Abstract
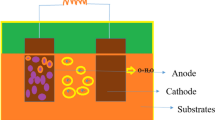
The organic matter consumption and hydrogen production rate were evaluated in a two-chamber microbial electrolysis cell (MEC). Three chemical oxygen demand (COD) concentration levels (400, 600 and 1200 mg/L) were tested. The COD was composed of a mixture of volatile fatty acids (VFAs) present in the effluent of a dark fermentation process. The two levels of voltage studied were 350 and 550 mV. The performance of the MEC was evaluated using either an anionic (AEM) or cationic exchange membrane (CEM). The robustness of the MEC was tested using two dark fermentation effluents, one with VFAs and another containing 1100 mg/L glucose. The highest production rates (81 mL/L/day) were obtained with 550 mV, and 85 % COD consumption was attained. No considerable differences in the hydrogen production rate were observed when the COD was increased from 400 to 1200 mg/L using 550 mV. However, maximal hydrogen production rates were obtained with the lower COD concentration using 350 mV. Neither the employment of AEM and CEM nor the change from synthetic substrate to real substrate resulted in remarkable changes in MEC performance. The substrate containing glucose was more slowly degraded because glucose was first transformed into VFAs, and then the VFAs were consumed to produce hydrogen. In this case, methane and carbon dioxide were detected.





Similar content being viewed by others
References
Show KY, Lee DJ, Tay JH, Lin CY, Chang JS (2012) Biohydrogen production: current perspectives and the way forward. Int J Hydrog Energy 30:1–16. doi:10.1016/j.ijhydene.2012.04.109
Lee HS, Vermaas WFJ, Rittmann BE (2010) Biological hydrogen production: prospects and challenges. Trends Biotechnol 28:262–271. doi:10.1016/j.tibtech.2010.01.007
Sleutels THJA, Ter Heijne A, Buisman CJN, Hamelers HVM (2012) Bioelectrochemical systems: an outlook for practical applications. ChemSusChem 5:1012–1019. doi:10.1002/cssc.201100732
Wrana N, Sparling R, Cicek N, Levin DB (2010) Hydrogen gas production in a microbial electrolysis cell by electrohydrogenesis. J Clean Prod 18:S105–S111. doi:10.1016/j.jclepro.2010.06.018
Pant D, Singh A, Van Bogaert G, Olsen SI, Nigam PS, Diels L, Vanbroekhoven K (2012) Bioelectrochemical systems (BES) for sustainable energy production product recovery from organic wastes and industrial wastewaters. RSC Adv 2:1248–1263. doi:10.1039/C1RA00839K
Logan BE, Call D, Cheng S, Hamelers HVM, Sleutels THJA, Jeremiasse AW, Rozendal RA (2008) Microbial electrolysis cells for high yield hydrogen gas production from organic matter. Environ Sci Technol 42:8639–8640. doi:10.1021/es801553z
Harnisch F, Schröder U (2010) From MFC to MXC: chemical and biological cathodes and their potential for microbial bioelectrochemical systems. Chem Soc Rev 39:4433–4448. doi:10.1039/C003068F
Logan BE (2007) Microbial fuel cells. Willey, Hoboken, p 127
Lovley D, Phillips E (1988) Novel mode of microbial energy metabolism: organic carbon coupled to dissimilatory reduction of iron or manganese. Appl Environ Microbiol 54:1472–1480
Buitrón G, Carvajal C (2010) Biohydrogen production from tequila vinasses in an anaerobic sequencing batch reactor: effect of initial substrate concentration, temperature and hydraulic retention time. Bioresour Technol 101:9071–9077. doi:10.1016/j.biortech.2010.06.127
Hernández-Mendoza CE, Buitrón G (2014) Suppression of methanogenic activity in anaerobic granular biomass for hydrogen production. J Chem Technol Biotechnol 89:143–149. doi:10.1002/jctb.4143
Zhang Y, Angelidaki I (2012) Innovative self-powered submersible microbial electrolysis cell (SMEC) for biohydrogen production from anaerobic reactors. Water Res 46:2727–2736. doi:10.1016/j.watres.2012.02.038
Gil-Carrera L, Escapa A, Moreno R, Morán A (2013) Reduced energy consumption during low strength domestic wastewater treatment in a semi-pilot tubular microbial electrolysis cell. J Environ Manag 122:1–7. doi:10.1016/j.jenvman.2013.03.001
Heidrich ES, Dolfing J, Scott K, Edwards SR, Jones C, Curtis TP (2013) Production of hydrogen from domestic wastewater in a pilot-scale microbial electrolysis cell. Appl Microbiol Biotechnol 97:6979–6989. doi:10.1007/s00253-012-4456-7
Sun M, Sheng GP, Mu ZX, Liu XW, Chen YZ, Wang HL, Yu HQ (2009) Manipulating the hydrogen production from acetate in a microbial electrolysis cell–microbial fuel cell-coupled system. J Power Sources 191:338–343. doi:10.1016/j.jpowsour.2009.01.087
Lu L, Ren N, Xing D, Logan BE (2009) Hydrogen production with effluent from an ethanol-H2-coproducing fermentation reactor using a single-chamber microbial electrolysis cell. Biosens Bioelectron 24:3055–3060. doi:10.1016/j.bios.2009.03.024
Rozendal RA, Hamelers HVM, Euverink GJW, Metz SJ, Buisman CJN (2006) Principle and perspectives of hydrogen production through biocatalyzed electrolysis. Int J Hydrog Ener 31:1632–1640. doi:10.1016/j.ijhydene.2005.12.006
Wang A, Sun D, Cao G, Wang H, Ren N, Wu WM, Logan BE (2011) Integrated hydrogen production process from cellulose by combining dark fermentation, microbial fuel cells, and a microbial electrolysis cell. Bioresour Technol 102:4137–4143. doi:10.1016/j.biortech.2010.10.137
Ruiz V, Ilhan ZE, Kang DW, Krajmalnik-Brown R, Buitrón G (2014) The source of inoculum plays a defining role in the development of MEC microbial consortia fed with acetic and propionic acid mixtures. J Biotechnol 182–183:11–18. doi:10.1016/j.jbiotec.2014.04.016
Lenin Babu M, Venkata Subhash G, Sarma PN, Venkata Mohan S (2013) Bio-electrolytic conversion of acidogenic effluents to biohydrogen: an integration strategy for higher substrate conversion and product recovery. Bioresour Technol 133:322–331. doi:10.1016/j.biortech.2013.01.029
Lu L, Xing D, Ren N, Logan BE (2012) Syntrophic interactions drive the hydrogen production from glucose at low temperature in microbial electrolysis cells. Bioresour Technol 124:68–76. doi:10.1016/j.biortech.2012.08.040
Sleutels THJA, Hamelers HVM, Rozendal RA, Buisman CJN (2009) Ion transport resistance in microbial electrolysis cells with anion and cation exchange membranes. Int J Hydrog Energy 34:3612–3620. doi:10.1016/j.ijhydene.2009.03.004
Kim KY, Chae KJ, Choi MJ, Ajayi FF, Jang A, Kim CW, Kim IS (2011) Enhanced coulombic efficiency in glucose-fed microbial fuel cells by reducing metabolite electron losses using dual-anode electrodes. Bioresour Technol 102(5):4144–4149. doi:10.1016/j.biortech.2010.12.036
Selembo PA, Perez JM, Lloyd WA, Logan BE (2009) High hydrogen production from glycerol or glucose by electrohydrogenesis using microbial electrolysis cells. Int J Hydrog Energy 34:5373–5381. doi:10.1016/j.ijhydene.2009.05.002
Acknowledgments
This study was supported by the International Fund of the Instituto de Ingeniería-UNAM and DGAPA-UNAM (PAPIIT IT100113). This research is part of the master supported by the CONACYT scholarship. The research was supported by TÁMOP 4.2.2/A-11/1/KONV-2012-0071, which was financed by the European Union and the European Social Fund. The János Bolyai Research Scholarship of the Hungarian Academy of Sciences is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rivera, I., Buitrón, G., Bakonyi, P. et al. Hydrogen production in a microbial electrolysis cell fed with a dark fermentation effluent. J Appl Electrochem 45, 1223–1229 (2015). https://doi.org/10.1007/s10800-015-0864-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-015-0864-6