Abstract
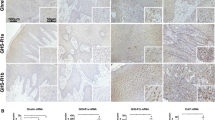
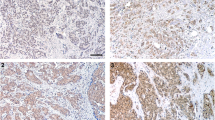
The underlying molecular mechanism of carcinogenesis in oral squamous cell carcinoma (OSCC) is poorly understood and appears to be controlled on many genetic, environmental, and hormonal factors. Obestatin and ghrelin, two recently discovered hormones, are co-expressed in endocrine cells. The purpose of this investigation was to examine the immunohistochemical features of OSCCs in relation to the tissue concentration of ghrelin and obestatin. The association between OSCC and Epstein Barr Virus (EBV) status was also explored. The expression of ghrelin and obestatin was examined by immunohistochemistry and immunoassay in oral biopsy specimens: 10 benign squamous epithelial cell samples, 10 microinvasive squamous cell carcinomas, and seven well-differentiated and seven poorly differentiated OSCCs. The presence of EBV was evaluated in these samples using immunohistochemistry. The concentrations of ghrelin and obestatin in tissue homogenates were measured by RIA and ELISA, respectively. Squamous cell carcinomas and benign tissue samples were positive for anti-EBV antibody, and obestatin and ghrelin were shown to be co-expressed in all stratified squamous epithelium samples. Expression of ghrelin and obestatin was decreased or absent in OSCCs in relation to the invasiveness of the carcinoma; ghrelin and obestatin levels in cancerous tissue homogenates were lower than in benign tissue homogenates. These results indicate that the concentrations and distribution of immunoreactive obestatin and ghrelin might be helpful in distinguishing OSCC from benign tumors. Maintaining normal levels of these hormones might be required for regulation of normal cell division. However, detailed studies will be required for better understanding of the complex mechanism of carcinogenesis relating to OSCCs.



Similar content being viewed by others
References
Patel MM, Pandya AN (2004) Relationship of oral cancer with age, sex, site distribution and habits. Indian J Pathol Microbiol 47:195–197
Talacko AA, Teo CG, Griffin BE, Johnson NW (1991) Epstein-Barr virus receptors but not viral DNA are present in normal and malignant oral epithelium. J Oral Pathol Med 20:20–25
Shamaa AA, Zyada MM, Wagner M, Awad SS, Osman MM, Azeem AA (2008) The significance of Epstein Barr Virus (EBV) & DNA Topoisomerase II alpha (DNA-Topo II alpha) immunoreactivity in normal oral mucosa, Oral Epithelial Dysplasia (OED) and Oral Squamous Cell Carcinoma (OSCC). Diagn Pathol 3:45
Mishra R, Das BR (2005) Activation of STAT 5-cyclin D1 pathway in chewing tobacco mediated oral squamous cell carcinoma. Mol Biol Rep 32:159–166
Aydin S (2007) Discovery of ghrelin hormone: research and clinical applications. Turk J Biochem 32:76–89
Chris JPA, Mark R (2008) Biochemistry of ghrelin precursor peptides. In: Gerald L (ed) Ghrelin vitamins and hormones, 1st edn. Academic Press, London, pp 13–30
Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K (1999) Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 402:656–659
Dagli AF, Aydin S, Karaoglu A, Akpolat N, Ozercan IH, Ozercan MR (2009) Ghrelin expression in normal kidney tissue and renal carcinomas. Pathol Res Pract 205:165–173
Jeffery PL, Murray RE, Yeh AH, McNamara JF, Duncan RP, Francis GD, Herington AC, Chopin LK (2005) Expression and function of the ghrelin axis, including a novel preproghrelin isoform, in human breast cancer tissues and cell lines. Endocr Relat Cancer 12:839–850
Murata M, Okimura Y, Iida K, Matsumoto M, Sowa H, Kaji H, Kojima M, Kangawa K, Chihara K (2002) Ghrelin modulates the downstream molecules of insulin signaling in hepatoma cells. J Biol Chem 277:5667–5674
Aydin S, Ozercan IH, Dagli F, Aydin S, Dogru O, Celebi S, Akin O, Guzel SP (2005) Ghrelin immunohistochemistry of gastric adenocarcinoma and mucoepidermoid carcinoma of salivary gland. Biotech Histochem 80:163–168
Mottershead M, Karteris E, Barclay JY, Suortamo S, Newbold M, Randeva H, Nwokolo CU (2007) Immunohistochemical and quantitative mRNA assessment of ghrelin expression in gastric and oesophageal adenocarcinoma. J Clin Pathol 60:405–409
Zhang JV, Ren PG, Avsian-Kretchmer O, Luo CW, Rauch R, Klein C, Hsueh AJ (2005) Obestatin, a peptide encoded by the ghrelin gene, opposes ghrelin’s effects on food intake. Science 310:996–999
Veldhorst M, Smeets A, Soenen S, Hochstenbach-Waelen A, Hursel R, Diepvens K, Lejeune M, Luscombe-Marsh N, Westerterp-Plantenga M (2008) Protein-induced satiety: effects and mechanisms of different proteins. Physiol Behav 94:300–307
Ozbay Y, Aydin S, Dagli AF, Akbulut M, Dagli N, Kilic N, Rahman A, Sahin I, Polat V, Ozercan HI, Arslan N, Sensoy D (2008) Obestatin is present in saliva: alterations in obestatin and ghrelin levels of saliva and serum in ischemic heart disease. BMB Rep 41:55–61
Aydin S, Ozkan Y, Erman F, Gurates B, Kilic N, Colak R, Gundogan T, Catak Z, Bozkurt M, Akin O, Sen Y, Sahin I (2008) Presence of obestatin in breast milk: relationship among obestatin, ghrelin, and leptin in lactating women. Nutrition 24:689–693
Tang SQ, Jiang QY, Zhang YL, Zhu XT, Shu G, Gao P, Feng DY, Wang XQ, Dong XY (2008) Obestatin: its physicochemical characteristics and physiological functions. Peptides 29:639–645
Ren AJ, Guo ZF, Wang YK, Lin L, Zheng X, Yuan WJ (2009) Obestatin, obesity and diabetes. Peptides 30:439–444
Barnes L, Evenson JW, Reichart P, Sidransky D (2005) World Health Organization classification of tumors pathology and genetics. Head and neck tumors. IARC Press, Lyon
Hsu SM, Raine L, Fanger H (1981) Use of avidin–biotin-peroxidase complex (ABC) in immunoperoxidase technique. J Histochem Cytochem 29:577–580
Hosoda H, Doi K, Nagaya N, Okumura H, Nakagawa E, Enomoto M, Ono F, Kangawa K (2004) Optimum collection and storage conditions for ghrelin measurements: octanoyl modification of ghrelin is rapidly hydrolyzed to desacyl ghrelin in blood samples. Clin Chem 50:1077–1080
Cruz I, Van den Brule AJ, Steenbergen RD, Snijders PJ, Meijer CJ, Walboomers JM, Snow GB, Van der Waal I (1997) Prevalence of Epstein-Barr virus in oral squamous cell carcinomas, premalignant lesions and normal mucosa—a study using the polymerase chain reaction. Oral Oncol 33:182–188
Mao EJ, Smith CJ (1993) Detection of Epstein-Barr virus (EBV) DNA by the polymerase chain reaction (PCR) in oral smears from healthy individuals and patients with squamous cell carcinoma. J Oral Pathol Med 22:12–17
Greenspan JS, Greenspan D, Lennette ET, Abrams DI, Conant MA, Petersen V, Freese UK (1985) Replication of Epstein-Barr virus within the epithelial cells of oral “hairy” leukoplakia, an AIDS-associated lesion. N Engl J Med 313:1564–1571
Horiuchi K, Mishima K, Ichijima K, Sugimura M, Ishida T, Kirita T (1995) Epstein-Barr virus in the proliferative diseases of squamous epithelium in the oral cavity. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 79:57–63
Sand LP, Jalouli J, Larsson PA, Hirsch JM (2002) Prevalence of Epstein-Barr virus in oral squamous cell carcinoma, oral lichen planus, and normal oral mucosa. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 93:586–592
Karaoglu A, Aydin S, Dagli AF, Cummings DE, Ozercan IH, Canatan H, Ozkan Y (2009) Expression of obestatin and ghrelin in papillary thyroid carcinoma. Mol Cell Biochem 323:113–118
Raffel A, Krausch M, Cupisti K, Gerharz CD, Eisenberger CF, Knoefel WT (2005) Ghrelin expression in neuroendocrine tumors of the gastrointestinal tract with multiple endocrine neoplasia type 1. Horm Metab Res 37:653–655
Tsolakis AV, Grimelius L, Stridsberg M, Falkmer SE, Waldum HL, Saras J, Janson ET (2009) Obestatin/ghrelin cells in normal mucosa and endocrine tumours of the stomach. Eur J Endocrinol 160:941–949
Conconi MT, Nico B, Guidolin D, Baiguera S, Spinazzi R, Rebuffat P (2004) Ghrelin inhibits FGF-2-mediated angiogenesis in vitro and in vivo. Peptides 25:2179–2185
Baiguera S, Conconi MT, Guidolin D, Mazzocchi G, Malendowicz LK, Parnigotto PP (2004) Ghrelin inhibits in vitro angiogenic activity of rat brain microvascular endothelial cells. Int J Mol Med 14:849–854
Haklar G, Sayin-Ozveri E, Yüksel M, Aktan AO, Yalçin AS (2001) Different kinds of reactive oxygen and nitrogen species were detected in colon and breast tumors. Cancer Lett 165:219–224
Thompson NM, Gill DA, Davies R, Loveridge N, Houston PA, Robinson IC, Wells T (2004) Ghrelin and des-octanoyl ghrelin promote adipogenesis directly in vivo by a mechanism independent of the type 1a growth hormone secretagogue receptor. Endocrinology 145:234–242
Sato M, Nakahara K, Goto S, Kaiya H, Miyazato M, Date Y, Nakazato M, Kangawa K, Murakami N (2006) Effects of ghrelin and des-acyl ghrelin on neurogenesis of the rat fetal spinal cord. Biochem Biophys Res Commun 350:598–603
Granata R, Settanni F, Biancone L, Trovato L, Nano R, Bertuzzi F, Destefanis S, Annunziata M, Martinetti M, Catapano F, Ghè C, Isgaard J, Papotti M, Ghigo E, Muccioli G (2007) Acylated and unacylated ghrelin promote proliferation and inhibit apoptosis of pancreatic beta-cells and human islets: involvement of 3′, 5′-cyclic adenosine monophosphate/protein kinase A, extracellular signal-regulated kinase 1/2, and phosphatidyl inositol 3-Kinase/Akt signaling. Endocrinology 148:512–529
Baldanzi G, Filigheddu N, Cutrupi S, Catapano F, Bonissoni S, Fubini A, Malan D, Baj G, Granata R, Broglio F, Papotti M, Surico N, Bussolino F, Isgaard J, Deghenghi R, Sinigaglia F, Prat M, Muccioli G, Ghigo E, Graziani A (2002) Ghrelin and des-acyl ghrelin inhibit cell death in cardiomyocytes and endothelial cells through ERK1/2 and PI 3-kinase/AKT. J Cell Biol 159:1029–1037
Granado M, Martín AI, López-Menduiña M, López-Calderón A, Villanúa MA (2008) GH-releasing peptide-2 administration prevents liver inflammatory response in endotoxemia. Am J Physiol Endocrinol Metab 294:E131–E141
El Eter E, Al Tuwaijiri A, Hagar H, Arafa M (2007) In vivo and in vitro antioxidant activity of ghrelin: Attenuation of gastric ischemic injury in the rat. J Gastroenterol Hepatol 22:1791–1799
Andrews ZB, Liu ZW, Walllingford N, Erion DM, Borok E, Friedman JM, Tschöp MH, Shanabrough M, Cline G, Shulman GI, Coppola A, Gao XB, Horvath TL, Diano S (2008) UCP2 mediates ghrelin’s action on NPY/AgRP neurons by lowering free radicals. Nature 454:846–851
Hou Y, An J, Hu XR, Sun BB, Lin J, Xu D (2009) Ghrelin inhibits interleukin-8 production induced by hydrogen peroxide in A549 cells via NF-kappa B pathway. Int Immunopharmacol 9:120–126
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alnema, M.M., Aydin, S., Ozkan, Y. et al. Ghrelin and obestatin expression in oral squamous cell carcinoma: an immunohistochemical and biochemical study. Mol Cell Biochem 339, 173–179 (2010). https://doi.org/10.1007/s11010-009-0381-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-009-0381-1