Abstract
Introduction
Atrial fibrillation (AF) is the most common type of arrhythmia. Atrial remodeling is a major factor to the AF substrate. The purpose of the study is to explore whether aliskiren (ALS) has a cardioprotective effect and its potential molecular mechanisms on atrial remodeling.
Methods
In acute experiments, dogs were randomly assigned to Sham, Paced and Paced+aliskiren (10 mg kg−1) (Paced+ALS) groups, with 7 dogs in each group. Rapid atrial pacing (RAP) was maintained at 600 bpm for 2 h for paced and Paced+ALS groups and atrial effective refractory periods (AERPs), inducibility of AF (AFi) and average duration time (ADT) were measured. In chronic experiments, there were 5 groups: Sham, Sham+ALS, Paced, Paced+ALS and Paced+ALS+PI3K antagonist wortmannin (WM) (70 μg kg−1 day−1). RAP at 500 beats/min was maintained for 2 weeks. Inflammation and oxidative stress indicators were measured by ELISA assay, echocardiogram and pathology were used to assess atrial structural remodeling, phosphatidylinositol 3-hydroxy kinase/protein kinase B (PI3K/Akt) signaling pathways were studied by RT-PCR and western blotting to evaluate whether the cardioprotective effect of ALS works through PI3K/Akt signaling pathway.
Results
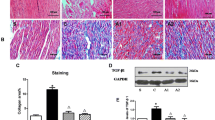
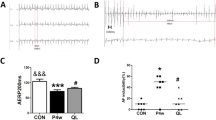
The electrophysiological changes were observed after 2-h pacing. The AERP shortened with increased AFi and ADT, which was attenuated by ALS (P < 0.05). After pacing for 2 weeks, oxidative stress and inflammation markers in the Paced group were significantly higher than those in the Sham group (P < 0.01) and were reduced by ALS treatment (P < 0.01). The reduced level of antioxidant enzymes caused by RAP was also found to be elevated in ALS-treated group (P < 0.01). The results of pathology and echocardiography showed that RAP can cause atrial enlargement, fibrosis (P < 0.01), and were attenuated in ALS treatment group. The PI3K/Akt signaling pathway were downregulated induced by RAP. ALS could upregulate the PI3K/Akt pathway expression (P < 0.05). Furthermore, the cardioprotective effects in structural remodeling of ALS were suppressed by WM.
Conclusions
ALS may offer cardioprotection in RAP-induced atrial remodeling, which may partly be ascribed to its anti-inflammatory and anti-oxidative stress action and the regulation of PI3K/Akt signaling pathway.




Similar content being viewed by others
References
Staszewsky L, Masson S, Barlera S, Disertori M, Boni S, Bertoli D, et al. Cardiac remodeling, circulating biomarkers and clinical events in patients with a history of atrial fibrillation. Data from the GISSI-AF trial. Cardiovasc Drugs Ther. 2015;29(6):551–61.
Kirchhof P. The future of atrial fibrillation management: integrated care and stratified therapy. Lancet. 2017;390(10105):1873–87.
Dilaveris PE, Kennedy HL. Silent atrial fibrillation: epidemiology, diagnosis, and clinical impact. Clin Cardiol. 2017;40(6):413–8.
Koivumäki JT, Seemann G, Maleckar MM, Tavi P. In silico screening of the key cellular remodeling targets in chronic atrial fibrillation. PLoS Comput Biol. 2014;10(5):e1003620.
Colman MA, Aslanidi OV, Kharche S, Boyett MR, Garratt C, Hancox JC, et al. Pro-arrhythmogenic effects of atrial fibrillation-induced electrical remodelling: insights from the three-dimensional virtual human atria. J Physiol. 2013;591(17):4249–72.
Kalla M, Sanders P, Kalman JM, Lee G. Radiofrequency catheter ablation for atrial fibrillation: approaches and outcomes. Heart Lung Circ. 2017;26(9):941–9.
Cappato R, Calkins H, Chen SA, Davies W, Iesaka Y, Kalman J, et al. Updated worldwide survey on the methods, efficacy, and safety of catheter ablation for human atrial fibrillation. Circ Arrhythm Electrophysiol. 2010;3(1):32–8.
Chrysostomakis SI, Karalis IK, Simantirakis EN, Koutsopoulos AV, Mavrakis HE, Chlouverakis GI, et al. Angiotensin II type 1 receptor inhibition is associated with reduced tachyarrhythmia-induced ventricular interstitial fibrosis in a goat atrial fibrillation model. Cardiovasc Drugs Ther. 2007;21(5):357–65.
Nair GM, Nery PB, Redpath CJ, Birnie DH. The role of renin angiotensin system in atrial fibrillation. J Atr Fibrillation. 2014;6(6):972.
Azizi M, Ménard J. Renin inhibitors and cardiovascular and renal protection: an endless quest? Cardiovasc Drugs Ther. 2013;27(2):145–53.
Brown MJ. Aliskiren. Circulation. 2008;118(7):773–84.
Zhao Z, Wang X, Li J, Yang W, Cheng L, Chen Y, et al. Protective effects of aliskiren on atrial ionic remodeling in a canine model of rapid atrial pacing. Cardiovasc Drugs Ther. 2014;28(2):137–43.
Zhao Z, Chen Y, Li W, Wang X, Li J, Yang W, et al. Aliskiren protecting atrial structural remodeling from rapid atrial pacing in a canine model. Naunyn Schmiedeberg's Arch Pharmacol. 2016;389(8):863–71.
Li J, Solus J, Chen Q, Rho YH, Milne G, Stein CM, et al. Role of inflammation and oxidative stress in atrial fibrillation. Heart Rhythm. 2010;7(4):438–44.
Nattel S, Li D. Ionic remodeling in the heart: pathophysiological significance and new therapeutic opportunities for atrial fibrillation. Circ Res. 2000;87(6):440–7.
Horio T, Akiyama M, Iwashima Y, Yoshihara F, Nakamura S, Tokudome T, et al. Preventive effect of renin-angiotensin system inhibitors on new-onset atrial fibrillation in hypertensive patients: a propensity score matching analysis. J Hum Hypertens. 2017;31(7):450–6.
Ellermann C, Mittelstedt A, Wolfes J, Willy K, Leitz P, Reinke F, et al. Action potential triangulation explains acute proarrhythmic effect of aliskiren in a whole-heart model of atrial fibrillation. Cardiovasc Toxicol. 2020;20(1):49–57.
Mascolo A, Urbanek K, De Angelis A, Sessa M, Scavone C, Berrino L, et al. Angiotensin II and angiotensin 1-7: which is their role in atrial fibrillation? Heart Fail Rev. 2019;2:367–80. https://doi.org/10.1007/s10741-019-09837-7.
Roşianu ŞH, Roşianu AN, Aldica M, Căpâlneanu R, Buzoianu AD. Inflammatory markers in paroxysmal atrial fibrillation and the protective role of renin-angiotensin-aldosterone system inhibitors. Clujul Med. 2013;86(3):217–21.
Karam BS, Chavez-Moreno A, Koh W, Akar JG, Akar FG. Oxidative stress and inflammation as central mediators of atrial fibrillation in obesity and diabetes. Cardiovasc Diabetol. 2017;16(1):120.
Zhao Q, Zhang S, Zhao H, Zhang S, Dai Z, Qian Y, et al. Median nerve stimulation prevents atrial electrical remodelling and inflammation in a canine model with rapid atrial pacing. Europace. 2018;20(4):712–8.
Dai M, Bao M, Zhang Y, Yu L, Cao Q, Tang Y, et al. Low-level carotid baroreflex stimulation suppresses atrial fibrillation by inhibiting left stellate ganglion activity in an acute canine model. Heart Rhythm. 2016;13(11):2203–12.
Chilukoti RK, Giese A, Malenke W, Homuth G, Bukowska A, Goette A, et al. Atrial fibrillation and rapid acute pacing regulate adipocyte/adipositas-related gene expression in the atria. Int J Cardiol. 2015;187:604–13.
Hu YF, Chen YJ, Lin YJ, Chen SA. Inflammation and the pathogenesis of atrial fibrillation. Nat Rev Cardiol. 2015;12(4):230–43.
Lazzerini PE, Laghi-Pasini F, Acampa M, Srivastava U, Bertolozzi I, Giabbani B, et al. Systemic inflammation rapidly induces reversible atrial electrical remodeling: the role of interleukin-6-mediated changes in connexin expression. J Am Heart Assoc. 2019;8(16):e011006.
Yao C, Veleva T, Scott L Jr, Cao S, Li L, Chen G, et al. Enhanced cardiomyocyte NLRP3 inflammasome signaling promotes atrial fibrillation. Circulation. 2018;138(20):2227–42.
Ribeiro Mesquita TR, Zhang R, de Couto G, Valle J, Sanchez L, Rogers RG, et al. Mechanisms of atrial fibrillation in aged rats with heart failure with preserved ejection fraction. Heart Rhythm. 2020. https://doi.org/10.1016/j.hrthm.2020.02.007.
Yoo S, Aistrup G, Shiferaw Y, Ng J, Mohler PJ, Hund TJ, et al. Oxidative stress creates a unique, CaMKII-mediated substrate for atrial fibrillation in heart failure. JCI Insight. 2018;3(21).
Ren X, Wang X, Yuan M, Tian C, Li H, Yang X, et al. Mechanisms and treatments of oxidative stress in atrial fibrillation. Curr Pharm Des. 2018;24(26):3062–71.
Takei Y, Ichikawa M, Kijima Y. Oral direct renin inhibitor aliskiren reduces in vivo oxidative stress and serum matrix metalloproteinase-2 levels in patients with permanent atrial fibrillation. J Arrhythm. 2015;31(2):76–7.
Nattel S, Harada M. Atrial remodeling and atrial fibrillation: recent advances and translational perspectives. J Am Coll Cardiol. 2014;63(22):2335–45.
Jalloul Y, Refaat MM. IL-6 rapidly induces reversible atrial electrical remodeling by downregulation of cardiac connexins. J Am Heart Assoc. 2019;8(16):e013638.
Bonadei I, Vizzardi E, D'Aloia A, Sciatti E, Raddino R, Metra M. Role of aliskiren on arterial stiffness and endothelial function in patients with primary hypertension. J Clin Hypertens (Greenwich). 2014;16(3):202–6.
Altarejo Marin T, Machado Bertassoli B, Alves de Siqueira de Carvalho A, Feder D. The use of aliskiren as an antifibrotic drug in experimental models: a systematic review. Drug Dev Res 2019 12. doi: https://doi.org/10.1002/ddr.21610.114, 126.
Chen L, Liu P, Feng X, Ma C. Salidroside suppressing LPS-induced myocardial injury by inhibiting ROS-mediated PI3K/Akt/mTOR pathway in vitro and in vivo. J Cell Mol Med. 2017;21(12):3178–89.
Ma L, Li XP, Ji HS, Liu YF, Li EZ. Baicalein protects rats with diabetic cardiomyopathy against oxidative stress and inflammation injury via phosphatidylinositol 3-kinase (PI3K)/AKT pathway. Med Sci Monit. 2018;24:5368–75.
Le Belle JE, Orozco NM, Paucar AA, Saxe JP, Mottahedeh J, Pyle AD, et al. Proliferative neural stem cells have high endogenous ROS levels that regulate self-renewal and neurogenesis in a PI3K/Akt- dependant manner. Cell Stem Cell. 2011;8(1):59–71.
Lu G, Li J, Zhai Y, Li Q, Xie D, Zhang J, et al. Spironolactone suppresses aldosterone-induced Kv1.5 expression by attenuating mineralocorticoid receptor-Nox1/2/4-mediated ROS generation in neonatal rat atrial myocytes. Biochem Biophys Res Commun. 2019;520(2):379–84.
Takahama H, Minamino T, Hirata A, Ogai A, Asanuma H, Fujita M, et al. Granulocyte colony-stimulating factor mediates cardioprotection against ischemia/reperfusion injury via phosphatidylinositol-3-kinase/Akt pathway in canine hearts. Cardiovasc Drugs Ther. 2006;20(3):159–65.
Qiu H, Ma J, Wu H, Ding C. DL-3-n-butylphthalide improves ventricular function, and prevents ventricular remodeling and arrhythmias in post-MI rats. Naunyn Schmiedeberg's Arch Pharmacol. 2018;391(6):627–37.
Ye Y, Qian J, Castillo AC, Perez-Polo JR, Birnbaum Y. Aliskiren and valsartan reduce myocardial AT1 receptor expression and limit myocardial infarct size in diabetic mice. Cardiovasc Drugs Ther. 2011;25(6):505–15.
Zhao J, Liu E, Li G, Qi L, Li J, Yang W. Effects of the angiotensin-(1-7)/Mas/PI3K/Akt/nitric oxide axis and the possible role of atrial natriuretic peptide in an acute atrial tachycardia canine model. J Renin-Angiotensin-Aldosterone Syst. 2015;16(4):1069–77.
Acknowledgments
The authors would like to express their special thanks to Beth Gabris in the Department of Medicine, Heart and Vascular Institute, University of Pittsburgh, USA, for her great help in editing the English language of the manuscript.
Funding
This study was funded by the Program of Natural Science Foundation of China (grant number 81370300), the China Education Ministry Colleges and Universities Special Scientific Research Foundation for Doctoral Advisor Class (grant number 20121202110004), the Tianjin Natural Science Foundation (grant number 17JCQNJC11400) and the Key Laboratory Science Foundation of Second Hospital of Tianjin Medical University (grant number 2017ZDSYS05).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, Z., Li, R., Wang, X. et al. Attenuation of atrial remodeling by aliskiren via affecting oxidative stress, inflammation and PI3K/Akt signaling pathway. Cardiovasc Drugs Ther 35, 587–598 (2021). https://doi.org/10.1007/s10557-020-07002-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-07002-z