Abstract
Evolutionary gradualism, the randomness of mutations, and the hypothesis that natural selection exerts a pervasive and substantial influence on evolutionary outcomes are pair-wise logically independent. Can the claims about selection and mutation be used to formulate an argument for gradualism? In his Genetical Theory of Natural Selection, R.A. Fisher made an important start at this project in his famous “geometric argument” by showing that a random mutation that has a smaller effect on two or more phenotypes will have a higher average fitness than a random mutation that has a larger phenotypic effect. Motoo Kimura’s demonstration that a gene’s probability of fixation depends on both the selection coefficient and the effective population size shows that Fisher’s argument for gradualism was mistaken. Here we analyze Fisher’s argument and explain how Kimura’s theory leads to a conclusion that Fisher did not anticipate. We identify a fallacy that reasoning about fitness differences and their evolutionary consequences should avoid. We then distinguish forward-directed from backward-directed versions of gradualism. The backward-directed thesis about a single mutation may be correct, but the forward-directed thesis is not. After that we consider a sequence of random mutations that all affect the same cluster of phenotypes and Allen Orr’s idea that there is an optimal sequence of mutation sizes, moving from larger to smaller as the population approaches a fixed optimum. This provides a likelihood justification for a forward-directed version of gradualism when there is a fixed optimum, but it also provides an argument against forward-directed gradualism if the optimum shifts substantially as the population evolves. Finally, we consider whether genome-wide association studies (GWASs) can furnish empirical evidence that bears on the truth of gradualism. The version of gradualism that GWASs directly bear on concerns the phenotypic effect size of a gene after it arises by mutation and has reached an appreciable frequency, not the phenotypic size of the mutation event itself.





Similar content being viewed by others
Notes
This formulation will be refined later in the paper. Note that the gradualism discussed here is not about rates of evolution. Thus it is not in conflict with the punctuated equilibrium thesis defended by Eldredge and Gould (1972).
Here we set aside the fact that Fisher assumes that that fitness is linearly related to the phenotypic distance from the organism’s present state to the optimum, and the fact that Fisher considers only a single optimum, rather than taking account of the possibility that there are multiple adaptive peaks (Orr 1998).
GWASs don’t allow you to measure the prior probabilities described in Odds. GWASs examine mutations that have managed to evolve to measurable frequencies, not the frequencies of mutations of various sizes, many of which may promptly go extinct.
Fisher (1930) uses d/2 as the diameter of the circle centered on the optimum, which simplifies the mathematics slightly, but at the cost of clarity.
References
Boyle EA, Li YI, Pritchard JK (2017) An expanded view of complex traits: from polygenic to omnigenic. Cell 169(7):1177–1186
Clarke B (1971) Natural selection and the evolution of proteins. Nature 232:487
Darwin C (1959) The Autobiography of Charles Darwin: With original omissions restored, edited with appendix and notes by his grand-daughter, Nora Barlow. Harcourt, Brace.
Darwin C (1868) The variation of animals and plants under domestication. Murray, London, p 2
Darwin, C. (1958). Autobiography, N. Barlow (ed.), London: Collins.
Edwards A (1972) Likelihood. Cambridge University Press
Eldredge N, Gould SJ (1972) Punctuated equilibria: an alternative to phyletic gradualism. Models Paleobiol 1972:82–115
Fisher RA (1930) The genetical theory of natural selection. Oxford University Press
Hacking I (1965) The logic of statistical inference. Cambridge University Press
Hartl DL, Taubes CH (1996) Compensatory nearly neutral mutations: selection without adaptation. J Theor Biol 182(3):303–309
Kimura M (1957) Some problems of stochastic processes in genetics. Ann Math Stat 28:882–901
Kimura M (1962) On the probability of fixation of mutant genes in a population. Genetics 47:713–719
Kimura M (1983) The neutral theory of molecular evolution. Cambridge University Press
Li S (2011) Concise formulas for the area and volume of a hyperspherical cap. Asian J Math Stat 4(1):66–70
Loos RJF et al (2013) Identification of heart rate-associated loci and their effects on cardiac conduction and rhythm disorders. Nat Genet 45(6):621–631. https://doi.org/10.1038/ng.2610
López-Cortegano E, Caballero A (2019) GWEHS: a genome-wide effect sizes and heritability screener. Genes 10(8):558
Manolio TA, Collins FS, Cox NJ, Goldstein DB, Hindorff LA, Hunter DJ, Visscher PM (2009) Finding the missing heritability of complex diseases. Nature 461(7265):747–753
Marouli E, Graff M, Medina-Gomez C et al (2017) Rare and low-frequency coding variants alter human adult height. Nature 542:186–190
Orr HA (1998) The population genetics of adaptation: the distribution of factors fixed during adaptive evolution. Evolution 52(4):935–949
Orr HA (2000) Adaptation and the cost of complexity. Evolution 54(1):13–20
Orr HA (2009) Darwin and darwinism: the (alleged) social implications of The Origin of Species. Genetics 183(3):767–772
Orr HA, Coyne JA (1992) The genetics of adaptation: a reassessment. Am Nat 140(5):725–742
Park JH, Wacholder S, Gail MH, Peters U, Jacobs KB, Chanock SJ, Chatterjee N (2010) Estimation of effect size distribution from genome-wide association studies and implications for future discoveries. Nat Genet 42(7):570–575
Royall R (1997) Statistical evidence–a likelihood Paradigm. Chapman and Hall
Simons YB, Bullaughey K, Hudson RR, Sella G (2018) A population genetic interpretation of GWAS findings for human quantitative traits. PLoS Biol 16(3):e2002985
Sober E (2008) Evidence and evolution. Cambridge University Press
Sober E (2015) Ockham’s Razors–a user’s manual. Cambridge University Press
Stoltzfus A (2017) Why we don’t want another “synthesis.” Biol Direct 12(1):1–12
Van Valen L (1973) A new evolutionary law. Evolutionary Theor 1:1–30
Visscher PM, McEvoy B, Yang J (2010) From Galton to GWAS: quantitative genetics of human height. Genet Res 92(5–6):371–379
Acknowledgements
We are grateful to AA, DB, BB, HC, JF, ML, BP, MS, MS, BV, and DW for useful discussion. We also thank the anonymous referee of this journal for helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1: the probability that a random mutation will improve fitness
In Fig. 1, let r be the radius of a circle centered on X and d be the radius of the circle centered on the optimum.Footnote 5 We use the law of cosines to find w, the distance from the phenotype resulting from \(\theta\) to the optimum:
As we want to know the probability that this mutation is advantageous (\(w<d\)) and given that all values are positive distances,
Rearranging and cancelling terms we find,
Taking the arccosine to find the angles,
As both the positive and negative angles are equal, the total angle will be \(2\mathrm{arccos}\left(\frac{r}{2d}\right)\). Dividing by the full circle, \(2\pi ,\) we get the proportion of the full circle which improves fitness,
Note that when \(r>2d\), there will be no solutions to this formula. In this case the circle centered on X will fully encompass the circle centered at the optimum, and thus all mutations will decrease fitness.
Appendix 2: the expected degree of improvement, given that the mutation improves fitness
We begin again with the law of cosines, where the mutation is of size r, but this time we solve for w, the fitness distance resulting from a mutation \(\theta\).
The improvement (decrease in fitness distance), \(\Delta w\), from mutation \(\theta\) will be equal to \(d-w\). Thus,
To find the expected improvement given that the mutation improves fitness we integrate over the \(\theta\) resulting in advantage and average over all \(\theta\) resulting in advantage. For the full circle we would integrate from/to \(\pm \mathrm{arccos}\left(\frac{r}{2d}\right),\) but as both the positive and negative portions are equal and we are only concerned with proportion, we simply integrate from 0 to the positive angle and divide by \(\mathrm{arccos}\left(\frac{r}{2d}\right)\) rather than \(2\mathrm{arccos}\left(\frac{r}{2d}\right)\).
Figure 6 shows the ratio of the expected improvement in fitness for a big mutation of size (0.1)f and the expected improvement in fitness of a small mutation of size 0.1, where f > 1. As can be seen, the ratio is not always greater than 1. The ratio begins to drop off when the big mutation is larger than d and begins to “overshoot” the optimum. Eventually, this overshooting becomes so deleterious that small is more advantageous than big.
Appendix 3: a random mutation’s expected improvement in fitness
To find the expected fitness from a random mutation, we take the average of all possible mutations of size r. This is done by first integrating over all possible \(\theta\) (0 to \(2\pi\)) and then dividing by the full circle (\(2\pi\)):
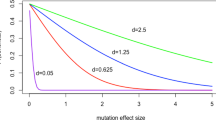
This is shown in Fig.
3.
Appendix 4: the full formula for (area of spherical cap)/(area of sphere) for an n-dimensional sphere
We begin with the formula for a spherical cap and sphere (Li 2011). n is the number of dimensions, r is the radius, and I the regularized incomplete beta function.
We divide the formula for the area of the spherical cap by the area of the full n-sphere to find the proportion of the sphere covered by the cap:
The areas of the sphere, \({A}_{n}\left(r\right)\) cancel, leaving us with:
Using trigonometric identities and the results from Appendix 1, we find, \({\mathrm{sin}}^{2}\Phi\):
Thus our final result for the probability of an advantageous mutation in n-dimensions is:
As in Appendix 1, this formula will not be valid for \(r>2d\). At \(r>2d\) all \(\uptheta\) will take the organism further from the optimum (and thus be disadvantageous). Hartl and Taubes (1996) arrive at the same result, but express it as a fraction of integrals:
Appendix 5: Probability of fixation for a mutation of size r
The selection coefficient, s, for a mutation B is given by:
where WA is the fitness of the wild type and WB the fitness of the mutant. From Appendix 2, let the fitness distance to optimum of the wild type be 1, and \(\mathrm{w}\) be the distance to optimum for the mutant. Halving the distance to optimum doubles the fitness, reducing distance by 2/3 triples it, and so on, thus:
Choosing the wild type as our reference, we let \({\mathrm{W}}_{\mathrm{A}}=1\), thus:
Substituting these values into the definition of s:
Substituting for w by the formula from Appendix 2:
The probability of fixation for a randomly mating diploid population of size \({N}_{e}\) is given by Kimura (1962):
Substituting the above value for s into Kimura’s formula and using the ‘exp’ notation for clarity [\(\mathrm{exp}\left(\mathrm{x}\right)={\mathrm{e}}^{\mathrm{x}}\)],
To find the expected probability of fixation for a random mutation of size r, we integrate the probability densities from the above formula over all mutations \(\uptheta\) and divide by all possible mutations (effectively summing the probability densities for all possible mutations and dividing by the number of possible mutations to find an average probability):
Letting \({\mathrm{N}}_{\mathrm{e}}=10\), we plot this curve in Fig.
7. This plot is valid for all \(\mathrm{r}\ge 0\). The greatest probability of fixation for occurs when \(r=0.7274\). Thus a mutation smaller than or larger than \(r=0.7274\) will have a lower average probability of fixation. This value is remarkably stable, going to 0.7304 as \({\mathrm{N}}_{\mathrm{e}}\to \infty\). Thus smaller mutations do not have a higher average probability of fixation than bigger mutations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maxwell, M.J., Sober, E. Gradualism, natural selection, and the randomness of mutation–fisher, Kimura, and Orr, connecting the dots. Biol Philos 38, 16 (2023). https://doi.org/10.1007/s10539-023-09904-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10539-023-09904-2