Abstract
Aims
This study aims to understand the mechanism by which grafting tomato (Solanum lycopersicum) onto Torubamu (Solanum melongena) reduces cadmium (Cd) accumulation in tomato.
Methods
Tomato plants were grafted onto Torubamu rootstocks, and both the un-grafted and grafted tomato plants were grown in the control soil and soil that contained 2.0 or 5.0 mg•kg−1 Cd for 30 d.
Results
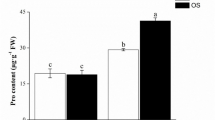
Grafting onto Torubamu rootstock markedly reduced the accumulation of both Cd and total sulfur in the leaves of tomato plants to 4.1–11.7% and 25.0–36.7% of the values in the un-grafted tomato plants, respectively. Moreover, the expression of miR166a and miR395b markedly increased in tomato scions. Tobacco dual-luciferase (LUC) assays and Agrobacterium-mediated tomato genetic transformation verified that miR166a and miR395b targeted Phavoluta (PHV), Revolute (REV), Class III Homeodomain Leucine Zipper (HD-ZIP), Sulfate transporter 2;1 (SULTR2;1), ATP sulfurylase (APS) (APS1/3), and High-affinity sulfate transporter (HAST) genes, which are directly or indirectly responsible for sulfate transport.
Conclusions
Based on the correlations between the Cd and sulfur accumulation in the tomato scions (r2 = 0.91) and in the miR166a/395b-transformed tomato scions (r2 = 0.96–0.98) grown in the soil with 2.0 mg•kg−1 Cd, we conclude that tomato grafting onto Torubamu rootstock can reduce Cd accumulation because of the sulfate transport regulatory ability of miR166a and miR395b. Understanding this mechanism can be of great benefit for Cd tolerance breeding.




Similar content being viewed by others
References
Adhikari S, Ghosh S, Azahar I, Adhikari A, Hossain Z (2018) Sulfate improves cadmium tolerance by limiting cadmium accumulation, modulation of sulfur metabolism and antioxidant defense system in maize. Environ Exp Bot 153:143–162. https://doi.org/10.1016/j.envexpbot.2018.05.008
Albacete A, Martinez-Andujar C, Martinez-Perez A, Thompson AJ, Dodd IC, Perez-Alfocea F (2015) Unravelling rootstockxscion interactions to improve food security. J Exp Bot 66:2211–2226. https://doi.org/10.1093/jxb/erv027
Baev V, Milev I, Naydenov M, Vachev T, Apostolova E, Mehterov N, Yahubyan G (2014) Insight into small RNA abundance and expression in high- and low-temperature stress response using deep sequencing in Arabidopsis. Plant Physiol Biochem 84:105–114. https://doi.org/10.1016/j.plaphy.2014.09.007
Basso MF, Ferreira PCG, Kobayashi AK, Harmon FG, Nepomuceno AL, Molinari HBC, Grossi‐De‐Sa MF (2019) MicroRNAs and new biotechnological tools for its modulation and improving stress tolerance in plants. Plant Biotechnol J 17:1482–1500. https://doi.org/10.1111/pbi.13116
Buhtz A, Pieritz J, Springer F, Kehr J (2010) Phloem small RNAs, nutrient stress responses, and systemic mobility. BMC Plant Biol 10:64. https://doi.org/10.1186/1471-2229-10-64
Carlsbecker A, Lee JY, Roberts CJ, Dettmer J, Lehesranta S, Zhou J, Benfey PN (2010) Cell signalling by microRNA165/6 directs gene dose-dependent root cell fate. Nature 465:316–321. https://doi.org/10.1038/nature08977
Chen Z, Tang YT, Yao AJ, Cao J, Wu ZH, Peng ZR, Qiu RL (2017) Mitigation of Cd accumulation in paddy rice ( Oryza sativa L.) by Fe fertilization. Environ Pollut 231:549–559. https://doi.org/10.1016/j.envpol.2017.08.055
Cheng Z, Ding Y, Liu H (2011) MiR398 and plant stress responses. Physiol Plant 143:1–9. https://doi.org/10.1111/j.1399-3054.2011.01477.x
Cimini S, Gualtieri C, Macovei A, Balestrazzi A, De Gara L, Locato V (2019) Redox balance-DDR-miRNA triangle: relevance in genome stability and stress responses in plants. Front Plant Sci 10:989. https://doi.org/10.3389/fpls.2019.00989
Ding Y, Yaoyao Y, Zhihua J, Yi W, Cheng Z (2016) MicroRNA390 is involved in cadmium tolerance and accumulation in rice. Front Plant Sci 7:235. https://doi.org/10.3389/fpls.2016.00235
Ding Y, Gong S, Wang Y, Wang F, Bao H, Sun J, Zhu C (2018) MicroRNA166 modulates cadmium tolerance and accumulation in rice. Plant Physiol 177:1691–1703. https://doi.org/10.1104/pp.18.00485
Djami-Tchatchou AT, Neeti S-M, Khayalethu N, Dubery IA (2017) Functional roles of microRNAs in agronomically important plants—potential as targets for crop improvement and protection. Front Plant Sci 8:378. https://doi.org/10.3389/fpls.2017.00378
Frary A, Eck JV (2005) Organogenesis from transformed tomato explants. Methods Mol Biol 286:141–150. https://doi.org/10.1385/1-59259-827-7:141
Gallego SM, Pena LB, Barcia RA, Azpilicueta CE, Iannone MF, Rosales EP et al (2012) Unravelling cadmium toxicity and tolerance in plants: insight into regulatory mechanisms. Environ Exp Bot 83:33–46. https://doi.org/10.1016/j.envexpbot.2012.04.006
Jeelani N, Zhu Z, Wang P, Zhang P, Song S, Yuan H, Leng X (2017) Assessment of trace metal contamination and accumulation in sediment and plants of the Suoxu river, China. Aquat Bot 140:92–95. https://doi.org/10.1016/j.aquabot.2016.11.007
Joseph P (2009) Mechanisms of cadmium carcinogenesis. Toxicol Appl Pharmacol 238:272–279. https://doi.org/10.1016/j.taap.2009.01.011
Kumar, Rajesh (2014) Role of MicroRNAs in biotic and abiotic stress responses in crop plants. Appl Biochem Biotechnol 174:93–115. https://doi.org/10.1007/s12010-014-0914-2
Kumar S, Verma S, Trivedi PK (2017) Involvement of Small RNAs in Phosphorus and Sulfur Sensing, Signaling and Stress: Current Update. Front Plant Sci 8:285. https://doi.org/10.3389/fpls.2017.00285
Li P, Wang X, Allinson G, Li X, Xiong X (2009) Risk assessment of heavy metals in soil previously irrigated with industrial wastewater in Shenyang, China. J Hazard Mater 161:516–521. https://doi.org/10.1016/j.jhazmat.2008.03.130
Li C, Yu X, Bai L, He C, Li Y (2016) Responses of miRNAs and their target genes to nitrogen- or phosphorus-deficiency in grafted cucumber seedlings. Hortic Environ Biotechnol 57:97–112. https://doi.org/10.1007/s13580-016-0092-y
Liang J, Shohag M, Yang X, Tian S, Zhang Y, Feng Y, He Z (2014) Role of sulfur assimilation pathway in cadmium hyperaccumulation by Sedum alfredii Hance. Ecotoxicol Environ Saf 100:159–165. https://doi.org/10.1016/j.ecoenv.2013.10.026
Liu Q, Axtell MJ (2015) Quantitating plant MicroRNA-mediated target repression using a dual-luciferase transient expression system. Methods Mol Biol 1284:287–303. https://doi.org/10.1007/978-1-4939-2444-8_14
Liu Q, Wang F, Axtell MJ (2014) Analysis of complementarity requirements for plant MicroRNA targeting using a nicotiana benthamiana quantitative transient assay. Plant Cell 26:741–753. https://doi.org/10.1105/tpc.113.120972
Liu Q, Yan S, Yang T, Zhang S, Liu B (2017) Small RNAs in regulating temperature stress response in plants: small RNAs regulation of plant temperature stress. J Integr Plant Biol 59:774–791. https://doi.org/10.1111/jipb.12571
Manara A (2012) Plant responses to heavy metal toxicity. Plants and Heavy Metals
Matthewman CA, Kawashima CG, Húska D, Csorba T, Dalmay T, Kopriva S (2012) miR395 is a general component of the sulfate assimilation regulatory network in Arabidopsis. FEBS Lett 586:3242–3248. https://doi.org/10.1016/j.febslet.2012.06.044
McConnell JR, Emery J, Eshed Y, Bao N, Bowman J, Barton MK (2001) Role of PHABULOSA and PHAVOLUTA in determining radial patterning in shoots. Nature 411:709–713. https://doi.org/10.1038/35079635
Megha S, Basu U, Kav NNV (2018) Regulation of low temperature stress in plants by microRNAs. Plant Cell Environ 41:1–15. https://doi.org/10.1111/pce.12956
Melnyk CW, Meyerowitz EM (2015) Plant grafting. Curr Biol 25:R183–R188. https://doi.org/10.1016/j.cub.2015.01.029
Moyle R, Carvalhais CL, Lara-Simone P, Ekaterina N, Gayathery S, Jessica D-M, Schenk PM (2017) An optimized transient dual luciferase assay for quantifying MicroRNA directed repression of targeted sequences. Front Plant Sci. https://doi.org/10.3389/fpls.2017.01631
Noman A, Aqeel M (2017) miRNA-based heavy metal homeostasis and plant growth. Environ Sci Pollut Res 24:10068–10082. https://doi.org/10.1007/s11356-017-8593-5
Noman A, Fahad S, Aqeel M, Ali U, Amanullah, Anwar S, Zainab M (2017) miRNAs: major modulators for crop growth and development under abiotic stresses. Biotechnol Lett 39:685–700. https://doi.org/10.1007/s10529-017-2302-9
Patron NJ, Durnford DG, Kopriva S (2008) Sulfate assimilation in eukaryotes: fusions, relocations and lateral transfers. BMC Evol Biol 8:39. https://doi.org/10.1186/1471-2148-8-39
Qin Q, Li X, Zhuang J, Weng L, Liu W, Tai P (2015) Long-distance transport of cadmium from roots to leaves ofSolanum melongena. Ecotoxicology 24:2224–2232. https://doi.org/10.1007/s10646-015-1546-1
Rosenfeld CE, Chaney RL, Martínez CE (2018) Soil geochemical factors regulate Cd accumulation by metal hyperaccumulating Noccaea caerulescens (J. Presl & C. Presl) F.K. Mey in field-contaminated soils. Sci Total Environ 616-617:279–287. https://doi.org/10.1016/j.scitotenv.2017.11.016
Rouphael Y, Cardarelli M, Rea E, Colla G (2008) Grafting of cucumber as a means to minimize copper toxicity. Environ Exp Bot 63:49–58. https://doi.org/10.1016/j.envexpbot.2007.10.015
Sauvé S, Norvell WA, McBride M, Hendershot W (2000) Speciation and complexation of cadmium in extracted soil solutions. Environ Sci Technol 34:291–296. https://doi.org/10.1021/es990202z
Savvas D, Colla G, Rouphael Y, Schwarz D (2010) Amelioration of heavy metal and nutrient stress in fruit vegetables by grafting. Sci Hortic 127:156–161. https://doi.org/10.1016/j.scienta.2010.09.011
Singh, Kumar N (2017) microRNAs databases: developmental methodologies, structural and functional annotations. Interdiscip Sci Comput Life Sci 9:357–377. https://doi.org/10.1007/s12539-016-0166-7
Singh A, Singh S, Panigrahi KC, Reski R, Sarkar AK (2014) Balanced activity of microRNA166/165 and its target transcripts from the class III homeodomain-leucine zipper family regulates root growth in Arabidopsis thaliana. Plant Cell Rep 33:945–953. https://doi.org/10.1007/s00299-014-1573-z
Sun Y, Zhou Q, Diao C (2008) Effects of cadmium and arsenic on growth and metal accumulation of Cd-hyperaccumulator Solanum nigrum L. Bioresour Technol 99:1103–1110. https://doi.org/10.1016/j.biortech.2007.02.035
Sun X, Lin L, Sui N (2019) Regulation mechanism of microRNA in plant response to abiotic stress and breeding. Mol Biol Rep 46:1447–1457. https://doi.org/10.1007/s11033-018-4511-2
Sunkar R, Li Y-F, Jagadeeswaran G (2012) Functions of microRNAs in plant stress responses. Trends Plant Sci 17:196–203. https://doi.org/10.1016/j.tplants.2012.01.010
Takahashi H, Watanabe-Takahashi A, Smith FW, Blake-Kalff M, Saito K (2001) The roles of three functional transporters involved in uptake and translocation of sulphate in Arabidopsis thaliana. Plant J 23:171–182. https://doi.org/10.1046/j.1365-313x.2000.00768.x
Thom A, Thom AS (2008) Reduction of cadmium translocation from roots to shoots in eggplant ( Solanum melongena ) by grafting onto Solanum torvum rootstock. Soil Sci Plant Nutr 54. https://doi.org/10.1111/j.1747-0765.2008.00269.x
Varsha S, Vinay K, Devarumath RM, Khare TS, Wani SH (2016) MicroRNAs as potential targets for abiotic stress tolerance in plants. Front Plant Sci 7:8178. https://doi.org/10.3389/fpls.2016.00817
Walker EL, Eggleston WB, Demopulos D, Kermicle J, Dellaporta SL (1997) Insertions of a novel class of transposable elements with a strong target site preference at the r locus of maize. Genetics 146:681–693
Wang H, He L, Song J, Cui W, Zhang Y, Jia C, Tai P (2016) Cadmium-induced genomic instability in Arabidopsis: molecular toxicological biomarkers for early diagnosis of cadmium stress. Chemosphere 150:258–265. https://doi.org/10.1016/j.chemosphere.2016.02.042
Wang J, Jiang L, Wu R (2017) Plant grafting: how genetic exchange promotes vascular reconnection. New Phytol 214:56–65. https://doi.org/10.1111/nph.14383
Wang W, Liu D, Chen D, Cheng Y, Zhang X, Song L, Shen F (2019) MicroRNA414c affects salt tolerance of cotton by regulating reactive oxygen species metabolism under salinity stress. RNA Biol 16:362–375. https://doi.org/10.1080/15476286.2019.1574163
Xu ZM, Tan XQ, Mei XQ, Li QS, Zhou C, Wang LL, Yang P (2018) Low-Cd tomato cultivars (Solanum lycopersicum L.) screened in non-saline soils also accumulated low Cd, Zn, and Cu in heavy metal-polluted saline soils. Environ Sci Pollut Res Int 25:27439–27450. https://doi.org/10.1007/s11356-018-2776-6
Yang Y, Zhou X, Tie B, Peng L, Li H, Wang K, Zeng Q (2017) Comparison of three types of oil crop rotation systems for effective use and remediation of heavy metal contaminated agricultural soil. Chemosphere 188:148–156. https://doi.org/10.1016/j.chemosphere.2017.08.140
Yu H, Wang J, Fang W, Yuan J, Yang Z (2006) Cadmium accumulation in different rice cultivars and screening for pollution-safe cultivars of rice. Sci Total Environ 370:302–309. https://doi.org/10.1016/j.scitotenv.2006.06.013
Yuan H, Sun L, Tai P, Liu W, Li X, Hao L (2019) Effects of grafting on root-to-shoot cadmium translocation in plants of eggplant (Solanum melongena) and tomato (Solanum lycopersicum). Sci Total Environ 652:989–995. https://doi.org/10.1016/j.scitotenv.2018.10.129
Zabala G, Vodkin LO (2005) The wp mutation of Glycine max carries a gene-fragment-rich transposon of the CACTA superfamily. Plant Cell 17:2619–2632. https://doi.org/10.1105/tpc.105.033506
Zhang B (2015) MicroRNA: a new target for improving plant tolerance to abiotic stress. J Exp Bot 66:1749–1761. https://doi.org/10.1093/jxb/erv013
Zhong R, Ye Z (2004) amphivasal vascular bundle 1, a gain-of-function mutation of the IFL1/REV Gene, is associated with alterations in the polarity of leaves, stems and carpels. Plant Cell Physiol 45:369–385. https://doi.org/10.1093/pcp/pch051
Zhu Y, Yu H, Wang J, Fang W, Yuan J, Yang Z (2007) Heavy metal accumulations of 24 asparagus bean cultivars grown in soil contaminated with Cd alone and with multiple metals (Cd, Pb, and Zn). J Agric Food Chem 55:1045–1052. https://doi.org/10.1021/jf062971p
Acknowledgements
This research was financially sponsored by National Natural Science Foundation of China (NSFC, 41877146, 41807488, 21677151, 41673132) and the National Key Research and Development Program of China (2016YFD0800305).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Antony Van der Ent.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hetong Wang is the Co-first author.
Electronic supplementary material
ESM 1
(DOC 336 kb)
Rights and permissions
About this article
Cite this article
He, L., Wang, H., Zhao, Q. et al. Tomato grafting onto Torubamu (Solanum melongena): miR166a and miR395b reduce scion Cd accumulation by regulating sulfur transport. Plant Soil 452, 267–279 (2020). https://doi.org/10.1007/s11104-020-04564-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04564-7