Abstract
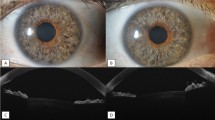
The standard of care for posterior segment disorders such as wet age-related macular degeneration, diabetic macular oedema and retinal vascular occlusions is pharmacotherapy by intravitreal drug delivery. Since the therapeutic effect of these drugs lasts only around 4 to 8 weeks, repeated intravitreal injections are required. Pain is experienced by the patients during injection as the needle courses through the sclera and choroid. The current work describes the design and development of a novel anodized titanium alloy implant that allows for intravitreal injections through the implant so that the needle transverses only the conjunctiva, thus minimizing discomfort to the patient. Both ex-vivo testing of the implant in enucleated goat’s eye as well as in-vivo validation in rabbit eyes was carried out. The implant was placed through pars plana via a minor surgical procedure and was sutured to the sclera and covered with conjunctiva. Subsequent intravitreal injections were administered under topical anaesthesia with a 30-gauge needle through the implant thus delivering the drug into the vitreous cavity. Repeated intravitreal injections were administered every 2 weeks via the implant for 3 months in 4 rabbits. Apart from cataract in 1 rabbit, no complications were observed. There was no evidence of intra-ocular inflammation or infection at final follow-up. Histopathological analysis did not reveal any inflammation or necrosis around the area of implant. The implants were subsequently removed at 5 months and scleral wound was closed with a single suture. The sclera and overlying conjunctiva healed well and no intraocular complications were observed after removal.





Similar content being viewed by others
References
E.M. Del Amo, A. Urtti, Exp. Eye Res. 137, 111 (2015)
P.S. Apaolaza, M. Busch, E. Asin-Prieto, K. Peynshaert, R. Rathod, K. Remaut, N. Dünker, A. Göpferich, Exp. Eye Res. 198, 108151 (2020)
T. Baba, G. Miura, T. Tatsumi, M. Sakurai, S. Yamamoto, Jpn J. Ophthalmol. 65, 492 (2021)
I. Bekerman, P. Gottlieb, M. Vaiman, J. Ophthalmol. 2014, (2014)
B. Bilgin, Ş Bilak, Middle East. Afr. J. Ophthalmol. 26, 55 (2019)
A.V. Bux, F. Fortunato, A. Barone, V. Russo, N.Delle Noci, C. Iaculli, Eur. J. Ophthalmol. 11206721211024804 (2021)
P.A. Campochiaro, D.M. Marcus, C.C. Awh, C. Regillo, A.P. Adamis, V. Bantseev, Y. Chiang, J.S. Ehrlich, S. Erickson, W.D. Hanley, J. Horvath, K.F. Maass, N. Singh, F. Tang, G. Barteselli, Ophthalmology 126, 1141 (2019)
Food and Drug Administration, J. Int. Assoc. Physicians AIDS Care 1, 37 (1995)
F. Gomi, R. Toyoda, A.H. Yoon, K. Imai, J. Clin. Med. 10, 3106 (2021)
M.S. Habib, Ther. Deliv 9, 547 (2018)
J.S. Heier, A.N. Antoszyk, P.R. Pavan, S.R. Leff, P.J. Rosenfeld, T.A. Ciulla, R.F. Dreyer, R.C. Gentile, J.P. Sy, G. Hantsbarger, N. Shams, Ophthalmology 113, 633 (2006)
N.M. Holekamp, P.A. Campochiaro, M.A. Chang, D. Miller, D. Pieramici, A.P. Adamis, C. Brittain, E. Evans, D. Kaufman, K.F. Maass, S. Patel, S. Ranade, N. Singh, G. Barteselli, C. Regillo, A. Investigators, Ophthalmology 129, 295 (2022)
M. Hu, G. Huang, F. Karasina, V.G. Wong, Invest. Ophthalmol. Vis. Sci. 49, 5627 (2008)
X. Huang, Y. Chau, Exp. Eye Res. 186, 107711 (2019)
Y.F.K. Islam, C.R. Blake, S.K. Gibran, Eye 35, 1850 (2021)
G.J. Jaffe, D. Martin, D. Callanan, P.A. Pearson, B. Levy, T. Comstock, Ophthalmology 113, 1020 (2006)
A.M. Khanani, D. Callanan, R. Dreyer, S. Chen, J.G. Howard, J.J. Hopkins, C. Lin, M. Lorenz-candlin, S. Makadia, S. Patel, T. Tam, S. Gune, Ophthalmol. Retin 5, 775 (2020)
R. Lee, T.Y. Wong, C. Sabanayagam, Eye Vis. 2, 1 (2015)
M.G. Maguire, D.F. Martin, G.-S. Ying, G.J. Jaffe, E. Daniel, J.E. Grunwald, C.A. Toth, F.L. Ferris, S.L. Fine, Physiol. Behav. 176, 139 (2017)
A. Martel, S. Nahon-Esteve, K. Martini, F. Almairac, S. Baillif, Graefe’s Arch. Clin. Exp. Ophthalmol. 258, 1395 (2020)
D.F. Martin, M.G. Maguire, S.L. Fine, G. Ying, G.J. Jaffe, J.E. Grunwald, C. Toth, M. Redford, F.L. Ferris, Ophthalmology 119, 1388 (2013)
E. Moisseiev, A. Loewenstein, Harefuah 158, 121 (2019)
E. Moisseiev, M. Waisbourd, E. Ben-Artsi, E. Levinger, A. Barak, T. Daniels, K. Csaky, A. Loewenstein, I.S. Barequet, Graefe’s Arch. Clin. Exp. Ophthalmol. 252, 331 (2014)
B.P. Nicholson, R.P. Singh, J.E. Sears, C.Y. Lowder, P.K. Kaiser, Am. J. Ophthalmol. 154, 969 (2012)
A. Nieto, H. Hou, M.J. Sailor, W.R. Freeman, L. Cheng, Exp. Eye Res. 116, 161 (2013)
S.F.E. Nilsson, J. Ver Hoeve, S. Wu, P.L. Kaufman, A. Alm, Adler’s Physiology of the Eye, 11th ed. (2011)
A. Oishi, M. Mandai, A. Nishida, M. Hata, T. Matsuki, Y. Kurimoto, Eur. J. Ophthalmol. 21, 777 (2011)
M. Pancholy, P.P. Storey, H.J. Levin, A. Obeid, S.N. Patel, B. Kuley, J. Hsu, M.J. Spirn, M. Fineman, M.A. Klufas, O. Gupta, A.C. Ho, S.J. Garg, Curr. Eye Res. 46, 1370 (2021)
V.T. Papastavrou, H. Zambarakji, I. Dooley, H. Eleftheriadis, T.L. Jackson, Retin. Cases Br. Reports 11, 44 (2017)
A. Reiner, M.E.C. Fitzgerald, N. Del Mar, C. Li, Prog. Retin. Eye Res. 64, 96 (2018)
K.A. Rezaei, J.C. Wen, MedEdPORTAL 12, 10502 (2016)
M.M. Sachdeva, A. Moshiri, H.A. Leder, A.W. Scott, J. Ophthalmic Inflamm. Infect. (2016)
H. Selvan, A. Pujari, A. Kishan, A.K. Behera, T. Sidhu, V. Gupta, T. Dada, R. Sihota, Curr. Eye Res. 46, 78 (2021)
S. Sen, C. Mishra, N.B. Kannan, K. Ramasamy, G. Rameshkumar, P. Lalitha, Semin Ophthalmol. 36, 413 (2021)
A. Sudhalkar, P. Chauhan, A. Sudhalkar, R.H. Trivedi, Ophthalmology 119, 198 (2012)
C. Wang, H. Hou, K. Nan, M.J. Sailor, W.R. Freeman, L. Cheng, Exp. Eye Res. 129, 74 (2014)
W.L. Wong, X. Su, X. Li, C.M.G. Cheung, R. Klein, C.Y. Cheng, T.Y. Wong, Lancet Glob. Heal. 2, e106 (2014)
L. Xu, T. Lu, L. Tuomi, N. Jumbe, J. Lu, S. Eppler, P. Kuebler, L.A. Damico-Beyer, A. Joshi, Investig Ophthalmol. Vis. Sci. 54, 1616 (2013)
N.A. Yannuzzi, N.Z. Gregori, P.J. Rosenfeld, N. Relhan, N.A. Patel, N. Si, D. Miller, S.R. Dubovy, W.E. Smiddy, S.G. Schwartz, Ophthalmic Surgery, Lasers Imaging Retin. 49, 313 (2018)
J. Zeroni, J. Slager, R. Hegenrother, T. Kloke, S. Varner, Invest. Ophthalmol. Vis. Sci. 48, 5805 (2007)
Acknowledgements
The author acknowledge the efforts and support of Mr. Mateen Amin, Focus Ophthalmics LLC, USA for design modifications and manufacturing of the prototypes and Mr. Tapas Kumar Roy, PhD student in pharmacology who assisted in the procedures and performed fundus imaging. The authors would also like to acknowledge Ms. Lata Kaushik for performing the initial ELISA experiments and in drafting the design as per the instructions of the manufacturer.
Funding
The authors would like to acknowledge the funding agencies, IRD, IIT Delhi and AIIMS for partial financial support to the project via multi-institutional project:MI01884 (IITD) /A-05 (AIIMS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest/Competing Interests
The authors declare that there are no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chawla, R., Bhattacharyya, J., Moksha, L. et al. A novel, minimally invasive implant to assist in repeated intraocular drug delivery. Biomed Microdevices 24, 17 (2022). https://doi.org/10.1007/s10544-022-00618-y
Accepted:
Published:
DOI: https://doi.org/10.1007/s10544-022-00618-y