Abstract
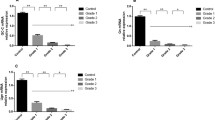
The role of colonic mucosal ornithine decarboxylase (ODC) in inflammatory bowel disease (IBD) remains controversial. This study assessed mucosal ODC activity in IBD patients segment by segment with regard to patient characteristics, disease activity/duration, medication, degree of mucosal inflammation, and presence/absence of epithelial regeneration and guanosine triphosphate (GTP) stimulation. Mucosal ODC activity was determined in biopsy specimens from the terminal ileum, cecum/ascending, transverse, and descending colon, and the sigmoid/rectum of 35 patients with IBD (18 with Crohn’s disease, 17 with ulcerative colitis) and 29 controls, using the amount of 14CO2 liberated from (carboxyl-14C)ornithine hydrochloride. GTP-stimulatable activity was expressed as the ratio of ODC activity in the presence and absence of GTP (70 μmol/L). Mucosal inflammation was assessed endoscopically/microscopically with previously described criteria. Presence/absence of mucosal regeneration also was determined by predefined criteria. Mucosal ODC-activity did not significantly differ in IBD patients and controls. There was a 4.4-fold activity gradient from the ileum to the rectum. Mucosal ODC activity was significantly higher in areas with epithelial regeneration compared to those without regeneration, and was stimulated by GTP by a factor of 1.42 in Crohn’s disease and 1.19 in ulcerative colitis patients compared to controls (p < 0.004). On the other hand, there was no significant association/relationship of mucosal ODC activity with disease activity/duration and the endoscopic/histologic degree of mucosal inflammation. The observation of unchanged mucosal ODC activity in patients with IBD and the absence of a significant relationship with clinical and endoscopic/histologic disease characteristics speaks against a major role of ODC in IBD as a major disease marker. The role of the ileorectal gradient, the enhanced activity in areas with epithelial regeneration, and the GTP-stimulatable form, however, need further investigation with regard to a possible involvement in carcinogenesis in IBD.

Similar content being viewed by others
References
Luk GD, Baylin SB (1983) Polyamines and intestinal growth–increased polyamine biosynthesis after jejunectomy. Am J Physiol 245:G656–G660
Luk GD, Goodwin G, Marton LJ, Baylin SB (1984) Polyamines are necessary for the survival of human small-cell lung carcinoma in culture. Proc Natl Acad USA 78:2355–2356
Richards JF, Janzen A, Peng T, Strumpfer A (1993) Function and origin of multiple ionic forms of hormone-induced ornithine decarboxylase. Arch Biochem Biophys 300:80–87
Luk GD, Baylin SB (1984) Ornithine decarboxylase as a biological marker in familial colonic polyposis. New Engl J Med 311:80–83
Pegg AE (1988) Polyamine metabolism and its importance in neoplastic growth and as target for chemotherapy. Cancer Res 48:759–774
Pillai RB, Tolia V, Rabah R, Simpson PM, Vijesurier R, Lin CH (1999) Increased colonic ornithine decarboxylase activity in inflammatory bowel disease in children. Dig Dis Sci 44:1565–1570
Rokkas T, Papatheodoron G, Karameris A (1992) Can ornithine decarboxylase serve as a biochemical marker for increased malignant potential in patients with ulcerative colitis? In: Dowling RH, Foelsch UR, Löser C (eds). Polyamines in the gastrointestinal tract. Kluwer Academic, Dordrecht, pp 187–191
Vatn MH, Elgjo K, Norheim A, Bergan A (1984) Measurement of enzyme activity in colonic biopsies. A test for premalignancy in ulcerative colitis? Scand J Gastroenterol 19:889–892
Obayashi M, Matsui-Yuasa I, Matsumoto T, Kitano A, Kobayashi K, Otani S (1992) Polyamine metabolism in colonic mucosa from patients with ulcerative colitis. Am J Gastroenterol 87:736–740
Fujita T, Saurai K (1995) Efficacy of glutamine–enriched enteral nutrition in an experimental model of mucosal ulcerative colitis. Br J Surg 82:749–751
Zehnter E, Roisch U, Kruis W, Breuer C, Diehl V (1996) Ornithine decarboxylase levels in patients with normal colonic mucosa. Eur J Clin Chem Clin Biochem 34:529–533
O’Brien TG, Hietala O, O’Donell K, Holmes M (1984) Activation of mouse epidermal tumor ornithine decarboxylase by GTP: evidence for different catalytic forms of the enzyme. Proc Natl Acad Sci USA 84:8927–8931
Cello JP, Schneideman DJ (1989) Ulcerative colitis. In: Sleisenger MH, Fordtran JS (eds) Gastrointestinal disease, pathophysiology, diagnosis, management. WB Saunders, Philadelphia, pp 1435–1477
Truelove SC, Witts LJ (1955) Cortisone in ulcerative colitis. Br Med J 2:1041–1048
Best WR, Becktel JM, Singleton JW, Kern F (1976) Development of a Crohn’s disease activity index. National Cooperative Crohn’s Disease Study. Gastroenterology 70:439–444
Gomes P, du Boulay C, Smith CL, Holdstock G (1986) Relationship between disease activity indices and colonoscopic findings in patients with colonic inflammatory bowel disease. Gut 27:92–95
Allgayer H, Kruis W, Paumgartner G, Wiebecke B, Brown L, Erdmann E (1988) Inverse relationship between colonic (Na+–K+)–ATPase activity and degree of mucosal inflammation in inflammatory bowel disease. Dig Dis Sci 33:417–422
Binder V (1970) A comparison between clinical state, macroscopic and microscopic appearances of rectal mucosa and cytologic picture of mucosal exudates in ulcerative colitis. Scand J Gastroenterol 5:627–632
Scheurlen C, Allgayer H, Hardt M, Kruis W (1998) Effect of short-term topical corticosteroid treatment on mucosal enzyme systems in patients with distal inflammatory bowel disease. Hepato-Gastroentrology 45:1539–1545
Fenoglio-Preiser CM, Noffsinger AE, Stemmermann GN, Lantz PE, Listorm MB (1998) Gastrointestinal Pathology. Lippincott-Raven, Philadelphia
Amasheh S, Barmeyer C, Koch CS, Tavalali S, Mankertz J, Epple HJ, Gehring MM, Florian P, Kroesen AJ, Zeitz M, Fromm M, Schulzke JD (2004) Cytokine-dependent transcriptional down-regulation of epithelial sodium channel (ENaC) in ulcerative colitis. Gastroenterology 126:1711–1720
Hietala OA, Yum KY, Pilon J, O’Donell K, Holroyde CP, Kline I, Reichard GA Litwin S, Gilmour SK, O’Brien TG (1990) Properties of ornithine decarboxylase in human colorectal adenocarcinomas. Cancer Res 50:2088–2094
Matsubara N, Hietala OA, Gilmour SK, Yum KY, LitWin S, Watts P, Brennan OB (1995) Association between high levels of ornithine decarboxylase activity and favorable prognosis in human colorectal carcinoma. Clin Cancer Res 1(6):665–671
Acknowledgment
This work was supported by the Frauke Weiskamp Stiftung (Foundation).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Allgayer, H., Roisch, U., Zehnter, E. et al. Colonic Ornithine Decarboxylase in Inflammatory Bowel Disease: Ileorectal Activity Gradient, Guanosine Triphosphate Stimulation, and Association with Epithelial Regeneration but Not the Degree of Inflammation and Clinical Features. Dig Dis Sci 52, 25–30 (2007). https://doi.org/10.1007/s10620-006-9515-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-006-9515-4