VK. Unisystem Langerhans cell histiocytosis in maxillofacial region in pediatrics: comprehensive and systematic review. Oral Maxillofac Surg. 2021;25:429–44.
Chou T, D’Angio GJ, Favara BE, Brabencova E, Tazi A, et al. Histiocytosis syndromes in children. Writing group of the histiocyte society. Lancet. 1987;1:208–9.
Kumar YP, Agrawal J, Mohanlakshmi J, Kumar PS. Langerhans cell histiocytosis revisited: case report with review. Contemp Clin Dent. 2015;6:432–6.
Anna-Maria B, Stephanie EW, Albrecht S, Annette A, Konrad S, Silke B, et al. Potential clinical implications of BRAF mutations in histiocytic proliferations. Oncotarget. 2014;5:4060–70.
Leung AKC, Lam JM, Leong KF. Childhood Langerhans cell histiocytosis: a disease with many faces. World J Pediatr. 2019;15:536–45.
Saeed N, Javad Y, Shahriar S, Mahsa M, Emran H. Outcome of surgery as sole treatment of eosinophilic granuloma of jaws. J Dent (Shiraz). 2019;20:210–4.
Whitcher BL, Webb DJ. Treatment of recurrent eosinophilic granuloma of the mandible following radiation therapy. J Oral Maxillofac Surg. 1986;44:565–70.
Wong GB, Pharoah MJ, Weinberg S, Brown DH. Eosinophilic granuloma of the mandibular condyle: report of three cases and review of the literature. J Oral Maxillofac Surg. 1997;55:870–8.
Allen CE, Merad M, McClain KL. Langerhans-Cell Histiocytosis. N Engl J Med. 2018;379:856–68.
Heritier S, Emile JF, Barkaoui MA, Thomas C, Fraitag S, Boudjemaa S, et al. BRAF mutation correlates with high-risk langerhans cell histiocytosis and increased resistance to first-line therapy. J Clin Oncol. 2016;34:3023–30.
Zou J. Oral health management for children. Hua Xi Kou Qiang Yi Xue Za Zhi. 2018;36:465–8.
Funding
This study was funded by Research and Develop Program, West China Hospital of Stomatology Sichuan University (RD-02–202110) to MM Meng and NSFC grants (82170947) to J Zou.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5). The study was approved by the West China Hospital of Stomatology Institutional Review Board, Sichuan University.
Informed consent
Informed consent was obtained from all patients for being included in the study. And identifying information of the patient is not included in the article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Meng, M., Zhang, Q., Zhou, X. et al. Progressive destruction of jaws caused by the delayed treatment of Langerhans cell histiocytosis in a 2-year-old boy: A case report. Oral Radiol 38, 433–437 (2022). https://doi.org/10.1007/s11282-022-00606-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11282-022-00606-1