Abstract
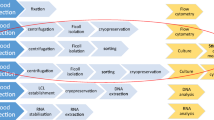
Peripheral blood mononuclear cells (PBMCs) are essential to the study of autoimmune, infectious, parasitic diseases, and cancer. In the rapidly growing field of cancer immunology, cellular phenotyping provides critical information about patient responses to treatments and treatment efficacies. Notably, the evaluation of T cell based therapies relies on the isolation of highly viable CD3+ T cell, CD4+ Helper T cell, and CD8+ Cytotoxic T cell populations before and during patient treatments. Cryopreservation of PBMC populations allows researchers to thaw and characterize clinical samples by flow cytometry, mass cytometry, sequencing, etc. in a high-throughput manner and in batches. Therefore, it is important to separate and bank an abundance of robust circulating immune cells. Here, we report our internal protocols for the high-quality separation, banking, and thawing of clinically relevant PBMC populations. We present quality control data from 11 melanoma patients and characterize their CD3+, CD4+, and CD8+ T cells by 4-color flow cytometry.





Similar content being viewed by others
References
Ben-Ami E et al (2017) Immunotherapy with single agent nivolumab for advanced leiomyosarcoma of the uterus: results of a phase 2 study. Cancer 123(17):3285–3290
Cosma G et al (2017) Identifying the presence of prostate cancer in individuals with PSA levels < 20 ng ml−1 using computational data extraction analysis of high dimensional peripheral blood flow cytometric phenotyping data. Front Immunol 8:1771
De la Cruz-Mosso U et al (2018) MIF promotes a differential Th1/Th2/Th17 inflammatory response in human primary cell cultures: predominance of Th17 cytokine profile in PBMC from healthy subjects and increase of IL-6 and TNF-α in PBMC from active SLE patients. Cell Immunol 324:42–49
Harty JT et al (2000) CD8+ T cell effector mechanisms in resistance to infection. Annu Rev Immunol 18:275–308
Kalos M (2011) Biomarkers in T cell therapy clinical trials. J Transl Med 9:138–147
Kutscher S, Dembek CJ, Allgayer S, Heltai S, Stadlbauer B, Biswas P, Nozza S, Tambussi G, Bogner JR, Stellbrink HJ, Goebel FD, Lusso P, Tinelli M, Poli G, Erfle V, Pohla H, Malnati M, Cosma A (2008) The intracellular detection of MIP-1Beta enhances the capacity to detect IFN-gamma mediated HIV-1-specific CD8 T-cell responses in a flow cytometric setting providing a sensitive alternative to the ELISPOT. AIDS Res Ther 6:5–22
Malyguine AM et al (2012) ELISPOT assay for monitoring cytotoxic T lymphocytes (CTL) activity in cancer vaccine clinical trials. Cells 1(2):111–126
Paredes RM et al (2018) Cryopreservation of human whole blood allows immunophenotyping by flow cytometry up to 30 days after cell isolation. J Immunol Methods 452:32–38
Patel T et al (2018) Development of an 8-color antibody panel for functional phenotyping of human CD8+ cytotoxic T cells from peripheral mononuclear cells. Cytotechnology 70:1–11
Pender MP et al (2014) Deficiency of CD8+ effector memory T cells is an early and persistent feature of multiple sclerosis. Mult Scler 20(14):1825–1832
Rao D et al (2018) Alterations in natural killer and dendritic cell subsets in individuals with HIV-associated neurotuberculosis. J Med Virol. https://doi.org/10.1002/jmv.25042
Salti SM et al (2011) Granzyme B regualtes antiviral CD8+ T cell responses. J Immunol 187(12):6301–6309
Schleker T et al (2018) Preserved in vitro immunoreactivity in children receiving long-term immunosuppressive therapy due to inflammatory bowel disease or autoimmune hepatitis. Mol Cell Pediatr 5(1):1
Singh N et al (2017) Inflammatory chemokines and their receptors in human visceral leishmaniasis: gene expression profile in peripheral blood, splenic cellular sources and their impact on trafficking of inflammatory cells. Mol Immunol 85:111–119
Speiser DE et al (2005) Rapid and strong human CD8+ T cell responses to vaccination with peptide, IFA, and CpG oligodeoxynucleotide 7909. J Clin Investig 15(3):739–746
Sridharan V et al (2016a) Definitive chemoradiation alters the immunologic landscape and immune checkpoints in head and neck cancer. Br J Cancer 115(2):252–260
Sridharan V et al (2016b) Immune profiling of adenoid cystic carcinoma: PD-L2 expression and associations with tumor-infiltrating lymphocytes. Cancer Immunol Res 4(8):679–687
Subrahmanyam PB et al (2018) Distinctive predictive biomarker candidates for response to anti-CTLA-4 and anti-PD-1 immunotherapy in melanoma patients. J Immunother Cancer 6:18
Weinberg A et al (2009) Optimization and limitations of use of cryopreserved peripheral blood mononuclear cells for functional and phenotypic T-cell characterization. Clin Vaccine Immunol 16(8):1176–1186
Funding
The Melanoma Research Alliance (F.S.H.), the Sharon Crowley Martin Memorial Fund for Melanoma Research (F.S.H.) and the Malcolm and Emily Mac Naught Fund for Melanoma Research (F.S.H.) at Dana-Farber Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
F.S. Hodi serves as a consultant to Genentech, Bristol-Myers Squibb, Merck, Novartis, Amgen, Sanofi, Bayer, Pfizer, EMD Serono, Verastem, Aduro, Celldex and Incyte.
Informed consent
All individuals gave written informed consent to participate in research prior to blood collection in accordance with Dana-Farber/Harvard Cancer Center Institutional Review Board (IRB) approved protocols.
Rights and permissions
About this article
Cite this article
Holland, M., Cunningham, R., Seymour, L. et al. Separation, banking, and quality control of peripheral blood mononuclear cells from whole blood of melanoma patients. Cell Tissue Bank 19, 783–790 (2018). https://doi.org/10.1007/s10561-018-9734-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-018-9734-x