Abstract
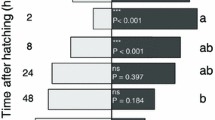
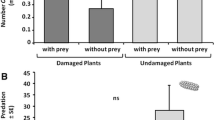
Insectivorous birds feed upon all developmental stages of herbivorous insects, including insect eggs if larvae and adults are unavailable. Insect egg deposition on plants can induce plant traits that are subsequently exploited by egg parasitoids searching for hosts. However, it is unknown whether avian predators can also use egg-induced plant changes for prey localization. Here, we studied whether great tits (Parus major) and blue tits (Cyanistes caeruleus) are attracted by traits of the Scots pine (Pinus sylvestris) induced by pine sawfly (Diprion pini) egg deposition. We chose this plant – insect system because sawfly egg deposition on pine needles is known to locally and systemically induce a change in pine volatile organic compounds (VOCs), and tits are known to prey upon sawfly eggs. In dual choice laboratory experiments, we simultaneously offered the birds an egg-free control branch and a systemically egg-induced branch. Significantly more birds visited the egg-induced branch first. We confirmed by GC-MS analyses that systemically egg-induced branches released more (E)-β-farnesene compared to control branches. Spectrophotometric analyses showed that control branches reflected more light than egg-induced branches throughout the avian visual range. Although a discrimination threshold model for blue tits suggests that the birds are poor at discriminating this visual difference, the role of visual stimuli in attracting the birds to egg-induced pines cannot be discounted. Our study shows, for the first time, that egg-induced odorous and/or visual plant traits can help birds to locate insect eggs without smelling or seeing those eggs.




Similar content being viewed by others
References
Adamik P, Koman M, Vojtek J (2003) The effect of habitat structure on guild patterns and the foraging strategies of insectivorous birds in forests. Biologia 58:275–286
Amo L, Jansen JJ, van Dam NM, Dicke M, Visser ME (2013) Birds exploit herbivore-induced plant volatiles to locate herbivorous prey. Ecol Lett 16:1348–1355. https://doi.org/10.1111/ele.12177
Amo L, Dicke M, Visser ME (2016) Are naïve birds attracted to herbivore-induced plant defences? Behaviour 153:353–366. https://doi.org/10.1163/1568539X-00003345
Anderson P, Anton S (2014) Experience-based modulation of behavioural responses to plant volatiles and other sensory cues in insect herbivores. Plant Cell Environ 37:1826–1835. https://doi.org/10.1111/pce.12342
Barbaro L, Battisti A (2011) Birds as predators of pine processionary moth (Lepidoptera: Notodonidae). Biol Control 56:107–114. https://doi.org/10.1016/j.biocontrol.2010.10.009
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Royal Stat Soc Series B 57:289–300
Bereczki K, Molnár D, Csóka G, Báldi A (2017) Factors affecting the bird predation of low density gypsy moth egg masses in three types of hardwood forests in Southwest Hungary. Bull Insectol 70:201–207
Betts MM (1955) The food of titmice in oak woodland. J Anim Ecol 24:282–323
Beyaert I, Wäschke N, Scholz A, Varama M, Reinecke A, Hilker M (2010) Relevance of resource-indicating key volatiles and habitat odour for insect orientation. Anim Behav 79:1077–1086. https://doi.org/10.1016/j.anbehav.2010.02.001
Biard C, Surai PF, Møller AP (2007) An analysis of pre- and post-hatching maternal effects mediated by carotenoids in the blue tit. Evol Biol 20:326–339. https://doi.org/10.1111/j.1420-9101.2006.01194.x
Bombosch S, Ramakers PMJ (1976) Zur Dauerzucht von Gilpinia hercyniae (Htg). Zeitschr. Pflanzenkrankh Pflanzenschutz 83:40–44
Cusumano A, Weldegergis BT, Colazza S, Dicke M, Fatouros NE (2015) Attraction of egg-killing parasitoids toward induced plant volatiles in a multi-herbivore context. Oecologia 179:163–174. https://doi.org/10.1007/s00442-015-3325-3
Cuthill IC (2006) Colour perception. In: Hill GE, McGraw KJ (eds) Bird Colouration, mechanisms and measurements. Harvard University Press, Cambridge, pp 3–40
D’Alessandro M, Turlings TCJ (2006) Advances and challenges in the identification of volatiles that mediate interactions among plants and arthropods. Analyst 131:24–32. https://doi.org/10.1039/B507589K
Dicke M (2009) Behavioural and community ecology of plants that cry for help. Plant Cell Environ 32:654–665. https://doi.org/10.1111/j.1365-3040.2008.01913.x
Dicke M, Baldwin IT (2010) The evolutionary context for herbivore-induced plant volatiles: beyond the ‘cry for help. Trends Plant Sci 15:167–175. https://doi.org/10.1016/j.tplants.2009.12.002
Dudoit S, Shaffer JP, Boldrick JC (2003) Multiple hypothesis testing in microarray experiments. Stat Sci 18:71–103
Eisner T, Johnessee JS, Carrel J, Hendry LB, Meinwald J (1974) Defensive use by an insect of a plant resin. Science 184:996–999
Gibb J (1957) Food requirement and other observations on captive tits. Bird Study 4:207–215
Hart NS, Partridge JC, Cuthill IC (2000) Retinal asymmetry in birds. Curr Biol 10:115–117. https://doi.org/10.1016/S0960-9822(00)00297-9
Haynes KJ, Allstadt AJ, Klimetzek D (2014) Forest defoliator outbreaks under climate change: effects on the frequency and severity of outbreaks of five pine insect pests. Glob Chang Biol 20:2004–2018. https://doi.org/10.1111/gcb.12506
Helms JA, Godfray AP, Ames T, Bridge ES (2016) Predator foraging altitudes reveal the structure of aerial insect communities. Sci Rep 6:28670. https://doi.org/10.1038/srep28670
Hilker M, Fatouros NE (2015) Plant responses to insect egg deposition. Annu Rev Entomol 60:493–515. https://doi.org/10.1146/annurev-ento-010814-020620
Hilker M, Meiners T (2002) Chemoecology of insect eggs and egg deposition. An introduction. In: Hilker M, Meiners T (eds) Chemoecology of insect eggs and egg deposition. Blackwell, Berlin, pp XV–XX. https://doi.org/10.1002/9780470760253
Hilker M, Meiners T (2006) Early herbivore alert: insect eggs induce plant defense. J Chem Ecol 32:1379–1397. https://doi.org/10.1007/s10886-006-9057-4
Hilker M, Meiners T (2011) Plants and insect eggs: how do they affect each other? Phytochemistry 72:1612–1623. https://doi.org/10.1016/j.phytochem.2011.02.018
Hilker M, Kobs C, Varama M, Schrank K (2002) Insect egg deposition induces Pinus sylvestris to attract egg parasitoids. J Exp Biol 205:455–461
Hilker M, Stein C, Schröder R, Varama M, Mumm R (2005) Insect egg deposition induces defence responses in Pinus sylvestris: characterisation of the elicitor. J Exp Biol 208:1849–1854
Holopainen JK, Gershenzon J (2010) Multiple stress factors and the emission of plant VOCs. Trends Plant Sci 15:176–184. https://doi.org/10.1016/j.tplants.2010.01.006
Izumi S, Yano K, Yamamoto Y, Susumu Y, Takahashi SY (1994) Yolk proteins from insect eggs: structure, biosynthesis and programmed degradation during embryogenesis. J Insect Physiol 40:735–746. https://doi.org/10.1016/0022-1910(94)90001-9
Karban R, Baldwin IT (1997) Induced responses to herbivory. University Chicago Press, Chicago
König WA, Joulain D, Hochmuth DH (2006) GC/MS Library: Terpenoids and Related Constituents of Essential Oils. Retrieved from http://massfinder.com/wiki/Terpenoids_Library (Accessed 18 June 2014)
Koski T-M, Lindstedt C, Klemola T, Troscianko J, Mäntylä E, Tyystjärvi E, Stevens M, Helander M, Laaksonen T (2017) Insect herbivory may cause changes in the visual properties of leaves and affect the camouflage of herbivores to avian predators. Behav Ecol Sociobiol 71:97. https://doi.org/10.1007/s00265-017-2326-0
Kouřimská L, Adámková A (2016) Nutritional and sensory quality of edible insects. NFS Journal 4:22–36. https://doi.org/10.1016/j.nfs.2016.07.001
Lind O, Mitkus M, Olsson P, Kelber A (2014) Ultraviolet vision in birds: the importance of transparent eye media. Proc R Soc B Biol Sci 281:20132209. https://doi.org/10.1098/rspb.2013.2209
Lindstedt C, Huttunen H, Kakko M, Mappes J (2011) Disengtangling the evolution of weak warning signals: high detection risk and low production costs of chemical defences in gregarious pine sawfly larvae. Evol Ecol 25:1029–1046. https://doi.org/10.1007/s10682-010-9456-4
Mäntylä E, Klemola T, Haukioja E (2004) Attraction of willow warblers to sawfly-damaged mountain birches: novel function of inducible plant defences? Ecol Lett 7:915–918. https://doi.org/10.1111/j.1461-0248.2004.00653.x
Mäntylä E, Alessio GA, Blande JD, Heijari J, Holopainen JK, Laaksonen T, Piirtola P, Klemola T (2008a) From plants to birds: higher avian predation rates in trees responding to insect herbivory. PLoS One 3(7):e2832. https://doi.org/10.1371/journal.pone.0002832
Mäntylä E, Klemola T, Sirkiä P, Laaksonen T (2008b) Low light reflectance may explain the attraction of birds to defoliated trees. Behav Ecol 19:325–330. https://doi.org/10.1093/beheco/arm135
Mäntylä E, Klemola T, Laaksonen T (2011) Birds help plants: a meta-analysis of top-down trophic cascades caused by avian predators. Oecologia 165:143–151. https://doi.org/10.1007/s00442-010-1774-2
Mäntylä E, Blande JD, Klemola T (2014) Do birches treated with methyl jasmonate attract insectivorous birds in the nature? Arthropod-Plant Interact 8:143–153. https://doi.org/10.1007/s11829-014-9296-1
Mäntylä E, Kleier S, Kipper S, Hilker M (2017) The attraction of insectivorous tit species to herbivore-damaged scots pines. J Ornith 158:479–491. https://doi.org/10.1007/s10336-016-1412-9
Mennerat A, Bonadonna F, Perret P, Lambrechts MM (2005) Olfactory conditioning experiments in a food-searching passerine bird in semi-natural conditions. Behav Process 70:264–270. https://doi.org/10.1016/j.beproc.2005.07.005
Mooney KA, Gruner DS, Barber NA, Van Bael SA, Philpott SM, Greenberg R (2010) Interactions among predators and the cascading effects of vertebrate insectivores on arthropod communities and plants. Proc Natl Acad Sci U S A 107:7335–7340. https://doi.org/10.1073/pnas.1001934107
Morris D (1956) The feather postures of birds and the problem of the origin of social signals. Behaviour 9:75–111
Mrazova A, Sam K (2017) Application of methyl jasmonate to grey willow (Salix cinerea) attracts insectivorous birds in nature. Arthropod-Plant Interact 12:1–8. https://doi.org/10.1007/s11829-017-9558-9
Mumm R, Hilker M (2006) Direct and indirect chemical defence of pine against folivorous insects. Trends Plant Sci 11:351–358. https://doi.org/10.1016/j.tplants.2006.05.007
Mumm R, Schrank K, Wegener R, Schulz S, Hilker M (2003) Chemical analysis of volatiles emitted by Pinus sylvestris after induction by insect oviposition. J Chem Ecol 29:1235–1252. https://doi.org/10.1023/A:1023841909199
Naef-Daenzer B, Keller LF (1999) The foraging performance of great and blue tits (Parus major and P. caeruleus) in relation to caterpillar development, and its consequences for nestling growth and fledging weight. J Anim Ecol 68:708–718. https://doi.org/10.1046/j.1365-2656.1999.00318.x
Olofsson E (1986) Winter feeding by the great tit, Parus major, on eggs of the European pine sawfly, Neodiprion sertifer. Ornis Fennica 63:91–92
Osorio D, Vorobyev M (2005) Photoreceptor spectral sensitivities in terrestrial animals: adaptations for luminance and colour vision. Proc R Soc B Biol Sci 272:1745–1752. https://doi.org/10.1098/rspb.2005.3156
Perrins CM (1991) Tits and their caterpillar food supply. Ibis 133:49–54
Sam K, Koane B, Novotny V (2015) Herbivore damage increases avian and ant predation of caterpillars on trees along a complete elevational forest gradient in Papua New Guinea. Ecography 38:293–300. https://doi.org/10.1111/ecog.00979
Schröder R, Forstreuter M, Hilker M (2005) A plant notices insect egg deposition and changes its rate of photosynthesis. Plant Physiol 138:470–477. https://doi.org/10.1104/pp.105.059915
Sipura M (1999) Tritrophic interactions: willows, herbivorous insects and insectivorous birds. Oecologia 121:537–545. https://doi.org/10.1007/s004420050960
Sneddon H, Hadden R, Hepper PG (1998) Chemosensory learning in the chicken embryo. Physiol Behav 64:133–139. https://doi.org/10.1016/S0031-9384(98)00037-7
Stein SE (2011) Retention indices. In: Linstrom PJ, Mallard WG (eds) NIST Chemistry WebBook, NIST Standard Reference Database Number 69. National Institute of Standards and Technology, Gaithersburg, retrieved from http://webbook.nist.gov/
Turlings TCJ, Tumlinson JH (1992) Systemic release of chemical signals by herbivore-injured corn. Proc Natl Acad Sci U S A 89:8399–8402
Van Bael SA, Philpott SM, Greenberg R, Bichier P, Barber NA, Mooney KA, Gruner DS (2008) Birds as predators in tropical agroforestry systems. Ecology 89:928–934. https://doi.org/10.1890/06-1976.1
van den Dool H, Kratz PD (1963) A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J Chromatogr 11:463–471
Velikova V, Salerno G, Frati F, Peri E, Conti E, Colazza S, Loreto F (2010) Influence of feeding and oviposition by phytophagous pentatomids on photosynthesis of herbaceous plants. J Chem Ecol 36:629–641. https://doi.org/10.1007/s10886-010-9801-7
Vet LEM, Dicke M (1992) Ecology of infochemical use by natural enemies in a tritrophic context. Annu Rev Entomol 37:141–172. https://doi.org/10.1146/annurev.en.37.010192.001041
Vorobyev M, Osorio D (1998) Receptor noise as a determinant of colour thresholds. Proc R Soc B Biol Sci 265:351–358. https://doi.org/10.1098/rspb.1998.0302
Vorobyev M, Osorio D, Bennett ATD, Marshall NJ, Cuthill IC (1998) Tetrachromacy, oil droplets and bird plumage colours. J Comp Physiol A 183:621–633. https://doi.org/10.1007/s003590050286
Wheelan CJ (2001) Foliage structure influences foraging of insectivorous birds: an experimental study. Ecology 82:219–231. https://doi.org/10.1890/0012-9658(2001)082[0219:FSIFOI]2.0.CO;2
Zangerl AR, Hamilton JG, Miller TJ, Crofts AR, Oxborough K, Berenbaum MR, de Lucia EH (2002) Impact of folivory on photosynthesis is greater than the sum of its holes. Proc Natl Acad Sci U S A 99:1088–1091. https://doi.org/10.1073/pnas.022647099
Zhou S, Lou Y-R, Tzin V, Jander G (2015) Alteration of plant primary metabolism in response to insect herbivory. Plant Physiol 169:1488–1498. https://doi.org/10.1104/pp.15.01405
Ziegler R (1997) Lipid synthesis by ovaries and fat bodies of Aedes aegypti (Diptera: Culicidae). Eur J Entomol 94:385–391
Acknowledgements
We thank Ute Braun, Freie Universität Berlin, for her help in rearing the sawflies and collecting pine branches. We also thank Prof. Dr. Holger Dau and Dr. Ivelina Zaharieva from the Physics Department, Freie Universität Berlin, for assistance with the spectrophotometer. The study was supported financially by the Finnish Cultural Foundation (grant to EM), the ERC grant no 669609 (EM) and the Academy of Finland via the project no 257581 (CL).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The birds were studied with a license from Landesamt für Gesundheit und Soziales, Berlin (no. 0149/12), and ringed with a license from Vogelwarte Radolfzell (no. 1882). The experimental procedure never caused damage or signs of severe distress to the birds. All birds were released back into the wild, close to their place of capture, immediately after the experiment. The time in captivity for each bird was 40.0 (31.0, 54.0) minutes [median (lower quartile, upper quartile)].
Rights and permissions
About this article
Cite this article
Mäntylä, E., Kleier, S., Lindstedt, C. et al. Insectivorous Birds Are Attracted by Plant Traits Induced by Insect Egg Deposition. J Chem Ecol 44, 1127–1138 (2018). https://doi.org/10.1007/s10886-018-1034-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-018-1034-1