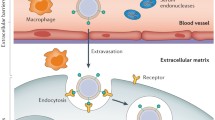
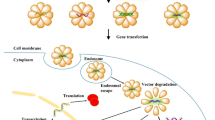
Abstract
Gene therapy has broad prospects as an effective treatment for some cancers and hereditary diseases. However, DNA and siRNA are easily degraded in vivo because of their biological activities as macromolecules, and they need the effective transmembrane delivery carrier Selecting the appropriate carrier for delivery will allow nucleic acid molecules to reach their site of action and enhance delivery efficiency. Currently used nucleic acid delivery vectors can be divided into two major categories: viral and non-viral vectors. Viral carrier transport efficiency is high, but there are safety issues. Non-viral vectors have attracted attention because of their advantages such as low immunogenicity, easy production, and non-tumorigenicity. The construction of safe, effective, and controllable vectors is the focus of current gene therapy research. This review presents the current types of nucleic acid delivery vehicles, which focuses on comparing their respective advantages and limitations, and proposes a novel delivery system, RNTs, a novel nanomolecular material, introducing the characteristics and nucleic acid delivery process of RNTs and their latest applications.






Similar content being viewed by others
References
A. Bianco, K. Kostarelos, M. Prato, Applications of carbon nanotubes in drug delivery. Curr. Opin. Chem. Biol. 9, 674–679 (2005)
A.U. Bielinska, J.F. Kukowska-Latallo, J.R. Baker, The interaction of plasmid DNA with polyamidoamine dendrimers: Mechanism of complex formation and analysis of alterations induced in nuclease sensitivity and transcriptional activity of the complexed DNA. Gene Structure and Expression 1353(2), 180–190 (1997)
K. Chamberlain, J.M. Riyad, T. Weber, et al., Cardiac gene therapy with adeno-associated, virus-based vectors. Curr. Opin. Cardiol. 4, 140–152 (2017)
Y. Chen, B. Bilgen, et al., Self-Assembled Rosette Nanotube/Hydrogel Composites For Cartilage Tissue Engineering. Tissue Engineering: Part C 16(6) (2010)
Y. Chen, S. Song, Z. Yan, et al., Self-assembled rosette nanotubes encapsulate and slowly release dexamethasone. Int. J. Nanomedicine 6, 1035–1044 (2011)
Z. Chen, T. Zhang, B. Wu, X. Zhang, Insights into the therapeutic potential of hypoxia-inducible factor-1α small interfering rNa in malignant melanoma delivered via folate-decorated cationic liposomes. Int. J. Nanomedicine 11, 991–1002 (2016)
J.W. Choi, J.S. Lee, S.W. Kim, et al., Evolution of oncolytic adenovirus for cancer treatment. Adv. Drug Deliv. Rev. 64, 720–729 (2012)
D.T. Curiel, J.T. Douglas, Adenoviral vectors for gene therapy (Academic Press, San Diego, 2002), pp. 329–348
M.E. Davis, Non-viral gene delivery systems. Curr OpinChem Biol 13, 28–131 (2002)
J. Dobson, Gene therapy progress and prospects: Magnetic nanoparticle-based gene delivery. Gene Ther. 13, 283–287 (2006)
K. Ewert, A. Ahmad, H.M. Evans, et al., Efficient synthesis and cell-transfection properties of a new multivalent cationic lipid for nonviral gene delivery. J. Med. Chem. 45, 5023–5029 (2002)
Y.X. Fan, Z.X. Liang, Q.Z. Liu, et al., Cell penetrating peptide of sodium-iodide symporter effect on the I-131 radiotherapy on thyroid cancer. Exp Ther Med 13(3), 989–994 (2017)
P.L. Felgner, T.R. Gadek, M. Holm, et al., Lipofection: A highly efficient, lipid-mediated DNA-transfection procedure. Proc. Natl. Acad. Sci. U. S. A. 84, 7413–7417 (1987)
H. Fenniri, P. Mathivanan, K.L. Vidale, et al., Helical rosette nano-tubes: Design, self-assembly and characterization. J. Am. Chem. Soc. 123(16), 3854–3855 (2001)
X. Gao, L. Huang, A novel cationic liposome reagent for efficient transfection of mammaliam cells. Biochem. Biophys. Res. Commun. 179, 280–285 (1991)
D.J. Gary, N. Puri, Y.Y. Won, Polymer-based si RNA delivery: Perspectives on the fundamental and phenomenological distinctions from polymer-based DNA delivery. J. Control. Release 121, 64–73 (2007)
D.A. Giljohann, D.S. Seferos, A.E. Prigodich, et al., Gene regulation with polyvalent si RNA-nanoparticle conjugates. Am. Chem. Soc. 131, 2072–2073 (2009)
I.M. Hafez, N. Maurer, P.R. Cullis, On the mechanism whereby cationic lipids promote intracellular delivery of polynucleic acids. Gene Ther. 8, 1188–1196 (2001)
K.M. Hui, P.T. Ang, L. Huang, et al., Phase I study of immunotherapy of cutaneous metastases of human carcinoma using allogeneic and xenogeneic MHC DNA-liposome complexes. Gene Ther. 4(8), 783–790 (1997)
S. Hu-Lieskovan, J.D. Heidel, D.W. Bartlett, Ea ta. Triche, sequence-specific knockdown of EWS-FLI1 by targeted, nonviral delivery of small interfering RNA inhibits tumor growth in a murine model of metastatic ewing's sarcoma. Cancer Res. 65(19), 8984–8992 (2005)
Q. Jiang, Y. Zhang, R. Zhuo, et al., Supramolecular host-guest polycationic gene delivery system based on poly (cyclodextrin) and azobenzene-terminated polycations. Colloids Surf. B: Biointerfaces 1(147), 25–35 (2016)
W.S. Journeay, S.S. Suri, J.G. Moralez, et al., Rosette nanotubes show low acute pulmonary toxicity in vivo. Int. J. Nanomedicine 3(3), 373–384 (2008a)
W.S. Journeay, S.S. Suri, J.G. Moralez, et al., Rosette nano-tubes show low acute pulmonary toxicity in vivo. Int. J. Nanomedicine 3(3), 373–383 (2008b)
W.S. Journeay, S.S. Suri, J.G. Moralez, et al., Low infamma-tory activation by self-assembling rosette nanotubes in human Calu-3 pulmonary epithelial cells. Small. 4(6), 817–823 (2008c)
K.J. Kauffman, M.J. Webber, D.G. Anderson, J. Control. Release 240, 227–234 (2016)
C.M. Lai, Y.K. Lai, P.E. Rakoczy, Adenovirus and adeno-associated virus vectors. DNA Cell Biol. 21, 895–913 (2002)
C. Le Guiner, L. Servais, M. Montus, et al., Long-term microdystrophin gene therapy is effective in a canine model of Duchenne muscular dystrophy. Nat. Commun. 8, 161–165 (2017)
C.H. Lee, Y.H. Ni, C.C. Chen, et al., Synergistic effect ofpolyethylenimine and cationic liposomes in nucleic acid de-livery to human cancer cells. Biochim. Biophys. Acta 1611, 55–62 (2003)
Y.L. Li, X. Duan, L.H. Jing, et al., Quantum dot-antisense oligonucleotide conjugates for multifunctional gene transfection, mRNA regulation, and tracking of biological processes. Biomaterials 32, 1923–1931 (2011)
B. Li, W. Guo, F. Zhang, et al., Synthesis and evaluation of L-arabinose-based cationic glycolipids as effective vectors for pDNA and siRNA in vitro. PLoS One 12(7), 180–276 (2017)
W. Li, Y. Liu, J. Du, et al., Cell penetrating peptide-based polyplexes shelled with polysaccharide to improve stability and gene transfection. Nanoscale 7(18), 8476–8488 (2015)
W.J. Li, F.C. Szoka, Lipid-based nanoparticles for nucleic acid delivery. Pharm. Res. 24, 438–449 (2007)
P. Li, R. Zhang, Y. Gan, et al., Effects of osteogenic protein-1 on intervertebral disc regeneration: A systematic review of animal studies. Biomed. Pharmacother. 88, 260–266 (2017)
A. Lieber, C.Y. He, L. Meuse, et al., The role of Kupffer cell activation and viral gene expression in early liver toxicity after infusion of recombination adenovirus vectors. J. Virol. 71, 8798–8807 (1997)
Z. Medarova, W. Pham, C. Farrar, et al., In vivo imaging of si RNA delivery and silencing in tumors. Nat. Med. 13, 372–377 (2007)
Z. Meng, J. O’Keeffe-Ahern, J. Lyu, et al., A new developing class of gene delivery: Messenger RNA-based. Biomater. Sci. (2017) https://doi.org/10.1039/C7BM00712D
L. Mi-Kyung, C. Soo-Kyung, C. Woo-Jeong, et al., The use ofchitosan as a condensing agent to enhance emulsion-mediatedgene transfer. Biomaterials 26, 2147–2156 (2005)
M.S. Mogensen, K. Scheibye-Alsing, P. Karlskov-Mortensen, et al., Validation of Genome-wide Intervertebral Disk Calcification Associations in Dachshund and Further Investigation of the Chromosome 12 Susceptibility Locus. Front. Genet. 3, 225 (2012)
N.A. Moore, P. Bracha, R.M. Hussain, et al., Gene therapy for age-related macular degeneration. Expert. Opin. Biol. Ther. 20, 1–10 (2017)
E. Ojeda, G. Puras, M. Agirre, J. Zarate, S. Grijalvo, R. Eritja, G. Martinez-Navarrete, C. Soto-Sánchez, A. Diaz-Tahoces, M. Aviles-Trigueros, E. Fernández, J.L. Pedraz, The influence of the polar head-group of synthetic cationic lipidson the transfection efficiency mediated by niosomes in rat retina and brain. Biomaterials 77, 267–279 (2016)
P.C. Patel, D.A. Giljohann, D.S. Seferos, et al., Peptide antisense nanoparticles. Proc. Natl. Acad. Sci. U. S. A. 105, 17222–17226 (2008)
H. Peng, H. Yang, L. Song, et al., Sustained delivery of siRNA/PEI complex from in situ forming hydrogels potently inhibits the proliferation of gastric cancer. Exp Clin Cancer Res 31, 35–57 (2016)
E.M. Pridgen, R. Langer, O.C. Farokhzad, Biodegradable, polymeric nanoparticle delivery systems for cancer therapy. Nanomed 2, 669–680 (2007)
W. Qiu, S.L. Li, W.L. Deng, et al., Strain sensor of carbon nanotubes in microscale:From model to metrology. Sci. World J. 2014, 406154 (2014)
M. Rodriguez, J. Lapierre, C.R. Ojha, et al., Intranasal drug delivery of small interfering RNA targeting Beclin1 encapsulated with polyethylenimine (PEI) in mouse brain to achieve HIV attenuation. Sci. Rep. 7(1), 18–62 (2017)
J.A. Roth, R.J. Cristiano, Gene therapy for cancer: What have we done and where are we going? Natl Cancer Inst 89, 21–39 (1997)
I.I. Slowing, J.L. Vivero-Escoto, C.W. Wu, et al., Mesoporous silica nanoparticles as controlled release drug delivery and gene transfection carriers. Adv. Drug Deliv. Rev. 60, 1278–1288 (2008)
J. Tao, W.-F. Ding, X.-H. Che, Y.-C. Chen, F. Chen, X.-D. Chen, X.-L. Ye, S.-B. Xiong, Optimization of a cationic liposome-based gene delivery system for the application of miR-145 in anticancer therapeutics. Int. J. Mol. Med. 37, 1345–1354 (2016)
D. Villaret, B. Glisson, D. Kenady, et al., A multicenter phase II study of tg DCC-E1A for the intratumoral treatment of patients with recurrent head and neck squamous cell carcinoma. Head Neck 24(7), 661–669 (2002)
Y. Wan, W. Dai, R.J. Nevagi, et al., Multifunctional peptide-lipid nanocomplexes for efficient targeted delivery of DNA and siRNA into breast cancer cells. Acta Biomater. 1(59), 257–268 (2017)
F. Wang, L. Gao, L.-Y. Meng, A neutralized noncharged polyethylenimine-Based system for efficient delivery of siRNA into heart without toxicity. Mater. Interfaces 8(49), 33529–33538 (2016)
T. Yoshizawa, Y. Hattori, M. Hakoshima, et al., Folate-linked lipid-based nanoparticles for synthetic si RNA delivery in KB tumor xenografts. Pharm Biopharm 70, 718–725 (2008)
J. Zheng, J. Yang, K. Zhao, et al., Low expression of microRNA-143 is related to degenerative scoliosis possibly by regulation of cyclooxygenase-2 expression. Int. J. Clin. Exp. Med. 8(3), 4140–4145 (2015)
X. Zhou, L. Chen, S. Grad, et al., The roles and perspectives of microRNAs as biomarkers for intervertebral disc degeneration. J. Tissue Eng. Regen. Med. (2017)
Acknowledgments
The authors thank Guo Xiong and Zhang Feng for reviewing the manuscript. We thank M. Arico from Liwen Bianji, Edanz Group China (www.liwenbianji.cn/ac), for editing the English text of a draft of this manuscript. This review was supported by grants from the National Nature Science Foundation of China (No. 81171761), Shaanxi Natural Science Foundation (No. 2017SF-088), and Shaanxi Provincial Department of Education Fund (No: 18JK0667). The funding sources had no role in this publication.
Author information
Authors and Affiliations
Contributions
Y.J. and Z.L.X. were responsible for study conception and design, acquisition of data, analysis and interpretation of data, and drafting the manuscript. L.J.Z. critically revised the manuscript for important intellectual content. H.J. and B.X. were responsible for the analysis and interpretation of data. S.F. was responsible for study conception and design, acquisition, analysis and interpretation of data, and critical revision of the manuscript for important intellectual content. All authors read and approved the final version of the manuscript and had full access to all of the data in the study.
Corresponding author
Ethics declarations
Conflict of interest
There are no financial or personal relations with other people or organizations, which could inappropriately bias this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiao, Y., Xia, Z.L., Ze, L.J. et al. Research Progress of nucleic acid delivery vectors for gene therapy. Biomed Microdevices 22, 16 (2020). https://doi.org/10.1007/s10544-020-0469-7
Published:
DOI: https://doi.org/10.1007/s10544-020-0469-7