Abstract
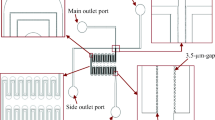
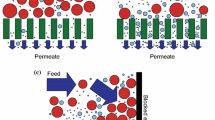
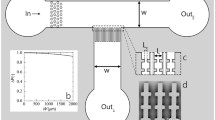
Steady state crossflow microfiltration (CMF) is an important and often necessary means of particle separation and concentration for both industrial and biomedical processes. The factors controlling the performance of CMF have been extensively reviewed. A major factor is transmembrane pressure (TMP). Because microchannels have small height, they tend to have high pressure gradients in the feed-flow direction. In the extreme, these gradients may even reverse the pressure across the membrane (inciting backflow). It is therefore desirable to compensate for the effect of feed-flow on the TMP, aiming at constant transmembrane pressure (cTMP) at a value which maximizes filtrate flux. This is especially critical during filtration of deformable particles (e.g. erythrocytes) through low intrinsic resistance membranes. Filtration flux is generally taken to be directly proportional to TMP, with pressure drop along the channel decreasing in the flow direction. A co-current flow of filtrate in a suitably designed filtrate collecting channel is shown to allow the TMP to remain constant and permit the sieving surface to perform optimally, permitting up to twice as much filtration over that of a naïve configuration. Manipulation of the filtrate channel may be even more beneficial if it prevents backflow that might otherwise occur at the end of a sufficiently long channel. Experiments with erythrocyte suspensions, reported here, validate these concepts.





Similar content being viewed by others
References
L.I. Amar, D. Guisado, M. Faria, et al., Erythrocyte fouling on micro-engineered membranes. Biomed. Microdevices 20, 55 (2018)
Bird RB, Stewart WE, Lightfoot EN. Transport phenomena: Wiley; 2007
C. Charcosset, Membrane processes in biotechnology: An overview. Biotechnol. Adv. 24, 482–492 (2006)
M. Dickson, L. Amar, M. Hill, J. Schwartz, E. Leonard, A scalable, micropore, platelet rich plasma separation device. Biomed. Microdevices 14, 1095–1102 (2012)
D.A. Drew, J.A. Schonberg, G. Belfort, Lateral inertial migration of a small sphere in fast laminar flow through a membrane duct. Chem. Eng. Sci. 46, 3219–3224 (1991)
R.W. Field, D. Wu, J.A. Howell, B.B. Gupta, Critical flux concept for microfiltration fouling. J. Membr. Sci. 100, 259–272 (1995)
H.M. Ji, V. Samper, Y. Chen, C.K. Heng, T.M. Lim, L. Yobas, Silicon-based microfilters for whole blood cell separation. Biomed. Microdevices 10, 251–257 (2008)
J. Kromkamp, A. Bastiaanse, J. Swarts, G. Brans, R.G.M. van der Sman, R.M.A. Boom, Suspension flow model for hydrodynamics and concentration polarisation in crossflow microfiltration. J. Membr. Sci. 253, 67–79 (2005)
E.F. Leonard, C.S. Vassilieff, The deposition of rejected matter in membrane separation processes. Chem. Eng. Commun. 30, 209–217 (1984)
M.R. Mackley, N.E. Sherman, Cross-flow cake filtration mechanisms and kinetics. Chem. Eng. Sci. 47, 3067–3084 (1992)
M.S. Maria, B. Kumar, T. Chandra, A. Sen, Development of a microfluidic device for cell concentration and blood cell-plasma separation. Biomed. Microdevices 17, 115 (2015)
R.O. Rodrigues, D. Pinho, V. Faustino, Lima R. A simple microfluidic device for the deformability assessment of blood cells in a continuous flow. Biomed. Microdevices 17, 108 (2015)
N. Rossignol, L. Vandanjon, P. Jaouen, F. Quemeneur, Membrane technology for the continuous separation microalgae/culture medium: Compared performances of cross-flow microfiltration and ultrafiltration. Aquac. Eng. 20, 191–208 (1999)
C.J. van Rijn, Nano and micro engineered membrane technology (Elsevier, 2004)
C.J. van Rijn, W. Nijdam, S. Kuiper, G.J. Veldhuis, H. van Wolferen, M. Elwenspoek, Microsieves made with laser interference lithography for micro-filtration applications. J. Micromech. Microeng. 9, 170 (1999)
N.A. Wagdare, A.T. Marcelis, O.B. Ho, R.M. Boom, C.J. van Rijn, High throughput vegetable oil-in-water emulsification with a high porosity micro-engineered membrane. J. Membr. Sci. 347, 1–7 (2010)
N. Weeranoppanant, L. Amar, E. Tong, M. Faria, M.I. Hill, E.F. Leonard, Modeling of fouling in cross-flow microfiltration of suspensions. AICHE J. (2018)
A.L. Zydney, C.K. Colton, A concentration polarization model for the filtrate flux in cross-flow microfiltration of particulate suspensions. Chem. Eng. Commun. 47, 1–21 (1986)
Acknowledgements
Support for this work was provided in part by Grant 1R21HL088162 from the National Institute of Health, and Vizio Medical Devices, LLC. The authors also thank Columbia Medical Center Blood Bank and blood donors. We acknowledge gratefully the assistance of Dr. Robert von Gutfeld as well as our whole medical team, most especially the late Dr. James Jones.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Topical area: Microfluidics, Separations: Materials, Devices and Processes, Artificial Organs.
Appendix
Appendix
1.1 Nomenclature
- B :
-
Half height of the channel (m)
- L :
-
Channel Length (m)
- W :
-
Channel width (m)
- ΔP :
-
Pressure drop across the channel (Torr)
- Q f :
-
Volumetric flowrate of the permeate (i.e. Filtration rate) (cm3/min)
- Q m :
-
Volumetric flowrate in main channel (cm3/min)
- TMP :
-
Transmembrane pressure (Torr)
- J f :
-
Filtrate flux (cm3/cm2-min)
- P 1 :
-
Inlet blood pressure (Torr)
- P 2 :
-
Outlet blood pressure (Torr)
- P 3 :
-
Inlet plasma pressure (Torr)
- P 4 :
-
Outlet plasma pressure (Torr)
Greek symbols
- γw:
-
Nominal wall shear rate (1/s)
- τw:
-
Wall shear stress (Pa)
- μ :
-
Viscosity of media (Poise)
1.2 Supporting equations
1.2.1 Microchannel height
As in a previous study (Amar et al. 2018), the thickness of each microchannel varied each time the apparatus was assembled and had to be accurately calculated each time. By measuring the change in pressure for various through-flow rates for a fluid of known viscosity (i.e. microfiltered water), the thickness (2B) of the channel was calculated using the equation for laminar flow in a narrow slit solved for B (Bird 2007):
The flow was assumed to be Newtonian, laminar, and fully developed, where Qm is the volumetric flow rate, ΔP is the difference in pressure between inlet and outlet, W and L are the width and length of the channel, respectively, and μ is the viscosity of the fluid (Bird 2007).
Since the heights above and below the microsieves were not equal, different flows above and below the sieves were required to create the same pressure drop. A simplified version of the slit flow equation (Bird 2007) was used:
The factor 3/2 compensates for the viscosity difference between the two fluids.
1.2.2 Shear rate and shear stress at the wall
The shear stress exerted at the flow boundaries, τw can be calculated by balancing the shear force at the wall against the pressure gradient for a slit channel (Bird 2007).
Shear rates (\( \dot{\gamma}=\frac{dV}{dZ} \)) at the wall are found as the shear stress (τw) divided by the viscosity:
where Qm is the volumetric flow rate, B is the half thickness of the channel, W is the width of the channel, and μ is the viscosity of the fluid (Bird 2007).
Rights and permissions
About this article
Cite this article
Amar, L.I., Hill, M.I., Faria, M. et al. Co-current crossflow microfiltration in a microchannel. Biomed Microdevices 21, 12 (2019). https://doi.org/10.1007/s10544-019-0362-4
Published:
DOI: https://doi.org/10.1007/s10544-019-0362-4