Abstract
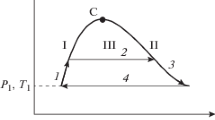
The (T 1−T 2)/T 1 efficiency of a very small temperature differential thermodynamic cycle operating near a solvent’s critical locus, where T 1 is the higher temperature and T 2 the lower, is shown to be a simple tool for determining the maximum difference in the excess heat of solution between the cycle’s low-temperature liquid side and its high-temperature supercritical fluid side.
Similar content being viewed by others
References
REFPROP, Version 7, National Institute of Standards and Technology, Boulder, Colorado (2002)
Zhao, S.Q., Wang, R.N., Yang, G.H.: A method for measuring solid solubility in supercritical carbon dioxide. J. Supercrit. Fluids 8, 15–19 (1995)
Young, H.D.: Fundamentals of Mechanics and Heat. McGraw-Hill, New York (1964), p. 540
Marceca, E., Fernández-Prini, R.: Solubility of solids in near-critical fluids. II. CHI3 and CI4 in ethane. J. Chem. Thermodyn. 25, 237–247 (1993)
Schofield, R.S., Post, M.E., McFall, T.A., Izatt, R.M., Christensen, J.J.: The excess enthalpies of liquid (dichlorodifluoromethane + N,N-dimethylacetamide) from 263.15 to 413.15 K and 4.33 to 4.53 MPa. J. Chem. Thermodyn. 15, 217–224 (1983)
Christensen, J.J., Zebolsky, D.M., Schofield, R.S., Cordray, D.R., Izatt, R.M.: The excess enthalpies of (ethane + chlorodifluoromethane) from 293.15 to 383.15 K at 5.15 MPa. J. Chem. Thermodyn. 16, 905–916 (1984)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Van Den Einde, D. Determining the Maximum Excess Heat of Solution Differentials Using the Carnot Cycle Temperature Efficiency. J Solution Chem 36, 1073–1077 (2007). https://doi.org/10.1007/s10953-007-9174-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-007-9174-x