Abstract
Purpose
Inflammatory breast cancer (IBC) is an aggressive variant for which axillary lymph node (LN) dissection following neoadjuvant chemotherapy (NACT) remains standard of care. But with increasingly effective systemic therapy, it is unclear whether more limited axillary surgery may be appropriate in some IBC patients. We sought to examine whether extent of axillary LN surgery was associated with overall survival (OS) for IBC.
Methods
Female breast cancer patients with non-metastatic IBC (cT4d) diagnosed 2010–2014 were identified in the National Cancer Data Base. Cox proportional hazards modeling was used to estimate the association between extent of axillary surgery (≤ 9 vs ≥ 10 LNs removed) and OS after adjusting for covariates, including post-NACT nodal status (ypN0 vs ypN1-3) and radiotherapy receipt (yes/no).
Results
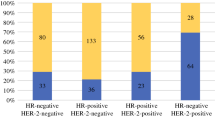
3471 patients were included: 597 (17.2%) had cN0 disease, 1833 (52.8%) had cN1 disease, and 1041 (30%) had cN2-3 disease. 49.9% of cN0 patients were confirmed to be ypN0 on post-NACT surgical pathology. Being ypN0 (vs ypN1-3) was associated with improved adjusted OS for all patients. Radiotherapy was associated with improved adjusted OS for cN1 and cN2-3 patients but not for cN0 patients. Regardless of ypN status, there was a trend towards improved adjusted OS with having ≥ 10 (vs ≤ 9) LNs removed for cN2-3 patients (HR 0.78, 95% CI 0.60–1.01, p = 0.06) but not for cN0 patients (p = 0.83).
Conclusions
A majority of IBC patients in our study presented with node-positive disease, and for those presenting with cN2-3 disease, more extensive axillary surgery is potentially associated with improved survival. For cN0 patients, however, more extensive axillary surgery was not associated with a survival benefit, suggesting an opportunity for more personalized care.


Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the National Cancer Data Base (NCDB) but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are, however, available from the authors upon reasonable request and with permission of the NCDB.
References
Hance KW, Anderson WF, Devesa SS, Young HA, Levine PH (2005) Trends in inflammatory breast carcinoma incidence and survival: the surveillance, epidemiology, and end results program at the National Cancer Institute. J Natl Cancer Inst 97(13):966–975
van Uden DJP, van Laarhoven HWM, Westenberg AH, de Wilt JHW, Blanken-Peeters CFJM (2015) Inflammatory breast cancer: an overview. Crit Rev Oncol/Hematol. 93(2):116–126
"Inflammatory Breast Cancer." NCCN Guideline Version 2.2019. https://www.nccn.org/. Accessed 2019 July 01.
Gianni L, Pienkowski T, Im Y-H et al (2016) 5-year analysis of neoadjuvant pertuzumab and trastuzumab in patients with locally advanced, inflammatory, or early-stage HER2-positive breast cancer (NeoSphere): a multicentre, open-label, phase 2 randomised trial. Lancet Oncol 17(6):791–800
Nahleh ZA, Barlow WE, Hayes DF et al (2016) SWOG S0800 (NCI CDR0000636131): addition of bevacizumab to neoadjuvant nab-paclitaxel with dose-dense doxorubicin and cyclophosphamide improves pathologic complete response (pCR) rates in inflammatory or locally advanced breast cancer. Breast Cancer Res Treat 158(3):485–495
Nakhlis F, Regan MM, Warren LE et al (2017) The impact of residual disease after preoperative systemic therapy on clinical outcomes in patients with inflammatory breast cancer. Ann Surg Oncol 24(9):2563–2569
Devi GR, Hough H, Barrett N et al (2019) Perspectives on inflammatory breast cancer (IBC) research, clinical management and community engagement from the Duke IBC consortium. J Cancer. 10(15):3344–3351
Classe JM, Bordes V, Campion L et al (2009) Sentinel lymph node biopsy after neoadjuvant chemotherapy for advanced breast cancer: results of Ganglion Sentinelle et Chimiotherapie Neoadjuvante, a French prospective multicentric study. J Clin Oncol: Off J Am Soc Clin Oncol 27(5):726–732
Mamounas EP, Brown A, Anderson S et al (2005) Sentinel node biopsy after neoadjuvant chemotherapy in breast cancer: results from national surgical adjuvant breast and bowel project protocol B-27. J Clin Oncol: Off J Am Soc Clin Oncol 23(12):2694–2702
van der Heiden-van der Loo M, de Munck L, Sonke GS et al (2015) Population based study on sentinel node biopsy before or after neoadjuvant chemotherapy in clinically node negative breast cancer patients: identification rate and influence on axillary treatment. Eur J Cancer. 51(8):915–921
Boileau JF, Poirier B, Basik M et al (2015) Sentinel node biopsy after neoadjuvant chemotherapy in biopsy-proven node-positive breast cancer: the SN FNAC study. J Clin Oncol: Off J Am Soc Clin Oncol. 33(3):258–264
Boughey JC, Suman VJ, Mittendorf EA et al (2013) Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (alliance) clinical trial. JAMA 310(14):1455–1461
Kuehn T, Bauerfeind I, Fehm T et al (2013) Sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy (SENTINA): a prospective, multicentre cohort study. Lancet Oncol 14(7):609–618
Imeokparia FO, Hughes TM, Dossett LA, Jeruss JS, Chang AE, Sabel MS (2019) Axillary pathologic complete response in inflammatory breast cancer patients: implications for SLNB? Ann Surg Oncol 26(10):3374–3379
Gianni L, Eiermann W, Semiglazov V et al (2010) Neoadjuvant chemotherapy with trastuzumab followed by adjuvant trastuzumab versus neoadjuvant chemotherapy alone, in patients with HER2-positive locally advanced breast cancer (the NOAH trial): a randomised controlled superiority trial with a parallel HER2-negative cohort. Lancet 375(9712):377–384
The National Cancer Data Base. The American College of Surgeons. www.ncdb.org.
"Surgical Axillary Stagng." NCCN Guideline Version 2.2019. https://www.nccn.org/. Accessed 1 Jul 2019.
Bilimoria KY, Bentrem DJ, Hansen NM et al (2009) Comparison of sentinel lymph node biopsy alone and completion axillary lymph node dissection for node-positive breast cancer. J Clin Oncol: Off J Am Soc Clin Oncol. 27(18):2946–2953
Rosenberger LH, Ren Y, Thomas SM, et al. Axillary lymph node dissection in node-positive breast cancer: are 10-nodes adequate and when is enough, enough? Breast Cancer Res Treat 2019 Nov 18 [Epub ahead of print].
Hidar S, Bibi M, Gharbi O et al (2009) Sentinel lymph node biopsy after neoadjuvant chemotherapy in inflammatory breast cancer. Int J Surg (London, England). 7(3):272–275
Stearns V, Ewing CA, Slack R, Penannen MF, Hayes DF, Tsangaris TN (2002) Sentinel lymphadenectomy after neoadjuvant chemotherapy for breast cancer may reliably represent the axilla except for inflammatory breast cancer. Ann Surg Oncol 9(3):235–242
DeSnyder SM, Mittendorf EA, Le-Petross C et al (2018) Prospective feasibility trial of sentinel lymph node biopsy in the setting of inflammatory breast cancer. Clin Breast Cancer. 18(1):e73–e77
Caretta-Weyer H, Sisney GA, Beckman C et al (2012) Impact of axillary ultrasound and core needle biopsy on the utility of intraoperative frozen section analysis and treatment decision making in women with invasive breast cancer. Am J Surg. 204(3):308–314
Britton PD, Provenzano E, Barter S et al (2009) Ultrasound guided percutaneous axillary lymph node core biopsy: how often is the sentinel lymph node being biopsied? Breast 18(1):13–16
Caudle AS, Yang WT, Krishnamurthy S et al (2016) Improved axillary evaluation following neoadjuvant therapy for patients with node-positive breast cancer using selective evaluation of clipped nodes: implementation of targeted axillary dissection. J Clin Oncol: Off J Am Soc Clin Oncol. 34(10):1072–1078
Boughey JC, Ballman KV, Le-Petross HT et al (2016) Identification and resection of clipped node decreases the false-negative rate of sentinel lymph node surgery in patients presenting with node-positive breast cancer (T0–T4, N1–N2) who receive neoadjuvant chemotherapy: results from ACOSOG Z1071 (alliance). Ann Surg 263(4):802–807
Fayanju OM, Ren Y, Thomas SM et al (2018) The clinical significance of breast-only and node-only pathologic complete response (pCR) after neoadjuvant chemotherapy (NACT): a review of 20,000 breast cancer patients in the National Cancer Data Base (NCDB). Ann Surg 268(4):591–601
Funding
Dr. Fayanju is currently supported by the National Institutes of Health (NIH) under Award Number 1K08CA241390 (PI: Fayanju) and previously by the National Center for Advancing Translational Sciences (NCATS) of the NIH under Award Number 1KL2TR002554 (PI: Svetkey). Dr. Greenup is supported by the NIH BIRCWH K12HD043446 (PI: Andrews). Dr. Suneja is supported by grants K08CA228631 (PI: Suneja) and P30AI064518 (PI: Weinhold) from the NIH. Dr. Devi is supported by grants from the Department of Defense (DoD) W81XWH-17–1-0297 (PI: Devi) and the IBC Network Foundation (PI: Devi). This work is also supported by the Duke Cancer Institute through NIH grant P30CA014236 (PI: Kastan). The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Force is a consultant for Genomic Health, Nanostring, and Pfizer, Dr. King is a consultant for Genomic Health, Dr. Hyslop is a consultant for AbbVie, and Dr. Fayanju, Mr. Ren, Dr. Greenup, Dr. Plichta, Dr. Rosenberger, Dr. Suneja, Dr. Devi, Dr. Nakhlis, and Dr. Hwang have no conflicts of interest to disclose.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with human participants or animals performed by any of the authors as de-identified data were used and the study was granted exempt status by our institutional review board.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fayanju, O.M., Ren, Y., Greenup, R.A. et al. Extent of axillary surgery in inflammatory breast cancer: a survival analysis of 3500 patients. Breast Cancer Res Treat 180, 207–217 (2020). https://doi.org/10.1007/s10549-020-05529-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-020-05529-1