Abstract
Key message
A first creation of high oleic acid peanut varieties by using transcription activator-like effecter nucleases (TALENs) mediated targeted mutagenesis of Fatty Acid Desaturase 2 (FAD2).
Abstract
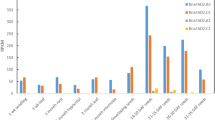
Transcription activator like effector nucleases (TALENs), which allow the precise editing of DNA, have already been developed and applied for genome engineering in diverse organisms. However, they are scarcely used in higher plant study and crop improvement, especially in allopolyploid plants. In the present study, we aimed to create targeted mutagenesis by TALENs in peanut. Targeted mutations in the conserved coding sequence of Arachis hypogaea fatty acid desaturase 2 (AhFAD2) were created by TALENs. Genetic stability of AhFAD2 mutations was identified by DNA sequencing in up to 9.52 and 4.11% of the regeneration plants at two different targeted sites, respectively. Mutation frequencies among AhFAD2 mutant lines were significantly correlated to oleic acid accumulation. Genetically, stable individuals of positive mutant lines displayed a 0.5–2 fold increase in the oleic acid content compared with non-transgenic controls. This finding suggested that TALEN-mediated targeted mutagenesis could increase the oleic acid content in edible peanut oil. Furthermore, this was the first report on peanut genome editing event, and the obtained high oleic mutants could serve for peanut breeding project.





Similar content being viewed by others
References
Akhtar S, Khalid N, Ahmed I, Shahzad A, Suleria HA (2014) Physicochemical characteristics, functional properties, and nutritional benefits of peanut oil: a review. Crit Rev Food Sci Nutr 54:1562–1575. https://doi.org/10.1080/10408398.2011.644353
Blanvillain-Baufume S, Reschke M, Sole M, Auguy F, Doucoure H, Szurek B, Meynard D, Portefaix M, Cunnac S, Guiderdoni E, Boch J, Koebnik R (2017) Targeted promoter editing for rice resistance to Xanthomonas oryzae pv. oryzae reveals differential activities for SWEET14-inducing TAL effectors. Plant Biotechnol J 15:306–317. https://doi.org/10.1111/pbi.12613
Broun P, Gettner S, Somerville C (1999) Genetic engineering of plant lipids. Annu Rev Nutr 19:197–216. https://doi.org/10.1146/annurev.nutr.19.1.197
Chen Z, Wang ML, Barkley NA, Pittman RN (2010) A simple allele-specific PCR assay for detecting FAD2 alleles in both A and B genomes of the cultivated peanut for high-oleate trait selection. Plant Mol Biol Rep 28:542–548. https://doi.org/10.1007/s11105-010-0181-5
Chi X, Yang Q, Pan L, Chen M, He Y, Yang Z, Yu S (2011). Isolation and characterization of fatty acid desaturase genes from peanut (Arachis hypogaea L.). Plant Cell Rep 30:1393–1404. https://doi.org/10.1007/s00299-011-1048-4
Clevenger J, Chu Y, Chavarro C, Agarwal G, Bertioli DJ, Leal-Bertioli S, Pandey MK, Vaughn J, Abernathy B, Barkley NA, Hovav R, Burow M, Nayak SN, Chitikineni A, Isleib TG, Holbrook CC, Jackson SA, Varshney RK, Ozias-Akins P (2017) Genome-wide SNP genotyping resolves signatures of selection and tetrasomic recombination in peanut. Mol Plant 10:309–322. https://doi.org/10.1016/j.molp.2016.11.015
Doyle EL, Booher NJ, Standage DS, Voytas DF, Brendel VP, Vandyk JK, Bogdanove AJ (2012) TAL effector-nucleotide targeter (TALE-NT) 2.0: tools for TAL effector design and target prediction. Nucleic Acids Res 40:W117-22. https://doi.org/10.1093/nar/gks608
Endo M, Nishizawa-Yokoi A, Toki S (2016). Targeted mutagenesis in rice using TALENs and the CRISPR/Cas9 system. Methods Mol Biol 1469:123–135. https://doi.org/10.1007/978-1-4939-4931-1_9
Frank MH, Chitwood DH (2016) Plant chimeras: the good, the bad, and the ‘Bizzaria’. Dev Biol 419:41–53. https://doi.org/10.1016/j.ydbio.2016.07.003
Haun W, Coffman A, Clasen BM, Demorest ZL, Lowy A, Ray E, Retterath A, Stoddard T, Juillerat A, Cedrone F, Mathis L, Voytas DF, Zhang F (2014). Improved soybean oil quality by targeted mutagenesis of the fatty acid desaturase 2 gene family. Plant Biotechnol J 12:934–940. https://doi.org/10.1111/pbi.12201
Janila P, Pandey MK, Shasidhar Y, Variath MT, Sriswathi M, Khera P, Manohar SS, Nagesh P, Vishwakarma MK, Mishra GP, Radhakrishnan T, Manivannan N, Dobariya KL, Vasanthi RP, Varshney RK (2016) Molecular breeding for introgression of fatty acid desaturase mutant alleles (ahFAD2A and ahFAD2B) enhances oil quality in high and low oil containing peanut genotypes. Plant Sci 242:203–213. https://doi.org/10.1016/j.plantsci.2015.08.013
Ji J, Tang D, Wang M, Li Y, Zhang L, Wang K, Li M, Cheng Z (2013). MRE11 is required for homologous synapsis and DSB processing in rice meiosis. Chromosoma 122:363–376. https://doi.org/10.1007/s00412-013-0421-1
Jung JH, Altpeter F (2016). TALEN mediated targeted mutagenesis of the caffeic acid O-methyltransferase in highly polyploid sugarcane improves cell wall composition for production of bioethanol. Plant Mol Biol 92:131–142. https://doi.org/10.1007/s11103-016-0499-y
Li T, Liu B, Spalding MH, Weeks DP, Yang B (2012) High-efficiency TALEN-based gene editing produces disease-resistant rice. Nat Biotechnol 30:390–392. https://doi.org/10.1038/nbt.2199
Lowder L, Malzahn A, Qi Y (2016) Rapid evolution of manifold CRISPR systems for plant genome editing. Front Plant Sci 7:1683. https://doi.org/10.3389/fpls.2016.01683
Mahfouz MM, Piatek A, Stewart CJ (2014) Genome engineering via TALENs and CRISPR/Cas9 systems: challenges and perspectives. Plant Biotechnol J 12:1006–1014. https://doi.org/10.1111/pbi.12256
Malzahn A, Lowder L, Qi Y (2017) Plant genome editing with TALEN and CRISPR. Cell Biosci 7:21. https://doi.org/10.1186/s13578-017-0148-4
McGinnis KM (2010) RNAi for functional genomics in plants. Brief Funct Genom 9:111–117. https://doi.org/10.1093/bfgp/elp052
Okuley J, Lightner J, Feldmann K, Yadav N, Lark E, Browse J (1994). Arabidopsis FAD2 gene encodes the enzyme that is essential for polyunsaturated lipid synthesis. Plant Cell 6:147–158. https://doi.org/10.1105/tpc.6.1.147
Pandey MK, Wang ML, Qiao L, Feng S, Khera P, Wang H, Tonnis B, Barkley NA, Wang J, Holbrook CC, Culbreath AK, Varshney RK, Guo B (2014) Identification of QTLs associated with oil content and mapping FAD2 genes and their relative contribution to oil quality in peanut (Arachis hypogaea L.). BMC Genet 15:133. https://doi.org/10.1186/s12863-014-0133-4
Riveros CG, Mestrallet MG, Gayol MF, Quiroga PR, Nepote V, Grosso NR (2010) Effect of storage on chemical and sensory profiles of peanut pastes prepared with high-oleic and normal peanuts. J Sci Food Agric 90:2694–2699. https://doi.org/10.1002/jsfa.4142
Rodriguez-Leal D, Lemmon ZH, Man J, Bartlett ME, Lippman ZB (2017) Engineering quantitative trait variation for crop improvement by genome editing. Cell 171:470–480.e8. https://doi.org/10.1016/j.cell.2017.08.030
Sharma KK, Anjaiah VV (2000) An efficient method for the production of transgenic plants of peanut (Arachis hypogaea L.) through Agrobacterium tumefaciens-mediated genetic transformation. Plant Sci 159:7–19
Tian HC, Marcotrigiano M (1993). Origin and development of adventitious shoot meristems initiated on plant chimeras. Dev Biol 155:259–269. https://doi.org/10.1006/dbio.1993.1023
Tiwari V, Chaturvedi AK, Mishra A, Jha B (2015). An efficient method of agrobacterium-mediated genetic transformation and regeneration in local Indian cultivar of groundnut (Arachis hypogaea) using grafting. Appl Biochem Biotechnol 175:436–453. https://doi.org/10.1007/s12010-014-1286-3
Tong C, Huang G, Ashton C, Wu H, Yan H, Ying QL (2012). Rapid and cost-effective gene targeting in rat embryonic stem cells by TALENs. J Genet Genom 39:275–280. https://doi.org/10.1016/j.jgg.2012.04.004
Van Eck J (2017) Genome editing and plant transformation of solanaceous food crops. Curr Opin Biotechnol 49:35–41. https://doi.org/10.1016/j.copbio.2017.07.012
Vassiliou EK, Gonzalez A, Garcia C, Tadros JH, Chakraborty G, Toney JH (2009) Oleic acid and peanut oil high in oleic acid reverse the inhibitory effect of insulin production of the inflammatory cytokine TNF-alpha both in vitro and in vivo systems. Lipids Health Dis 8:25. https://doi.org/10.1186/1476-511X-8-25
Wang Y, Zhang XG, Zhao YL, Prakash CS, He GH, Yin DM (2015) Insights into the novel members of the FAD2 gene family involved in high-oleate fluxes in peanut. Genome 58:375–383. https://doi.org/10.1139/gen-2015-0008
Yin K, Gao C, Qiu JL (2017) Progress and prospects in plant genome editing. Nat Plants 3:17107. https://doi.org/10.1038/nplants.2017.107
Zaloga GP, Harvey KA, Stillwell W, Siddiqui R (2006). Trans fatty acids and coronary heart disease. Nutr Clin Pract 21:505–512. https://doi.org/10.1177/0115426506021005505
Acknowledgements
This study was supported by the Science and Technology Planning Project of Guangdong Province (2015B020231006, 2016B020201003 and 2016LM3161), the National Natural Science Foundation of China (31501246), and the Modern Agro-industry Technology Research System (CARS-14).
Author information
Authors and Affiliations
Contributions
HL, QL, and YH conceived the original screening and research plans. XC supervised the experiments. SW, XL performed most of the experiments. HL designed the experiments and analyzed the data. QL conceived the project and wrote the article with contributions of all the authors. SW, HL supervised and complemented the writing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wen, S., Liu, H., Li, X. et al. TALEN-mediated targeted mutagenesis of fatty acid desaturase 2 (FAD2) in peanut (Arachis hypogaea L.) promotes the accumulation of oleic acid. Plant Mol Biol 97, 177–185 (2018). https://doi.org/10.1007/s11103-018-0731-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-018-0731-z