Abstract
Background
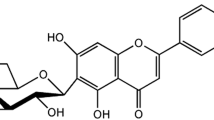
Isofraxidin is a coumarin compound mainly isolated from several traditional and functional edible plants beneficial for neurodegenerative diseases, including Sarcandra glabra and Apium graveolens, and Siberian Ginseng.
Objective
This study aimed to assess effects of isofraxidin against memory impairments and cognition deficits in a scopolamine-induced mouse model.
Materials & methods
Animals were randomly divided into 6 groups, control, vehicle, donepezil (10 mg/kg, p.o.), and isofraxidin (3, 10, and 30 mg/kg, p.o.). Isofraxidin or donepezil was administered for 44 days, once per day. The scopolamine insults (1 mg/kg, i.p.) was given from the 21st day, once per day. Morris water maze test and Y-maze test were used for the behavioral test. After that, brain samples were collected for analysis.
Results
Firstly, isofraxidin significantly improved scopolamine-induced behavioral impairments and cognition deficits in Morris water maze and Y-maze test. Then, isofraxidin facilitated cholinergic activity via inhibiting acetylcholinesterase (AChE) activity. Besides, isofraxidin decreased lipid peroxidation level but enhanced levels of glutathione, glutathione peroxidase, and superoxide dismutase. Moreover, isofraxidin suppressed the expression of inflammatory mediators and cytokines. Further investigations showed that isofraxidin up-regulated expression of brain-derived neurotrophic factor (BDNF), and promoted phosphorylation of tropomyosin-related kinase B (TrkB), cyclic AMP-response element-binding protein (CREB), and extracellular signal-regulated kinase (ERK).
Discussion & Conclusions
These results suggested that isofraxidin ameliorated scopolamine-induced cognitive and memory impairments, possibly through regulating AChE activity, suppressing oxidative stress and inflammatory response, and modulating BDNF-CREB-ERK pathways.








Similar content being viewed by others
Data availability statement
The data that support the findings of this study are available on requests from the corresponding authors.
References
Ahmad A, Ramasamy K, Jaafar SM, Majeed AB, Mani V (2014) Total isoflavones from soybean and tempeh reversed scopolamine-induced amnesia, improved cholinergic activities and reduced neuroinflammation in brain. Food Chem Toxicol 65:120–128 English
Bajo R, Pusil S, Lopez ME, Canuet L, Pereda E, Osipova D, Maestu F, Pekkonen E (2015) Scopolamine effects on functional brain connectivity: a pharmacological model of Alzheimer’s disease. Sci Rep 5:9748
Butterfield DA (2018) Perspectives on Oxidative Stress in Alzheimer’s Disease and Predictions of Future Research Emphases. J Alzheimers Dis
Chen H, Xiang S, Huang L, Lin J, Hu S, Mak SH, Wang C, Wang Q, Cui W, Han Y (2018) Tacrine(10)-hupyridone, a dual-binding acetylcholinesterase inhibitor, potently attenuates scopolamine-induced impairments of cognition in mice. Metab Brain Dis 33(4):1131–1139
Conrad CD, Galea LA, Kuroda Y, McEwen BS (1996) Chronic stress impairs rat spatial memory on the Y maze, and this effect is blocked by tianeptine pretreatment. Behav Neurosci 110(6):1321–1334
Deng X, Zhao S, Liu X, Han L, Wang R, Hao H, Jiao Y, Han S, Bai C (2020) Polygala tenuifolia: a source for anti-Alzheimer’s disease drugs. Pharm Biol 58(1):410–416 eng
Ennaceur A, Delacour J (1988) A new one-trial test for neurobiological studies of memory in rats. 1: Behavioral data. Behav Brain Res 31(1):47–59
Fabiani C, Murray AP, Corradi J, Antollini SS (2018) A novel pharmacological activity of caffeine in the cholinergic system. Neuropharmacology 135:464–473
Gan L, Johnson JA (2014) Oxidative damage and the Nrf2-ARE pathway in neurodegenerative diseases. Biochim Biophys Acta 1842(8):1208–1218
Hafez HS, Ghareeb DA, Saleh SR, Abady MM, El Demellawy MA, Hussien H, Abdel-Monem N (2017) Neuroprotective effect of ipriflavone against scopolamine-induced memory impairment in rats. Psychopharmacology 234(20):3037–3053 English
Hescham S, Grace L, Kellaway LA, Bugarith K, Russell VA (2009) Effect of exercise on synaptophysin and calcium/calmodulin-dependent protein kinase levels in prefrontal cortex and hippocampus of a rat model of developmental stress. Metab Brain Dis 24(4):701–709
Hoang THX, Ho DV, Van Phan K, Le QV, Raal A, Nguyen HT (2020) Effects of Hippeastrum reticulatum on memory, spatial learning and object recognition in a scopolamine-induced animal model of Alzheimer’s disease. Pharm Biol 58(1):1098–1104 eng
Huang HY, Ko HH, Jin YJ, Yang SZ, Shih YA, Chen IS (2012) Dihydrochalcone glucosides and antioxidant activity from the roots of Anneslea fragrans var. lanceolata [Article]. Phytochemistry 78:120–125
Hughes RN (2004) The value of spontaneous alternation behavior (SAB) as a test of retention in pharmacological investigations of memory. Neurosci Biobehav Rev 28(5):497–505
Ko YH, Kim SY, Lee SY, Jang CG (2018) 6,7,4’-Trihydroxyisoflavone, a major metabolite of daidzein, improves learning and memory via the cholinergic system and the p-CREB/BDNF signaling pathway in mice. Eur J Pharmacol 826:140–147 English
Kumar H, Koppula S, Kim IS, More SV, Kim BW, Choi DK (2012) Nuclear factor erythroid 2-related factor 2 signaling in Parkinson disease: a promising multi therapeutic target against oxidative stress, neuroinflammation and cell death. CNS Neurol Disord Drug Targets 11(8):1015–1029
Li C, Tang B, Feng Y, Tang F, Pui-Man Hoi M, Su Z, Ming-Yuen Lee S (2018) Pinostrobin Exerts Neuroprotective Actions in Neurotoxin-Induced Parkinson’s Disease Models through Nrf2 Induction. J Agric Food Chem 66(31):8307–8318
Li J, Li X, Li Z, Zhang L, Liu Y, Ding H, Yin S (2017) Isofraxidin, a coumarin component improves high-fat diet induced hepatic lipid homeostasis disorder and macrophage inflammation in mice. Food Funct 8(8):2886–2896 English
Li P, Zhao QL, Wu LH, Jawaid P, Jiao YF, Kadowaki M, Kondo T (2014) Isofraxidin, a potent reactive oxygen species (ROS) scavenger, protects human leukemia cells from radiation-induced apoptosis via ROS/mitochondria pathway in p53-independent manner [Article]. Apoptosis 19(6):1043–1053
Lim DW, Son HJ, Um MY, Kim IH, Han D, Cho S, Lee CH (2016) Enhanced Cognitive Effects of Demethoxycurcumin, a Natural Derivative of Curcumin on Scopolamine-Induced Memory Impairment in Mice.Molecules.21(8)
Liu W, Rabinovich A, Nash Y, Frenkel D, Wang YQ, Youdim MBH, Weinreb O (2017) Anti-inflammatory and protective effects of MT-031, a novel multitarget MAO-A and AChE/BuChE inhibitor in scopolamine mouse model and inflammatory cells. Neuropharmacology 113:445–456 English
Lombardo S, Maskos U (2015) Role of the nicotinic acetylcholine receptor in Alzheimer’s disease pathology and treatment. Neuropharmacology 96:255–262 English
Mietto BS, Mostacada K, Martinez AM (2015) Neurotrauma and inflammation: CNS and PNS responses. Mediat Inflamm. 2015:251204. English
Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods 11(1):47–60
Niu XF, Wang Y, Li WF, Mu QL, Li HN, Yao H, Zhang HL (2015) Protective effects of Isofraxidin against lipopolysaccharide-induced acute lung injury in mice. Int Immunopharmacol 24(2):432–439 English
Onasanwo SA, Adamaigbo VO, Adebayo OG, Eleazer SE (2021) Moringa oleifera-supplemented diet protect against cortico-hippocampal neuronal degeneration in scopolamine-induced spatial memory deficit in mice: role of oxido-inflammatory and cholinergic neurotransmission pathway. Metab Brain Dis 36(8):2445–2460
Polatoglu K, Yucel YY, Nalbantsoy A, Yalcin HT, Goren N (2017) Cytotoxic, antimicrobial activities, AChE and BChE inhibitory effects of compounds from Tanacetum chiliophyllum (Fisch & Mey.) Schultz Bip. var. oligocephalum (DC) Sosn. and T. chiliophyllum (Fisch & Mey.) Schultz Bip. var. monocephalum Grierson. Phytochem Lett 22:199–204 English
Ponne S, Kumar CR, Boopathy R (2020) Verapamil attenuates scopolamine induced cognitive deficits by averting oxidative stress and mitochondrial injury - A potential therapeutic agent for Alzheimer’s Disease. Metab Brain Dis 35(3):503–515
Rabiei Z, Setorki M (2018) Effect of hydroalcoholic Echium amoenum extract on scopolamine-induced learning and memory impairment in rats. Pharm Biol 56(1):672–677 eng
Rosenblum K, Futter M, Jones M, Hulme EC, Bliss TVP (2000) ERKI/II regulation by the muscarinic acetylcholine receptors in neurons. J Neurosci 20(3):977–985 English
Selles MC, Oliveira MM, Ferreira ST (2018) Brain Inflammation Connects Cognitive and Non-Cognitive Symptoms in Alzheimer’s Disease. J Alzheimers Dis
Tancredi V, D’Antuono M, Cafe C, Giovedi S, Bue MC, D’Arcangelo G, Onofri F, Benfenati F (2000) The inhibitory effects of interleukin-6 on synaptic plasticity in the rat hippocampus are associated with an inhibition of mitogen-activated protein kinase ERK. J Neurochem 75(2):634–643 English
Tyler WJ, Alonso M, Bramham CR, Pozzo-Miller LD (2002) From acquisition to consolidation: On the role of brain-derived neurotrophic factor signaling in hippocampal-dependent learning. Learn Memory 9(5):224–237 English
Um MY, Lim DW, Son HJ, Cho S, Lee C (2018) Phlorotannin-rich fraction from Ishige foliacea brown seaweed prevents the scopolamine-induced memory impairment via regulation of ERK-CREB-BDNF pathway. J Funct Foods 40:110–116
Weinreb O, Amit T, Bar-Am O, Youdim MB (2016) Neuroprotective effects of multifaceted hybrid agents targeting MAO, cholinesterase, iron and beta-amyloid in ageing and Alzheimer’s disease. Br J Pharmacol 173(13):2080–2094 English
Wojsiat J, Zoltowska KM, Laskowska-Kaszub K, Wojda U (2018) Oxidant/Antioxidant Imbalance in Alzheimer’s Disease: Therapeutic and Diagnostic Prospects. Oxid Med Cell Longev. English
Wu X, Kosaraju J, Zhou W, Tam KY (2018) SLOH, a carbazole-based fluorophore, mitigates neuropathology and behavioral impairment in the triple-transgenic mouse model of Alzheimer’s disease. Neuropharmacology 131:351–363
Wu X, Tang B, Liao X, Su Z, Lee SM, Cai Y, Li C (2019) Suppressive effects of the supercritical-carbon dioxide fluid extract of Chrysanthemum indicum on chronic unpredictable mild stress-induced depressive-like behavior in mice. Food Funct 10(2):1212–1224
Xanthos DN, Sandkuhler J (2014) Neurogenic neuroinflammation: inflammatory CNS reactions in response to neuronal activity. Nat Rev Neurosci 15(1):43–53 English
Xian YF, Ip SP, Mao QQ, Su ZR, Chen JN, Lai XP, Lin ZX (2015) Honokiol improves learning and memory impairments induced by scopolamine in mice. Eur J Pharmacol 760:88–95
Acknowledgements
This work is supported in part by the National Natural Science Foundation of China (82003615), the National Key R&D Program of China (2018YFA0800603), the Key Research and Development Program of Guangdong Province for “Innovative drug creation” (2019B020201015), and the Guangdong Innovative Research Team Program (2016ZT06Y432).
Author information
Authors and Affiliations
Contributions
Designed the study: Bingliang Lian and Xiaoli Wu. Performed the experiments:Bingliang Lian, Jingwen Gu, Chen Zhang, Zhicong Zou and Meng Yu. Data collection and Statistical analyses: Bingliang Lian, Jingwen Gu, Chen Zhang, Zhicong Zou and Meng Yu. Wrote the main manuscript text: Bingliang Lian and Xiaoli Wu. Revised the main manuscript text: Fanghong Li and Allan Zijian Zhao. All authors reviewed and approved the manuscript.
Corresponding authors
Ethics declarations
Disclosure statement
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lian, B., Gu, J., Zhang, C. et al. Protective effects of isofraxidin against scopolamine-induced cognitive and memory impairments in mice involve modulation of the BDNF-CREB-ERK signaling pathway. Metab Brain Dis 37, 2751–2762 (2022). https://doi.org/10.1007/s11011-022-00980-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-00980-z