Abstract
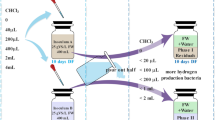
Manipueira is a carbohydrate-rich agro-industrial waste from cassava processing. It is considered well suitable for biotechnological processes, such as hydrogen and carboxylic acids production, due to the high content of easily degradable organic matter. However, the proper methanogenesis inhibition method, inoculum type, and organic loads are factors still limiting the processes. The objective in this work was to evaluate the effects of such factors on byproducts production in anaerobic reactors. Batch experiments were conducted with 2.3-L flasks during two operational phases. In the first phase (P1), inhibition of methanogens in the sludge was evaluated using acetylene (1% v/v of headspace) and heat treatment (120 °C, 1 atm for 30 min). In the second phase (P2), three inoculum types obtained from common anaerobic sludges (bovine rumen and sludges from municipal and textile industrial wastewater treatment plants) were individually assayed. P2 aimed to identify the best inoculum, based on hydrogen production ability, which was tested for three initial concentrations of manipueira in terms of chemical oxygen demand (COD) (10, 20 and 40 g O2/L). Results of P1 indicated that either acetylene or heat treatment efficiently inhibited methanogenesis, with no methane production. However, the maximum H2 production potential by applying heat treatment (~ 563 mL) was more than twice compared with that by acetylene treatment (~ 257 mL); and butyrate was the main carboxylic acid by-product (~ 3 g/L). In P2 experiments after sludge heat treatment, the highest hydrogen yield (1.66 ± 0.07 mol H2/mol glucose) and caproic acid production (~ 2 g/L) were observed at 20 g O2/L of manipueira COD, when bovine rumen was the inoculum. The primary metabolic degradation products in all P2 experiments were ethanol, acetic, butyric, propionic and caproic acids. The finding of caproic acid detection indicated that the applied conditions in manipueira anaerobic degradation favored carbon chain elongation over methanogenesis.







Similar content being viewed by others
References
Agler MT, Wrenn BA, Zinder SH, Angenent LT (2011) Waste to bioproduct conversion with undefined mixed cultures: the carboxilate platform. Trends Biotechnol 29(2):70–78. https://doi.org/10.1016/j.tibtech.2010.11.006
Angenent LT, Karim K, Al-Dahhan MH, Wrenn BA, Domíguez-Espinosa R (2004) Production of bioenergy and biochemicals from industrial and agricultural wastewater. Trends Biotechnol 22(9):477–485. https://doi.org/10.1016/j.tibtech.2004.07.001
APHA (2012) Standard methods for the examination for water and wastewater, 22nd edn. Washington, DC, American Public Health Association/American Water Works Association/Water Environmental Federation
Archer DB, Kirsop BH (1990) The microbiology and control of anaerobic digestion. In: Wheatley A (ed) Anaerobic digestion: a waste treatment technology. Elsevier Applied Science, London, pp 43–91
Cappelletti BM, Reginatto V, Amante ER, Antônio RV (2011) Fermentative production of hydrogen from cassava processing wastewater by Clostridium acetobutylicum. Renew Energy 36(12):3367–3372. https://doi.org/10.1016/j.renene.2011.05.015
Chang J-J, Lin J-J, Ho C-Y, Chin W-C, Huang C-C (2010) Establishment of rumen-mimic bacterial consortia: a functional union for bio-hydrogen production from cellulosic bioresource. Int J Hydrog Energy 35:13399–13406. https://doi.org/10.1016/j.ijhydene.2009.11.119
Chen CC, Lin CY, Lin MC (2002) Acid-base enrichment enhances anaerobic hydrogen production process. Appl Microbiol Biotechnol 58:224–228. https://doi.org/10.1007/s002530100814
Chen W-H, Chen S-Y, Khanal SK, Sung S (2006) Kinetic study of biological hydrogen production by anaerobic fermentation. Int J Hydrog Energy 31:2170–2178. https://doi.org/10.1016/j.ijhydene.2006.02.020
Chidthaisong A, Conrad R (2000) Specificity of chloroform, 2-bromoethanosulfonate and fluoroacetate to inhibit methanogenesis and other anaerobic processes in anoxic rice field soil. Soil Biol Biochem 32(7):977–988. https://doi.org/10.1016/S0038-0717(00)00006-7
De Gioannis G, Muntoni A, Polettini A, Pomi R (2013) A review of dark fermentative hydrogen production from biodegradable municipal waste fractions. Waste Manag 33:1345–1361. https://doi.org/10.1016/j.wasman.2013.02.019
Dubois SM, Gilles KA, Hamilton JL, Rebers PA, Smith F (1956) Colorimetric methods for determination of sugar and related substance. Anal Chem 28(3):350–356. https://doi.org/10.1021/ac60111a017
Eker S, Sarp M (2016) Hydrogen gas production from waste paper by dark fermentation: effects of initial substrate and biomass concentrations. Int J Hydrog Energy 42(4):2562–2568. https://doi.org/10.1016/j.ijhydene.2016.04.020
Fernandes BS, Peixoto G, Albrecht FR, Saavedra del Aguila NK, Zaiat M (2010) Potential to produce biohydrogen from various wastewaters. Energy Sustain Dev 14(2):143–148. https://doi.org/10.1016/j.esd.2010.03.004
Gokfiliz P, Karapinar I (2016) The effect of support particle type on thermophilic hydrogen production by immobilized batch dark fermentation. Int J Hydrog Energy 42(4):2553–2561. https://doi.org/10.1016/j.ijhydene.2016.03.041
Grootscholten TIM, Kinsky dal Borgo F, Hamelers HVM, Buisman CJN (2013) Promoting chain elongation in mixed culture acidification reactors by addition of ethanol. Biomas Bioenerg 48:10–16. https://doi.org/10.1016/j.biombioe.2012.11.019
Grootscholten TIM, Strik DPBTB, Steinbusch KJJ, Buisman CJN, Hamelers HVM (2014) Two-stage medium chain fatty acid (MCFA) production from municipal solid waste and ethanol. Appl Energ 116:223–229. https://doi.org/10.1016/j.apenergy.2013.11.061
Harper SR, Pohland FG (1986) Recent development in hydrogen management during wastewater treatment. Biotechnol Bioeng 28:585–602. https://doi.org/10.1002/bit.260280416
Ho C-Y, Chang J-J, Lin J-J, Chin T-Y, Mathew GM, Huang C-C (2011) Establishment of functional rumen bacterial consortia (FRBC) for simultaneous biohydrogen and bioethanol production from lignocellulose. Int J Hydrog Energy 36:12168–12176. https://doi.org/10.1016/j.ijhydene.2011.06.125
Kenealy WR, Cao Y, Weimer PJ (1995) Production of caproic acid by cocultures of ruminal cellulolytic bacteria and Clostridium kluyveri grown on cellulose and ethanol. Appl Microbiol Biotechnol 44:507–513. https://doi.org/10.1007/BF00169952
Kim SH, Han SK, Shin HS (2004) Feasibility of biohydrogen production by anaerobic co-digestion of food waste and sewage sludge. Int J Hydrog Energy 29(15):1607–1616. https://doi.org/10.1016/j.ijhydene.2004.02.018
Köchling T, Ferraz AD Jr, Florencio L, Kato MT, Gavazza S (2017) Pyrosequencing analysis of highly adapted azo dye-degrading microbial communities in a two-stage anaerobic-aerobic bioreactor treating textile effluent. Environ Technol 38(6):687–693. https://doi.org/10.1080/09593330.2016.1208681
Lee K-S, Hsu Y-F, Lo Y-C, Lin P-J, Lin C-Y, Chang J-S (2008) Exploring optimal environmental factors for fermentative hydrogen production from starch using mixed anaerobic microflora. Int J Hydrog Energy 33:1565–1572. https://doi.org/10.1016/j.ijhydene.2007.10.019
Li CL, Fang HHP (2007) Fermentative hydrogen production from wastewater and solid wastes by mixed cultures. CRC Rev Environ Sci Technol 37(1):1–39. https://doi.org/10.1080/10643380600729071
Liu H, Wang J, Wang A, Chen J (2011) Chemical inhibitors of methanogenesis and putative applications. Appl Microbiol Biot 89(5):1333–1340. https://doi.org/10.1007/s00253-010-3066-5
Logan BE, Call D, Cheng S, Hamelers HVM, Sleutels T, Jeremiasse AW, Rozendal RA (2008) Microbial electrolysis cells for high yield hydrogen gas production from organic matter. Environ Sci Technol 42:8630–8640. https://doi.org/10.1021/es801553z
Lucena RM, Gavazza S, Florencio L, Kato MT, Morais MA Jr (2011) Study of the microbial diversity in a full-scale UASB reactor treating domestic wastewater. World J Microb Biotechnol 27(12):2893–2902. https://doi.org/10.1007/s11274-011-0771-x
Luo G, Xie L, Zou Z, Wang W, Zhou Q (2010) Exploring optimal conditions for thermophilic fermentative hydrogen production from cassava stillage. Int J Hydrog Energy 35:6161–6169. https://doi.org/10.1016/j.ijhydene.2010.03.126
Maintinguer SI, Fernandes BS, Duarte ICS, Saavedra NC, Adorno MAT, Varesche MB (2008) Fermentative hydrogen production by microbial consortium. Int J Hydrog Energy 33(16):4309–4317. https://doi.org/10.1016/j.ijhydene.2008.06.053
Marshall CW, LaBelle EV, May HD (2013) Production of fuels and chemicals from waste by microbiomes. Curr Opin Biotechnol 24:391–397. https://doi.org/10.1016/j.copbio.2013.03.016
Moraes EM, Adorno MAT, Zaiat M, Foresti E (2000) Determinação de ácidos voláteis por cromatografia gasosa em efluentes de reatores anaeróbios tratando resíduos líquidos e sólidos. In: VI Oficina e Seminário Latino-Amerocano de Digestão Anaeróbia. Anais da VI Oficina e Seminário Latino-Amerocano de Digestão Anaeróbia, v 2. Recife—PE: Universitária—UFPE, pp. 235–238 (in Portuguese)
Nandi R, Sengupta S (1998) Microbial production of hydrogen: an overview. Crit Rev Microbiol 24(1):61–84. https://doi.org/10.1080/10408419891294181
Nissila ME, Tahti HP, Rintala JA, Puhakka JA (2011) Thermophilic hydrogen production from cellulose with rumen fluid enrichment cultures: effects of different heat treatments. Int J Hydrog Energy 36:1482–1490. https://doi.org/10.1016/j.ijhydene.2010.11.010
Nocek JE, Russell JB (1988) Protein and energy as an integrated system. Relationship of ruminal protein and carbohydrate availability to microbial synthesis and milk production. J Dairy Sci 71:2070–2107. https://doi.org/10.3168/jds.S0022-0302(88)79782-9
O-Thong S, Prasertsan P, Karakashev D, Angelidaki I (2008) Thermophilic fermentative hydrogen production by the newly isolated Thermoanaerobacterium thermosaccharolyticum PSU-2. Int J Hydrog Energ 33(4):1204–1214. https://doi.org/10.1016/j.ijhydene.2007.12.015
Ottaviano LM, Ramos LR, Botta LS, Varesche MBA, Silva EL (2016) Continuous thermophilic hydrogen production from cheese whey powder solution in an anaerobic fluidized bed reactor: effect of hydraulic retention time and initial substrate concentration. Int J Hydrog Energy. https://doi.org/doi.org/10.1016/j.ijhydene.2016.11.168
Pakarinen O, Lehtoma A, Rintala J (2008) Batch dark fermentative hydrogen production from grass silage: the effect of inoculum, pH, temperature and VS ratio. Int J Hydrog Energy 33:594–601. https://doi.org/10.1016/j.ijhydene.2007.10.008
Phowan P, Danvirutai P (2014) Hydrogen production from cassava pulp hydrolysate by mixed seed cultures: effects of initial pH, substrate and biomass concentrations. Biomass Bioenergy 64:1–10. https://doi.org/10.1016/j.biombioe.2014.03.057
Ren N-Q, Guo W-Q, Wang X-J, Xiang W-S, Liu B-F, Wang X-Z, Ding J, Chen Z-B (2008) Effects of different pretreatment methods on fermentation types and dominant bacteria for hydrogen production. Int J Hydrog Energy 33:4318–4324. https://doi.org/10.1016/j.ijhydene.2008.06.003
Sparling R, Risbey D, Poggi-Varaldo HM (1997) Hydrogen production from inhibited anaerobic composters. Int J Hydrog Energy 22(6):563–566. https://doi.org/10.1016/S0360-3199(96)00137-1
Sprott GD, Jarrell KF, Shaw KM, Knowles R (1982) Acetylene as an inhibitor of methanogenic bacteria. J Gen Microbiol 128:2453–2462. https://doi.org/10.1099/00221287-128-10-2453
Steinbusch KJJ, Hamelers HVM, Buisman CJN (2008) Alcohol production through volatile fatty acids reduction with hydrogen as electron donor by mixed cultures. Water Res 42:4059–4066. https://doi.org/10.1016/j.watres.2008.05.032
Steinbusch KJJ, Hamelers HVM, Plugge CM, Buisman CJN (2011) Biological formation of caproate and caprylate from acetate: fuel and chemical production from low grade biomass. Energy Environ Sci 4:216–224. https://doi.org/10.1039/C0EE00282H
Valdez-Vazquez I, Poggi-Varaldo HM (2009) Hydrogen production by fermentative consortia. Renew Sust Energy Rev 13:1000–1013. https://doi.org/10.1016/j.rser.2008.03.003
Valdez-Vazquez I, Sparling R, Risbey D, Rinderknecht-Seijas N, Poggi-Varaldo HM (2005) Hydrogen generation via anaerobic fermentation of paper mill wastes. Bioresour Technol 96:1907–1913. https://doi.org/10.1016/j.biortech.2005.01.036
Van Ginkel SW, Logan B (2005) Increased biological hydrogen production with reduced organic loading. Water Res 39(16):3819–3826. https://doi.org/10.1016/j.watres.2005.07.021
Van Ginkel S, Sung S, Lay JJ (2001) Biohydrogen production as a function of pH and substrate concentration. Environ Sci Technol 35:4726–4730. https://doi.org/10.1021/es001979r
Wang J, Wan W (2009a) Factors influencing hydrogen production: a review. Int J Hydrog Energy 34:799–811. https://doi.org/10.1016/j.ijhydene.2008.11.015
Wang J, Wan W (2009b) Experimental design methods for fermentative hydrogen production: a review. Int J Hydrog Energy 34:235–244. https://doi.org/10.1016/j.ijhydene.2008.10.008
Wang A, Gao L, Ren N, Xu J, Liu C, Lee D-J (2010) Enrichment strategy to select functional consortium from mixed cultures: consortium from rumen liquor for simultaneous cellulose degradation and hydrogen production. Int J Hydrog Energy 35:13413–13418. https://doi.org/10.1016/j.ijhydene.2009.11.117
Wang K, Yin J, Shen D, Li N (2014) Anaerobic digestion of food waste for volatile fatty acids (VFAs) production with different types of inoculum: effect of pH. Bioresour Technol 161:395–401. https://doi.org/10.1016/j.biortech.2014.03.088
Wang S, Zhang T, Su H (2016) Enhanced hydrogen production from corn starch wastewater as nitrogen source by mixed cultures. Renew Energy 96:1135–1141. https://doi.org/10.1016/j.renene.2015.11.072
Wu KJ, Chang CF, Chang JS (2007) Simultaneous production of biohydrogen and bioethanol with fluidized-bed and packed bed bioreactors containing immobilized anaerobic sludge. Process Biochem 42:1165–1171. https://doi.org/10.1016/j.procbio.2007.05.012
Zheng H, Zeng RJ, Angelidaki I (2008) Biohydrogen production from glucose in upflow biofilm reactors with plastic carriers under extreme thermophilic conditions (70 °C). Biotechnol Bioeng 100(5):1034–1038. https://doi.org/10.1002/bit.21826
Acknowledgements
The authors gratefully acknowledge the financial support of the Brazilian agencies, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPQ, Project 441495/2014-1, granted to the second author) and Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE, Project Pronex APQ 0603-3.07/14, granted to the third author).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amorim, N.C.S., Amorim, E.L.C., Kato, M.T. et al. The effect of methanogenesis inhibition, inoculum and substrate concentration on hydrogen and carboxylic acids production from cassava wastewater. Biodegradation 29, 41–58 (2018). https://doi.org/10.1007/s10532-017-9812-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-017-9812-y