Abstract
Background and objectives
Lysinuric protein intolerance (LPI) is a rare multisystemic metabolic disease. The objective of the study was to describe presentation and course of lung involvement in a cohort of ten children.
Patients and methods
Retrospective review of patients followed at Necker-Enfants Malades University Hospital between 1980 and 2012 for a LPI. In patients with lung involvement, clinical data, chest radiographs, pulmonary function tests, bronchoalveolar lavages, and lung biopsies were analyzed. The first and last high-resolution computed tomography (HRCT) were also reviewed.
Results
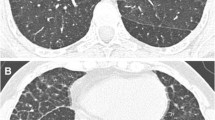
Lung involvement was observed in ten of 14 patients (71 %). Five patients had an acute onset of respiratory symptoms, three had a progressive onset and two were free of symptoms. During the period studied, six patients (60 %) died, all in a context of respiratory failure. Clinical presentation and course were highly variable, even in the same family. HRCT were performed in seven cases, showing in all cases an interstitial pattern and fibrosis in four. All ten patients had pulmonary alveolar proteinosis (PAP) confirmed by histopathological analysis. Five patients had pulmonary fibrosis (at biopsy and/or HRCT scan). Two patients underwent whole lung lavages, without efficiency.
Conclusion
PAP is a constant feature in children with LPI and lung involvement. Pulmonary fibrosis is frequent and these two pathologies may develop independently. This study shows the heterogeneity of presentation and outcome. Lung injury could be secondary to impaired phagocytic function and abnormal inflammatory and immune responses intrinsic to the SLC7A7 mutant phenotype. HRCT is recommended to detect lung involvement.


Similar content being viewed by others
References
Barilli A, Rotoli BM, Visigalli R et al (2010) In lysinuric protein intolerance system y + L activity is defective in monocytes and in GM-CSF-differentiated macrophages. Orphanet J Rare Dis 5:32
Barilli A, Rotoli BM, Visigalli R et al (2012) Impaired phagocytosis in macrophages from patients affected by lysinuric protein intolerance. Mol Genet Metab 105(4):585–589
Borsani G, Bassi MT, Sperandeo MP et al (1999) SLC7A7, encoding a putative permease-related protein, is mutated in patients with lysinuric protein intolerance. Nat Genet 21(3):297–301
Carey B, Trapnell BC (2010) The molecular basis of pulmonary alveolar proteinosis. Clin Immunol 135(2):223–235
Ceruti M, Rodi G, Stella GM et al (2007) Successful whole lung lavage in pulmonary alveolar proteinosis secondary to lysinuric protein intolerance: a case report. Orphanet J Rare Dis 2:14
de Blic J (2004) Pulmonary alveolar proteinosis in children. Paediatr Respir Rev 5(4):316–322
de Blic J, Midulla F, Barbato A et al (2000) Bronchoalveolar lavage in children. ERS task force on bronchoalveolar lavage in children. European respiratory society. Eur Respir J 15(1):217–231
DiRocco M, Garibotto G, Rossi GA et al (1993) Role of haematological, pulmonary and renal complications in the long-term prognosis of patients with lysinuric protein intolerance. Eur J Pediatr 152(5):437–440
Douda DN, Farmakovski N, Dell S et al (2009) SP-D counteracts GM-CSF-mediated increase of granuloma formation by alveolar macrophages in lysinuric protein intolerance. Orphanet J Rare Dis 4:29
Gordon WC, Gibson B, Leach MT et al (2007) Haemophagocytosis by myeloid precursors in lysinuric protein intolerance. Br J Haematol 138(1):1
Granillo OM, Brahmajothi MV, Li S et al (2008) Pulmonary alveolar epithelial uptake of S-nitrosothiols is regulated by L-type amino acid transporter. Am J Physiol Lung Cell Mol Physiol 295(1):L38–43
Guzel-Ozanturk A, Ozgul RK, Unal O et al (2013) Molecular and clinical evaluation of Turkish patients with lysinuric protein intolerance. Gene 521(2):293–295
Hansell DM, Bankier AA, MacMahon H et al (2008) Fleischner society: glossary of terms for thoracic imaging. Radiology 246(3):697–722
Kerem E, Elpelg ON, Shalev RS et al (1993) Lysinuric protein intolerance with chronic interstitial lung disease and pulmonary cholesterol granulomas at onset. J Pediatr 123(2):275–278
Kitamura T, Tanaka N, Watanabe J et al (1999) Idiopathic pulmonary alveolar proteinosis as an autoimmune disease with neutralizing antibody against granulocyte/macrophage colony-stimulating factor. J Exp Med 190(6):875–880
Koizumi A, Shoji Y, Nozaki J et al (2000) A cluster of lysinuric protein intolerance (LPI) patients in a northern part of Iwate, Japan due to a founder effect. The Mass Screening Group. Hum Mutat 16(3):270–271
Ogier De Baulny H, Schiff M, Dionisi-Vici C (2012) Lysinuric protein intolerance (LPI): a multi organ disease by far more complex than a classic urea cycle disorder. Mol Genet Metab 106(1):12–17
Palacin M, Borsani G, Sebastio G (2001) The molecular bases of cystinuria and lysinuric protein intolerance. Curr Opin Genet Dev 11(3):328–335
Palacin M, Bertran J, Chillaron J et al (2004) Lysinuric protein intolerance: mechanisms of pathophysiology. Mol Genet Metab 81(Suppl 1):S27–37
Parenti G, Sebastio G, Strisciuglio P et al (1995) Lysinuric protein intolerance characterized by bone marrow abnormalities and severe clinical course. J Pediatr 126(2):246–251
Parto K, Penttinen R, Paronen I et al (1993a) Osteoporosis in lysinuric protein intolerance. J Inherit Metab Dis 16(2):441–450
Parto K, Svedstrom E, Majurin ML et al (1993b) Pulmonary manifestations in lysinuric protein intolerance. Chest 104(4):1176–1182
Santamaria F, Parenti G, Guidi G et al (1996) Early detection of lung involvement in lysinuric protein intolerance: role of high-resolution computed tomography and radioisotopic methods. Am J Respir Crit Care Med 153(2):731–735
Santamaria F, Brancaccio G, Parenti G et al (2004) Recurrent fatal pulmonary alveolar proteinosis after heart-lung transplantation in a child with lysinuric protein intolerance. J Pediatr 145(2):268–272
Sebastio G, Sperandeo MP, Andria G (2011) Lysinuric protein intolerance: reviewing concepts on a multisystem disease. Am J Med Genet C: Semin Med Genet 157(1):54–62
Simell O (2001) Lysinuric protein intolerance and other cationic aminoacidurias. In: Valle D et al (eds) Scriver CR BD. The metabolic and molecular bases of inherited disease. New York, McGraw-Hill, pp 4933–4955
Tanner LM, Nanto-Salonen K, Niinikoski H et al (2007) Nephropathy advancing to end-stage renal disease: a novel complication of lysinuric protein intolerance. J Pediatr 150(6):631–634
Torrents D, Mykkanen J, Pineda M et al (1999) Identification of SLC7A7, encoding y + LAT-1, as the lysinuric protein intolerance gene. Nat Genet 21(3):293–296
Trapnell BC, Whitsett JA, Nakata K (2003) Pulmonary alveolar proteinosis. N Engl J Med 349(26):2527–2539
Compliance with ethics guidelines
Conflict of interest
None.
Informed consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000.
This study was approved by the Institutional Review Board of the Société de Pneumologie de Langue Française.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: John H Walter
Rights and permissions
About this article
Cite this article
Valimahamed-Mitha, S., Berteloot, L., Ducoin, H. et al. Lung involvement in children with lysinuric protein intolerance. J Inherit Metab Dis 38, 257–263 (2015). https://doi.org/10.1007/s10545-014-9777-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-014-9777-5