Abstract
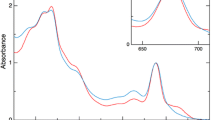
Heliobacteria contain a very simple photosynthetic apparatus, consisting of a homodimeric type I reaction center (RC) without a peripheral antenna system and using the unique pigment bacteriochlorophyll (BChl) g. They are thought to use a light-driven cyclic electron transport pathway to pump protons, and thereby phosphorylate ADP, although some of the details of this cycle are yet to be worked out. We previously reported that the fluorescence emission from the heliobacterial RC in vivo was increased by exposure to actinic light, although this variable fluorescence phenomenon exhibited very different characteristics to that in oxygenic phototrophs (Collins et al. 2010). Here, we describe the underlying mechanism behind the variable fluorescence in heliobacterial cells. We find that the ability to stably photobleach P800, the primary donor of the RC, using brief flashes is inversely correlated to the variable fluorescence. Using pump-probe spectroscopy in the nanosecond timescale, we found that illumination of cells with bright light for a few seconds put them in a state in which a significant fraction of the RCs underwent charge recombination from P800 +A0 − with a time constant of ~20 ns. The fraction of RCs in the rapidly back-reacting state correlated very well with the variable fluorescence, indicating that nearly all of the increase in fluorescence could be explained by charge recombination of P800 +A0 −, some of which regenerated the singlet excited state. This hypothesis was tested directly by time-resolved fluorescence studies in the ps and ns timescales. The major decay component in whole cells had a 20-ps decay time, representing trapping by the RC. Treatment of cells with dithionite resulted in the appearance of a ~18-ns decay component, which accounted for ~0.6 % of the decay, but was almost undetectable in the untreated cells. We conclude that strong illumination of heliobacterial cells can result in saturation of the electron acceptor pool, leading to reduction of the acceptor side of the RC and the creation of a back-reacting RC state that gives rise to delayed fluorescence.






Similar content being viewed by others
References
Albert I, Rutherford AW, Grav H, Kellermann J, Michel H (1998) The 18-kDa cytochrome c 553 from Heliobacterium gestii: gene sequence and characterization of the mature protein. Biochemistry 37(25):9001–9008
Amesz J (1995) The antenna-reaction center complex of heliobacteria. Advances in Photosynthesis 2 (Anoxygenic Photosynthetic Bacteria):687–697
Béal D, Rappaport F, Joliot P (1999) A new high-sensitivity 10-ns time-resolution spectrophotometric technique adapted to in vivo analysis of the photosynthetic apparatus. Rev Sci Instrum 70:202–207
Bowes JM, Crofts AR (1980) Binary oscillations in the rate of reoxidation of the primary acceptor of Photosystem II. Biochim Biophys Acta 590(3):373–384
Brettel K, Leibl W, Liebl U (1998) Electron transfer in the heliobacterial reaction center: evidence against a quinone-type electron acceptor functioning analogous to A1 in Photosystem I. Biochim Biophys Acta 1363(3):175–181
Broess K, Trinkunas G, van der Weij-de Wit CD, Dekker JP, van Hoek A, van Amerongen H (2006) Excitation energy transfer and charge separation in Photosystem II membranes revisited. Biophys J 91(10):3776–3786
Buttler W (1978) Energy distribution in the photochemical apparatus of photosynthesis. Ann Rev Plant Physiol 29:345–378
Byrdin M, Rimke I, Schlodder E, Stehlik D, Roelofs TA (2000) Decay kinetics and quantum yields of fluorescence in Photosystem I from Synechococcus elongatus with P700 in the reduced and oxidized state: Are the kinetics of excited state decay trap-limited or transfer-limited? Biophys J 79(2):992–1007
Caffarri S, Broess K, Croce R, van Amerongen H (2011) Excitation energy transfer and trapping in higher plant Photosystem II complexes with different antenna sizes. Biophys J 100(9):2094–2103
Chauvet A, Sarrou J, Lin S, Romberger SP, Golbeck JH, Savikhin S, Redding KE (2013) Temporal and spectral characterization of the photosynthetic reaction center from Heliobacterium modesticaldum. Photosynth Res 116(1):1–9
Clayton RK (1965) Characteristics of fluorescence and delayed light emission from green photosynthetic bacteria and algae. J Gen Physiol 48:633–646
Clayton RK (1967) An analysis of the relations between fluorescence and photochemistry during photosynthesis. J Theor Biol 14(2):173–186
Collins AM, Redding KE, Blankenship RE (2010) Modulation of fluorescence in Heliobacterium modesticaldum cells. Photosynth Res 104(2–3):283–292
Ducluzeau AL, Chenu E, Capowiez L, Baymann F (2008) The Rieske/cytochrome b complex of Heliobacteria. Biochim Biophys Acta 1777(9):1140–1146
Duysens LN (1978) Transfer and trapping of excitation energy in Photosystem II. Ciba Found Symp 61:323–340
Engelmann EC, Zucchelli G, Garlaschi FM, Casazza AP, Jennings RC (2005) The effect of outer antenna complexes on the photochemical trapping rate in barley thylakoid Photosystem II. Biochim Biophys Acta 1706(3):276–286
Engelmann E, Zucchelli G, Casazza AP, Brogioli D, Garlaschi FM, Jennings RC (2006) Influence of the Photosystem I-light harvesting complex I antenna domains on fluorescence decay. Biochemistry 45(22):6947–6955
Galka P, Santabarbara S, Khuong TT, Degand H, Morsomme P, Jennings RC, Boekema EJ, Caffarri S (2012) Functional analyses of the plant Photosystem I-light-harvesting complex II supercomplex reveal that light-harvesting complex II loosely bound to Photosystem II is a very efficient antenna for Photosystem I in state II. Plant Cell 24(7):2963–2978
Giera W, Ramesh VM, Webber AN, van Stokkum I, van Grondelle R, Gibasiewicz K (2010) Effect of the P700 pre-oxidation and point mutations near A0 on the reversibility of the primary charge separation in Photosystem I from Chlamydomonas reinhardtii. Biochim Biophys Acta 1797(1):106–112
Gobets B, van Grondelle R (2001) Energy transfer and trapping in Photosystem I. Biochim Biophys Acta 1507(1–3):80–99
Guergova-Kuras M, Boudreaux B, Joliot A, Joliot P, Redding K (2001) Evidence for two active branches for electron transfer in Photosystem I. Proc Natl Acad Sci USA 98(8):4437–4442
Hastings G, Kleinherenbrink FA, Lin S, Blankenship RE (1994) Time-resolved fluorescence and absorption spectroscopy of Photosystem I. Biochemistry 33(11):3185–3192
Heinnickel M, Shen G, Agalarov R, Golbeck JH (2005) Resolution and reconstitution of a bound Fe–S protein from the photosynthetic reaction center of Heliobacterium modesticaldum. Biochemistry 44(29):9950–9960
Heinnickel M, Agalarov R, Svensen N, Krebs C, Golbeck JH (2006) Identification of FX in the heliobacterial reaction center as a [4Fe–4S] cluster with an S = 3/2 ground spin state. Biochemistry 45(21):6756–6764
Heinnickel M, Shen G, Golbeck JH (2007) Identification and characterization of PshB, the dicluster ferredoxin that harbors the terminal electron acceptors F(A) and F(B) in Heliobacterium modesticaldum. Biochemistry 46(9):2530–2536
Hiraishi A (1989) Occurrence of menaquinone is the sole isoprenoid quinone in the photosynthetic bacterium Heliobacterium chlorum. Arch Microbiol 151:378–379
Hodges M, Moya I (1986) Time-resolved chlorophyll fluorescence studies of photosynthetic membranes: resolution and characterisation of four kinetic components. Biochim Biophys Acta 849:193–202
Hohmann-Marriott MF, Blankenship RE (2007) Variable fluorescence in green sulfur bacteria. Biochim Biophys Acta 1767(1):106–113
Holzwarth AR, Wendler J, Haehnel W (1985) Time-resolved picosecond fluorescence spectra of the antenna chlorophylls in Chlorella vulgaris. Resolution of Photosystem I fluorescence. Biochim Biophys Acta 807:155–167
Holzwarth AR, Muller MG, Niklas J, Lubitz W (2006) Ultrafast transient absorption studies on Photosystem I reaction centers from Chlamydomonas reinhardtii. 2 Mutations near the P700 reaction center chlorophylls provide new insight into the nature of the primary electron donor. Biophys J 90(2):552–565
Ihalainen JA, van Stokkum IH, Gibasiewicz K, Germano M, van Grondelle R, Dekker JP (2005) Kinetics of excitation trapping in intact Photosystem I of Chlamydomonas reinhardtii and Arabidopsis thaliana. Biochim Biophys Acta 1706(3):267–275
Ikegami I (1976) Fluorescence changes related in the primary photochemical reaction in the P700-enriched particles isolated from spinach chloroplasts. Biochim Biophys Acta 449(2):245–258
Jagannathan B, Golbeck JH (2009a) Breaking biological symmetry in membrane proteins: the asymmetrical orientation of PsaC on the pseudo-C2 symmetric Photosystem I core. Cell Mol Life Sci 66(7):1257–1270
Jagannathan B, Golbeck JH (2009b) Understanding of the binding interface between PsaC and the PsaA/PsaB heterodimer in Photosystem I. Biochemistry 48(23):5405–5416
Jennings RC, Zucchelli G, Santabarbara S (2013) Photochemical trapping heterogeneity as a function of wavelength, in plant Photosystem I (PSI-LHCI). Biochim Biophys Acta 1827(6):779–785
Kleinherenbrink FAM, Aartsma TJ, Amesz J (1991) Charge separation and formation of bacteriochlorophyll triplets in Heliobacterium chlorum. Biochim Biophys Acta 1057(3):346–352
Kleinherenbrink FA, Ikegami I, Hiraishi A, Otte SCM, Amesz J (1993) Electron transfer in menaquinone-depleted membranes of Heliobacterium chlorum. Biochim Biophys Acta 1142:69–73
Kleinherenbrink FAM, Hastings G, Wittmershaus BP, Blankenship RE (1994) Delayed fluorescence from Fe–S Type photosynthetic reaction centers at low redox potential. Biochem 33(10):3096–3105
Kramer DM, Schoepp B, Liebl U, Nitschke W (1997) Cyclic electron transfer in Heliobacillus mobilis involving a menaquinol-oxidizing cytochrome bc complex and an RCI-type reaction center. Biochemistry 36(14):4203–4211
Liddell PA, Gervaldo M, Bridgewater JW, Keirstead AE, Lin S, Moore TA, Moore AL, Gust D (2008) Porphyrin-based hole conducting electropolymer. Chem Mater 20:135–142
Lin S, Chiou HC, Blankenship RE (1995) Secondary electron transfer processes in membranes of Heliobacillus mobilis. Biochemistry 34(39):12761–12767
Miloslavina Y, Szczepaniak M, Muller MG, Sander J, Nowaczyk M, Rogner M, Holzwarth AR (2006) Charge separation kinetics in intact Photosystem II core particles is trap-limited. A picosecond fluorescence study. Biochemistry 45(7):2436–2442
Miyamoto R, Mino H, Kondo T, Itoh S, Oh-Oka H (2008) An electron spin-polarized signal of the P800 +A1(Q)− state in the homodimeric reaction center core complex of Heliobacterium modesticaldum. Biochemistry 47(15):4386–4393
Moya I, Hodges M, Barbet J-C (1986) Modification of room-temperature picosecond chlorophyll fluorescence kinetics in green algae by Photosystem II trap closure. FEBS Lett 198:256–262
Muhiuddin IP, Rigby SE, Evans MC, Amesz J, Heathcote P (1999) ENDOR and special TRIPLE resonance spectroscopy of photoaccumulated semiquinone electron acceptors in the reaction centers of green sulfur bacteria and heliobacteria. Biochemistry 38(22):7159–7167
Müller MG, Niklas J, Lubitz W, Holzwarth AR (2003) Ultrafast transient absoprtion studies on Photosystem I reaction centers from Chlamydomonas reinhardtii I. A new interpretation of the energy trapping and early electron transfer steps in Photosystem I. Biophys J 85(6):3899–3922
Müller MG, Slavov C, Luthra R, Redding KE, Holzwarth AR (2010) Independent initiation of primary electron transfer in the two branches of the Photosystem I reaction center. Proc Natl Acad Sci USA 107(9):4123–4128
Neerken S, Amesz J (2001) The antenna reaction center complex of heliobacteria: composition, energy conversion and electron transfer. Biochim Biophys Acta 1507(1–3):278–290
Nitschke W, Liebl U, Matsuura K, Kramer DM (1995) Membrane-bound c-type cytochromes in Heliobacillus mobilis. In vivo study of the hemes involved in electron donation to the photosynthetic reaction center. Biochemistry 34(37):11831–11839
Oh-oka H, Iwaki M, Itoh S (2002) Electron donation from membrane-bound cytochrome c to the photosynthetic reaction center in whole cells and isolated membranes of Heliobacterium gestii. Photosynth Res 71(1–2):137–147
Parrett KG, Mehari T, Warren PG, Golbeck JH (1989) Purification and properties of the intact P-700 and FX-containing Photosystem I core protein. Biochim Biophys Acta 973(2):324–332
Rappaport F, Diner BA, Redding K (2006) Optical measurements of secondary electron transfer in Photosystem I. In: Golbeck J (ed) Photosystem I: the plastocyanin:ferredoxin oxidoreductase in photosynthesis. Kluwer Academic, Dordrecht, pp 223–244
Reed DW, Zankel KL, Clayton RK (1969) The effect of redox potential on P870 fluorescence in reaction centers from Rhodopseudomonas spheroides. Proc Natl Acad Sci USA 63(1):42–46
Roelofs TA, Lee CH, Holzwarth AR (1992) Global target analysis of picosecond chlorophyll fluorescence kinetics from pea chloroplasts: a new approach to the characterization of the primary processes in Photosystem II alpha- and beta-units. Biophys J 61(5):1147–1163
Romberger SP, Golbeck JH (2012) The FX iron-sulfur cluster serves as the terminal bound electron acceptor in heliobacterial reaction centers. Photosynth Res 111:285–290
Romberger SP, Castro C, Sun Y, Golbeck JH (2010) Identification and characterization of PshBII, a second F A/F B-containing polypeptide in the photosynthetic reaction center of Heliobacterium modesticaldum. Photosynth Res 104(2–3):293–303
Samson G, Prasil O, Yaakoubd B (1999) Photochemical and thermal phases of chlorophyll a fluorescence. Photosynthetica 37(2):163–182
Santabarbara S, Heathcote P, Evans MC (2005) Modelling of the electron transfer reactions in Photosystem I by electron tunnelling theory: the phylloquinones bound to the PsaA and the PsaB reaction centre subunits of PS I are almost isoenergetic to the iron-sulfur cluster FX. Biochim Biophys Acta 1708(3):283–310
Sarrou I, Khan Z, Cowgill J, Lin S, Brune D, Romberger S, Golbeck JH, Redding KE (2012) Purification of the photosynthetic reaction center from Heliobacterium modesticaldum. Photosynth Res 111(3):291–302
Schatz GH, Brock H, Holzwarth AR (1988) Kinetic and energetic model for the primary processes in Photosystem II. Biophys J 54(3):397–405
Sétif P, Brettel K (1993) Forward electron transfer from phylloquinone A1 to iron–sulfur centers in spinach Photosystem I. Biochem 32(31):7846–7854
Slavov C, Ballottari M, Morosinotto T, Bassi R, Holzwarth AR (2008) Trap-limited charge separation kinetics in higher plant Photosystem I complexes. Biophys J 94(9):3601–3612
Tripathy BC, Draheim JE, Anderson GP, Gross EL (1984) Variable fluorescence of Photosystem I particles and its application to the study of the structure and function of Photosystem I. Arch Biochem Biophys 235(2):449–460
van der Est A, Hager-Braun C, Leibl W, Hauska G, Stehlik D (1998) Transient electron paramagnetic resonance spectroscopy on green-sulfur bacteria and heliobacteria at two microwave frequencies. Biochim Biophys Acta 1409(2):87–98
van Grondelle R, Holmes NG, Rademaker H, Duysens LN (1978) Bacteriochlorophyll fluorescence of purple bacteria at low redox potentials. The relationship between reaction center triplet yield and the emission yield. Biochim Biophys Acta 503(1):10–25
Vrieze J, Van de Meent EJ, Hoff AJ (1998) Triplet properties and interactions of the primary electron donor and antenna chromophores in membranes of Heliobacterium chlorum, studied with ADMR spectroscopy. Biochemistry 37(42):14900–14909
White NTH, Beddard GS, Thorne JRG, Feehan TM, Keyes TE, Heathcote P (1996) Primary charge separation and energy transfer in the Photosystem I reaction center of higher plants. J Phys Chem 100(29):12086–12099
Wientjes E, van Stokkum IH, van Amerongen H, Croce R (2011) The role of the individual Lhcas in Photosystem I excitation energy trapping. Biophys J 101(3):745–754
Zankel KL, Reed DW, Clayton RK (1968) Fluorescence and photochemical quenching in photosynthetic reaction centers. Proc Natl Acad Sci USA 61(4):1243–1249
Acknowledgments
Much of this work was performed while KR was visiting the group of Francis-André Wollman at the Institut de Biologie Physico-Chimique (Paris) as a J. William Fulbright Scholar. KR is grateful to the FAW and the CNRS, as well as the Fulbright program, for their support during his stay in Paris. Work performed in KR’s laboratory at ASU was supported by the Division of Chemical Sciences, Geosciences, and Biosciences, Office of Basic Energy Sciences of the U.S. Department of Energy through Grant DE-SC0010575.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Redding, K.E., Sarrou, I., Rappaport, F. et al. Modulation of the fluorescence yield in heliobacterial cells by induction of charge recombination in the photosynthetic reaction center. Photosynth Res 120, 221–235 (2014). https://doi.org/10.1007/s11120-013-9957-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-013-9957-4