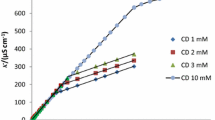
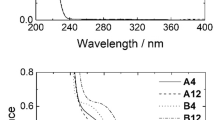
Abstract
As a continuation of our previous investigation, interactions between cyclodextrin (β-CD), γ-cyclodextrin (γ-CD) and alkyl trimethylammonium bromides in aqueous solutions have been studied with titration calorimetry and 1H NMR at 298.15 K. The results are discussed in terms of the amphiphilic interaction of CD with surfactants and the iceberg structure formed by water molecules existing around the hydrophobic tail of surfactant molecules. The stoichiometry of the β-CD–surfactant system is 1:1 whereas that of the γ-CD–surfactant system is 1:2. The corresponding formation enthalpy (negative) of the complexes of the two systems decreases with an increase in the number of carbon atoms (n) in hydrophobic chain of surfactant molecule, C n H2n+1, whereas the entropy increases with the enlargement of n.
Similar content being viewed by others
References
Eastburn, S.D., Tao, B.Y.: Applications of modified cyclodextrins. Biotechnol. Adv. 12, 325–339 (1994)
Jesus, M.B., Pinto, L.M.A., Fraceto, L.F., Takahata, Y., Lino, A.C.S., Jaime, C., Paula, E.: Theoretical and experimental study of a praziquantel and β-cyclodextrin inclusion complex using molecular mechanic calculations and 1H-nuclear magnetic resonance. J. Pharm. Biomed. Anal. 41, 1428–1432 (2006)
Zhang, H.-R., Chen, G., Wang, L., Ding, L., Tian, Y., Jin, W.-Q., Zhang, H.-Q.: Study on the inclusion complexes of cyclodextrin and sulphonated azo dyes by electrospray ionization mass spectrometry. Int. J. Mass Spectrom. 252, 1–10 (2006)
Illapakurthy, A.C., Wyandt, C.M., Stodghill, S.P.: Isothermal titration calorimetry method for determination of cyclodextrin complexation thermodynamics between artemisinin and naproxen under varying environmental conditions. Eur. J. Pharm. Biopharm. 59, 325–332 (2005)
Martín, L., León, A., Olives, A.I., Castillo, B., Marín, M.A.: Spectrofluorimetric determination of stoichiometry and association constants of the complexes of harmane and harmine with β-cyclodextrin and chemically modified β-cyclodextrins. Talanta 60, 493–503 (2003)
Chan, J., Sun, D.-Z., Feng, C.-G.: Spectrophotometric studies on the inclusion formation of β-cyclodextrin with 3,4-(methylenedioxyphenyl) benzylideneaniline. J. Solution Chem. 31, 211–222 (2002)
Guo, M.-Q., Zhang, S.-Q., Song, F.-R., Wang, D.-W., Liu, Z.-Q., Liu, S.-Y.: Studies on the non-covalent complexes between oleanolic acid and cyclodextrins using electrospray ionization tandem mass spectrometry. J. Mass Spectrom. 38, 723–731 (2003)
Carlos, B.D., Elisa, G.R.: Monitoring dediazoniation product formation by high-performance liquid chromatography after derivatization. J. Chromatogr. A 989, 221–229 (2003)
Nathalie, M., Philippe, M., Ribet, J.P.: Spectrofluorimetric study of eflucimibe–γ-cyclodextrin inclusion complex. Eur. J. Pharm. Biopharm. 59, 523–526 (2005)
Pacioni, N.L., Veglia, A.V.: Determination of carbaryl and carbofuran in fruits and tap water by β-cyclodextrin enhanced fluorimetric method. Anal. Chim. Acta 488, 193–202 (2003)
Manzoori, J.L., Amjiadi, M.: Spectrofluorimetric study of host–guest complexation of ibuprofen with β-cyclodextrin and its analytical application. Spectrochim. Acta A: Mol. Biomol. Spectrosc. 59, 909–916 (2003)
Spencer, J.N., He, Q., Ke, X.-M., Wu, Z.-Q., Fetter, E.: Complexation of inorganic anions and small organic molecules with alpha-cyclodextrin in water. J. Solution Chem. 27, 1009–1019 (1998)
Bernini, A., Spiga, O., Ciutti, A., Scarselli, M., Bottoni, G., Mascagni, G., Niccolai, N.: NMR studies of the inclusion complex between β-cyclodextrin and paroxetine. Eur. J. Pharm. Sci. 22, 445–450 (2004)
Srinivasan, K., Bartlett, M.G.: Analysis of methylphenidate and its metabolite ritalinic acid in monkey plasma by liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 14, 619–923 (2000)
Sun, D.-Z., Li, L., Qiu, X.-M., Liu, F., Yin, B.-L.: Isothermal titration calorimetry and 1H NMR studies on host–guest interaction of paeonol and two of its isomers with β-cyclodextrin. Int. J. Pharm. 316, 7–13 (2006)
Buckton, G., Beezer, A.: A calorimetric study of powder surface energetics. Int. J. Pharm. 41, 139–145 (1988)
Jacobson, A.M., Casassa, Z.: Micropore filling of supercritical NO on Cu-doped iron oxide dispersed activated carbon fibers. J. Colloid. Interface Sci. 142, 489 (1991)
Bai, G.-Y., Wang, Y.-J., Yan, H.-K., Robert, T.K., Kwak, J.C.T.: Thermodynamics of interaction between cationic gemini surfactants and hydrophobically modified polymers in aqueous solutions. J. Phys. Chem. B 106, 2153–2159 (2002)
Gómez-Oreíllana, I., Hallén, D.: The thermodynamics of the binding of the benzene to β-cyclodextrin in aqueous solution. Thermochim. Acta 221, 183–193 (1993)
Sun, D.-Z., Wang, S.-B., Wei, X.-L., Yin, B.-L.: A calorimetric study of β-cyclodextrin with 3-alkoxyl-2-hydroxypropyl trimethylammonium bromides in aqueous solutions. J. Chem. Thermodyn. 37, 431–436 (2005)
Sun, D.-Z., Wang, S.-B., Song, M.-Z., Wei, X.-L., Yin, B.-L.: A calorimetric study of host–guest complexes of α-cyclodextrin with alkyl trimethyl ammonium bromides in aqueous solutions. J. Solution Chem. 34, 701–712 (2005)
Frank, H.S., Evans, M.W.: Free volume and entropy in condensed systems. III. Entropy in binary liquid mixtures; partial molal entropy in dilute solutions; structure and thermodynamics in aqueous electrolytes. J. Chem. Phys. 13, 507–532 (1945)
Szejtli, J.: Introduction and general overview of cyclodextrin chemistry. Chem. Rev. 98, 1743–1754 (1998)
Fernandes, C.M., Caralho, R.A., Pereira da, C.S., Veiga, F.J.B.: Multimodal molecular encapsulation of nicardipine hydrochloride by β-cyclodextrin and triaxetyl–β-cyclodextrin in solution structural studies by 1H NMR and ROESY experiments. Eur. J. Pharm. Sci. 18, 285–296 (2003)
Tong, L.-H.: Cyclodextrin Chemistry—Foundation and Application. Science Press, Beijing (2001)
Ventura, C.A., Puglisi, G., Zappalà, M., Mazzone, G.: A physico-chemical study on the interaction between papaverine and natural and modified β-cyclodextrins. Int. J. Pharm. 160, 163–172 (1998)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qu, XK., Zhu, LY., Li, L. et al. Host–Guest Complexation of β-, γ-Cyclodextrin with Alkyl Trimethyl Ammonium Bromides in Aqueous Solution. J Solution Chem 36, 643–650 (2007). https://doi.org/10.1007/s10953-007-9132-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-007-9132-7