Abstract
The area of semi-natural grasslands has decreased dramatically causing many grassland specialist species to persist in small habitat fragments. Furthermore, ecological communities once shaped by disturbances related to traditional agriculture now face the impacts of modern agriculture and urbanization. Many of the species have become endangered due to the combined effects of habitat destruction and degradation. We studied the responses of vascular plants and bryophytes to present and past connectivity in semi-natural grasslands, while accounting for the effects of local environmental conditions. We conducted vegetation surveys in 33 fragmented, unmanaged dry grasslands in Southwest Finland. A total of 191 vascular plant species and 60 bryophyte species were recorded. Higher vascular plant diversity was associated with higher historical connectivity, whereas bryophyte diversity declined with increasing current connectivity. Edaphic conditions, microclimate and disturbances were found to structure both vascular plant and bryophyte communities, but the responses to individual environmental variables were very different. The contrasting responses could be explained by the differences between the typical life-history strategies in vascular plants and bryophytes as well as resource competition between the two groups. These results highlight the need to consider different species groups when planning conservation measures. Vascular plant richness may best be conserved in connected grasslands whereas bryophyte richness in isolated ones. The results also indicated that trampling by humans could result in positive disturbance and to some extent help maintain richness of grassland specialist species.
Similar content being viewed by others
Introduction
Habitat destruction is the primary driver of global biodiversity loss (Fahrig 2003; Hanski 2011). It is especially detrimental to habitats and species that are already rare or threatened. In Europe, one of the most threatened habitat groups are dry semi-natural grasslands (European commission 2017; Kontula and Raunio 2018). The area of dry semi-natural grasslands in Finland has reduced from an estimated 150 000 hectares in the 1860s to 13,000 hectares in the 1960s and still to only 700 hectares in 2018, meaning 95% decrease in 60 years. (Lehtomaa et al. 2018b). This area provides primary habitat for almost 300 nationally endangered species in 17 taxonomic groups (such as plants, fungi, Arachnida, Coleoptera, Lepidoptera). In terms of plants, semi-natural grasslands provide habitat for 30 endangered bryophyte and 62 endangered vascular species in Finland. (Hyvärinen et al. 2019).
The semi-natural grasslands have not been under traditional management practices other than grazing or haymaking with scythe. However, grasslands of high conservation value may also develop from abandoned agricultural fields given time under some conditions (Ejrnæs et al. 2008). One of the primary reasons for habitat loss in dry semi-natural grasslands is the lack of suitable management such as grazing and the subsequent progress of secondary succession (Lehtomaa et al. 2018a). In semi-natural grasslands, the onset of secondary succession often leads to an increased cover of bushes and trees and at the same time to a decrease in species richness of vascular plants in the field layer (Lazzaro et al. 2020). A considerable threat are also invasive alien species such as Amelanchier spicata and Lupinus polyphyllus (Hyvärinen et al. 2019). As those examples present a major threat for especially mesic meadows and freshwater meadows (Lehtomaa et al. 2018b), dry semi-natural grasslands, particularly near city center (Štajerová et al. 2017) provide them a habitat for seed dispersal to more vulnerable environments.
Based on island biogeography and metapopulation theory, conservation planning has traditionally aimed to protect and restore networks of large, well-connected habitats (Wintle et al. 2019). Such an approach is seldom applicable in dry grassland conservation, because in most landscapes these habitats exist only as small, fragmented remnants of their previous extent. Despite their small size and isolation, such remnant grasslands may contribute to landscape-scale biodiversity by supporting communities and endangered species not found in other habitats (Albrecht and Haider 2013; Janišová et al. 2014; Lampinen 2019). This highlights the importance of acknowledging them in conservation planning (Wintle et al. 2019). It also calls for increased understanding of how different species inhabiting dry grasslands respond to the abandonment, fragmentation and isolation of their habitats, and to the following changes in environmental conditions.
Species communities assemble based on both local and landscape-scale conditions that filter species according to variation in the environment (Löbel et al. 2006; Raatikainen et al. 2009; Frey et al. 2011; Reitalu et al. 2012). In dry grasslands, local conditions are central aspects of habitat quality that define whether grassland species persist in a grassland (Pykälä et al. 2005; Lampinen 2019). From the perspective of a given plant species, the most important local conditions comprise those related to prevailing microclimate and soil properties. For example, exposure to heat and sunlight (Tinya et al. 2009), soil pH (Hydbom et al. 2012; Ewald 2003) and consequent nutrient availability (Bedford et al. 1999), the amount of living and dead biomass (Carson and Peterson 1990), and the amount of exposed bare rock or trampled bare ground (Jägerbrand and Alatalo 2015) in the habitat all covary with species occurrence. Landscape-scale conditions such as structural connectivity between suitable grasslands are, in turn, central to species dispersal (Crooks and Sanjayan 2006; Auffret et al. 2015). Species occurrence may also depend on the interaction between local and landscape-scale conditions (Matter et al. 2009): high structural connectivity is unlikely to increase the occurrence of a grassland species if the remaining grassland patches are of poor quality. Furthermore, responses to local or landscape-scale conditions differ between taxonomic groups. Depending on the mobility of a given species and dispersal distance, a grassland fragment can be perceived as highly connected or isolated. Thus, planning grassland conservation based on data collected only at a single scale (Reitalu et al. 2012) or from a single taxonomic group may lead to unintended outcomes in protecting different groups of species adapted to grasslands (Takala et al. 2012, 2014; Zulka et al. 2014).
Despite the need to consider more than one species group, the majority of research underlying grassland conservation concerns vascular plants and their responses to varying local and landscape-scale conditions (e.g. Pykälä et al. 2005; Öster et al. 2007; Reitalu et al. 2012). Bryophytes form an important part of the vegetation, but the patterns and underlying drivers of bryophyte diversity in semi-natural grasslands are insufficiently understood (Löbel et al. 2006; Müller et al. 2012; Takala et al. 2012; García de León et al. 2016; Simmel et al. 2021). Yet, certain conditions important to both bryophyte and vascular plant diversity patterns have been identified. On a local scale, especially soil pH (Virtanen et al. 2000; Löbel et al. 2006; Hydbom et al. 2012), productivity (Haworth et al. 2007; Müller et al. 2012), and disturbance (Takala et al. 2012; Tyler et al. 2018) are all associated with changes in the occurrence and species richness of bryophytes and vascular plants in semi-natural grasslands. Unfortunately, little is still known about the interactions between the two species groups in grasslands. Arguments for the existence of both facilitative (e.g. Ingerpuu et al. 2005) and competitive (e.g. Virtanen et al. 2000; Peintinger and Bergamini 2006) interactions between bryophytes and vascular plants exist, but a consensus is lacking in terms of the dominant interaction in different conditions.
At a landscape-scale, several studies have related the diversity patterns and community composition of vascular plants to present and past connectivity between grasslands in the surrounding landscape (Uroy et al. 2019) and the age of the grassland (Redhead et al 2014; Schmid et al. 2017; Karlík and Poschlod 2019). The richness of grassland plants in a given grassland often reflects the past rather than the present structural connectivity of that grassland to those in the surrounding landscape (e.g. Lindborg and Eriksson 2004; Helm et al. 2006; Husáková and Münzbergová 2014; Lampinen et al. 2018). This slow response of plant communities to changes in the surrounding landscape indicates that a further decline of vascular plant species richness may be expected in grasslands with reduced connectivity or habitat area. The responses of bryophyte species richness to connectivity have seldom been studied in semi-natural grasslands and the results are contradictory. For example, Zulka et al. (2014) suggest that the species richness of bryophytes in dry grasslands is positively associated with the present area of similar types of habitats in the surrounding landscape, while Löbel et al. (2006) report no statistically significant relationship between these two factors. Evidence from single-taxon studies suggest that bryophytes may respond to a loss of connectivity with a similar time lag as vascular plants (Snäll et al. 2004). Nonetheless, we do not currently fully understand how bryophytes respond to changes in present or past connectivity in semi-natural grasslands, or how these landscape-scale conditions interact with local conditions to shape bryophyte communities in them.
Without a deeper understanding of how different groups of species interact in or respond to varying environmental conditions in fragmented dry grasslands, it is difficult to plan the restoration of the remaining grasslands in an ecologically informed manner. It is equally difficult to manage the grasslands in such ways that species or groups of species prioritized for protection benefit from management. To fill this gap in our understanding, we explore the drivers behind the species richness of vascular plants and bryophytes in a network of fragmented dry grasslands varying in their degree of structural connectivity. We address the following research questions:
-
(1)
What are the primary local environmental drivers of vascular plant and bryophyte species richness in fragmented, unmanaged dry grasslands?
-
(2)
Is the current species richness of bryophytes and vascular species correlated similarly with present or historical structural connectivity between dry grasslands?
Materials and methods
Study area and sampling
The study was conducted in the city of Turku, South-Western Finland (city center coordinates in WGS 84 coordinate system: 60° 27′ 6.08″ N, 22° 16′ 0.72″ E). The city is situated on the hemiboreal vegetation zone (Kalliola 1973), and has a continental, humid climate of warm summers (the Dfb-type of the Köppen climate classification) (Peel et al. 2007). The geological rock classes consist mainly of metamorphic rock (sedimentary protolith-composition-texture) and microcline granite (Geological Survey of Finland (2017)[please, update the link between list and text]). During the 30-year period 1991–2020 in Turku, the monthly temperature average varied between − 4.5 °C (February) and + 17.5 °C (July), and the annual average was + 5.8 °C. Monthly precipitation average varied between 32 mm (April) and 74 mm (July), and the average annual precipitation was 684 mm (Jokinen et al. 2021).
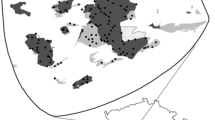
Due to the 4000 years of agriculture in the surrounding region (Cramp et al. 2014), there are several hundred fragments of ancient dry grassland in the city as remnants of historical, open agricultural landscapes preceding urban development. The majority of the fragments are rocky with shallow soil indicating grazing rather than haymaking as the historical mode of management. Indeed, the most recent records of agricultural management in the studied grasslands, grazing by cattle, date to the 1950’s (Lampinen and Koskela 2016). The grasslands used in this study were sampled from a GIS-dataset depicting the distribution of these remnant fragments in an area of 8 × 9 km surrounding the center of the city (Fig. 1). All sampled grasslands were interpreted as ancient, i.e. sharing a history related to early onset of habitation and agriculture in the surrounding region, and contemporary grasslands such as those developed in road verges or abandoned cultivated fields were omitted from the sampling. The habitat types of all mapped fragments vary from rocky meadows to low herb-rich dry meadows and Avenula pubescens dry meadows (as described by Lehtomaa et al. 2018b). According to a vegetation survey conducted in similar grassland fragments in the surrounding countryside, common dry grassland specialist species in similar fragments include Galium verum, Filipendula vulgaris, Helictochloa pratensis, Helictotrichon pubescens, Silene nutans and Luzula campestris (Lampinen 2017). On average, the mapped grassland fragments are 0.07 ha in size and the sampled grassland fragments 0.05 ha in size.
Historical (grey) and current (black) dry grasslands in the city of Turku, south-western Finland. Encircled polygons depict grassland fragments included in this study (n = 33), exemplified by photographs of three grassland fragments with corresponding numbers in the map. Background map
To decide which grassland fragment to sample, we first created a preliminary classification of structural connectivity of each fragment. First, we calculated the average geographical distance of each grassland to ten nearest grasslands. We then divided the grasslands into three categories based on this distance: grasslands with the lowest (< 695 m), intermediate (700–935 m) and highest average distances (> 940 m) to other grasslands, respectively. We randomly sampled 11 dry grasslands from each category resulting in a total of 33 study sites varying in their geographical distance (i.e. current structural connectivity) to other dry grasslands.
Data collection and types of data
The data consist of counts of vascular plant and bryophyte species richness and of environmental variables describing the local conditions and structural connectivity of each grassland (Table 1). The data collection began by preliminary field visits to each sampled grassland in early summer 2018. During these visits each grassland was delineated on aerial images (Map data © 2015 Google) based on the presence of vegetation and structural features typical to dry grasslands in the study region (Lehtomaa et al. 2018b). The grassland boundaries were digitized into GIS-format for further data collection (QGIS version 2.18.26).
After the preliminary field visits to the sampled grasslands, both the species richness and environmental data were collected between 4.6. and 17.8.2018 at three different scales: some variables were measured at the scale of 1 m2 study plots established in each grassland, some at the scale of individual grasslands, and some at the scale of the surrounding landscape (Table 1). The 1 m2 study plots (five in each grassland) were laid evenly with one-meter intervals along a 10-m-long transect established in each grassland. The transect crossed the grassland in the middle and was oriented parallel to the slope of the grassland.
Species richness data
The species richness data were collected both at the scale of plots and individual grasslands. At the scale of plots, the data described the richness of all vascular plant and bryophyte species observed in the study plots. At the scale of individual grasslands, the data described the richness of vascular plant species observed during a standardized 10-min survey of each grassland. The survey enabled gathering data of the most common species as well as grassland specialist species at the site. At both plot and grassland scales, the species were identified on site or, especially in case of bryophytes, later in the laboratory under a microscope.
In addition to the species richness of all observed vascular plant species, the richness of grassland specialist vascular plant species was recorded at the scale of plots and individual grasslands. We defined grassland specialist species as those shown to occur both historically and currently primarily in semi-natural grasslands following the classification of Pykälä (2001), Pykälä et al. (2005). The classification of specialist species includes species that are shown to benefit from traditional agriculture such as grazing and excludes those that may suffer from it to some extent even when they may be commonly met at meadows (Pykälä 2001).
Environmental data
The local environmental data collected in the study plots consisted of visual estimates of the percentage cover of trampled or bare ground, of bare rock, and of plant litter (dead and decomposing plant material) in each plot. The ground was interpreted as trampled when it was located on a path and or was bare and when the vegetation in the field layer was clearly stomped. The rock or ground was defined as bare when it had no living or decaying vegetation. The local environmental data collected from the surrounding grassland described the area, slope, aspect, soil acidity and successional stage of each grassland. The area of each grassland, delineated during the preliminary surveys, was calculated with QGIS (QGIS Development Team 2018). The slope was visually estimated in degrees of declination. The aspect was counted as a deviation from exact South by subtracting the cardinal direction of the slope in compass degrees from 180. Soil pH was measured in five 50–100 ml soil samples collected from the center of each study plot a few centimeters below the soil surface or less when the soil surface was thin. The soil samples were stored in individual plastic bags and analyzed in December 2018 and January 2019, when they were filtered through a 5 mm sieve and mixed in 90 ml of tap water with a soil:water ratio of 1:5. The acidity of the suspension was measured after 11 h in the shaker (Heidolph Unimax 1010) with a calibrated pH meter (VWF pH1000H). As semi-natural grasslands undergo a successional shift towards woody vegetation types in the absence of disturbance, we also estimated the extent to which such a shift had progressed in each grassland. To achieve this, we used open-access, LIDAR-derived vegetation canopy height model raster with 1 m2 resolution from the year 2017 (Finnish Forest Center 2021), overlaid with the GIS lineations of the sampled grassland. We considered areas in each grassland covered by vegetation lower than 1 m not to have undergone a successional shift and areas with vegetation higher than 1 m as in active state of successional shift. The extent of successional shift in each grassland was estimated as the proportional cover of trees and bushes higher than 1 m. The larger the proportion of each fragment covered by trees and bushes higher than 1 m, and the higher those trees and bushes across the grassland, the higher the estimated extent of successional shift.
The structural connectivity of each grassland was measured at the scale of the surrounding landscape and described the present and historical cover (in ha) of each studied grassland to others within a 250-m-wide buffer. While the present connectivity was calculated with the same dataset from which the studied grasslands were sampled, the historical connectivity was calculated based on digitized Senate maps, a nineteenth century series of 1:21 000 maps depicting the land use and land cover in southern Finland (National Archives Service of Finland 2022; Lampinen et al. 2015, 2018). In these maps, grasslands are depicted as three distinct types: mesic grasslands, wet grasslands and non-forested dry grasslands and pastures (Lampinen et al. 2015). The last of these has been identified as linking to current species richness patterns of species adapted to semi-natural grasslands (Lampinen et al. 2018). Because of this, we calculated the historical connectivity data based on the cover of dry grasslands and pastures, but also included the total cover of all aforementioned historical grassland types as an additional measure of the historical landscape configuration surrounding each grassland. The buffer zone was set to 250 m to avoid redundancy in the structural connectivity data: the grasslands available for the study are located in an area less than eight kilometers in diameter, and wider buffers would have overlapped with those surrounding other sampled grasslands. For further description of the data used in the study, see Table 1.
Statistical analyses
We explored the responses of vascular plant and bryophyte species richness to the environmental variables with exploratory hierarchical partitioning (HP), generalized linear models (GLM), and generalized linear mixed models (GLMM) utilizing Poisson distribution and a log-linear link function. We used HP models to explore how much variation each of the environmental variables explains independently of others in the species richness data of the studied species groups. We then used univariate GLMs to explain the species-richness of the studied species groups at the scale of grasslands (n = 33), and univariate GLMMs to explain it at the scale of individual vegetation plots (n = 165) to develop a more precise understanding of the strength and direction of each relationship between the environmental and species richness variables.
As the HP algorithm can process only nine variables (Nally and Walsh 2004), certain variables presumed to be correlated with others were omitted from the exploratory models. For example, only the historical connectivity to all grasslands was included in the analysis due to its correlation with the historical cover of dry grasslands, the latter being a subset of the former. In each HP model, we tested the statistical significance of the proportion of explained variance with 1000 permutations. On the contrary, the explanatory variables in the univariate grassland-scale GLMs included all environmental variables measured at the scale of grasslands and those measured on the plot-scale, with the latter averaged over all five plots placed in each grassland. The explanatory variables in the univariate plot-scale GLMMs included all environmental variables measured at the scale of individual study plots and those measured at the scale of each grassland. The GLMM model for bryophyte species richness also included the total cover of all vascular plants in the plot. As each grassland included a total of five vegetation plots, we included grassland identity as a random variable in each GLMM.
We checked the residuals of the explanatory variables for normality with the Shapiro–Wilk -test in GLMs and GLMMs. This resulted in the exclusion of the plant litter variable from the models due to a non-normal distribution and lack of variation between the grasslands. To test spatial autocorrelation in the data, we calculated and compared the expected and observed Moran’s I of the species richness of each studied species group across the sampled grasslands. This revealed no evidence of spatial autocorrelation in the species richness data (Online Appendix 1). Finally, the p-values in the models were corrected against false discovery rate (Verhoeven et al. 2005). All analyses were conducted with R software (Version 1.1.463) and the associated packages stats (R Core Team 2020), hier.part (Nally and Walsh 2004), Hmisc (Harrell and Dupont 2019), ape (Paradis and Schliep 2018) and lme4 (Bates et al. 2015).
Results
We observed in total 191 vascular plant species and 60 bryophyte species in the studied grasslands. Of the former, 25 species are classified as grassland specialists in the study region (Pykälä 2001, 2005) (Table 2). On average, one grassland/plot supported 38/8 vascular plant species, 6/1.5 vascular grassland specialist plant species and 9–10/3.3 bryophyte species (Table 1). Six of the observed vascular plant species, all of them grassland specialists, are currently considered either near threatened (NT) or nationally threatened in Finland (VU, CR) (Ryttäri et al. 2012; Hyvärinen et al. 2019) (Table 2). The woody species Crataegus monogyna and Fraxinus excelsior are both in their very northern limits of distribution in the present study area and they are rare and not competitively strong. In the study area, they indicate traditional land use (moderate grazing) as without that they would not be competitive. Of the 191 vascular plants identified, 23 belonged to alien taxa in Finland (Table 2). All the observed bryophyte species have current conservation status of least concern (LC).
The average size of the grasslands sampled for this study is 0.05 ha, minimum size was 16 m2 and maximum size 0.23 ha. The range in each measured environmental variable among the grassland fragments sampled in this study are shown in Table 1. Initial inspections of the correlations between grassland area and species richness patterns revealed only moderate positive or negative correlations, for example between area and number of grassland specialist species the correlation coefficient was 0.25 and as for number of bryophyte species it was -0.23.
The grasslands were situated on moderate slopes of 17° and at an aspect of 167°. The soil in the grasslands was moderately acidic with an average pH 5.6. Bare rock covered on average 8.5%, plant litter 88.7% and trampled ground 39.5% of each grassland. The present structural connectivity of each studied grassland (i.e. the cover in hectares of present dry grasslands within a buffer 250 m in diameter) was low, on average below 0.01 ha. Historical structural connectivity was higher, totaling on average 3.7 ha (Fig. 1).
Vascular plant species richness
According to the HP model, only historical connectivity to other grasslands contributed significantly and independently to the species richness of all vascular plants at the scale of grasslands (Fig. 2a), and the same was evident in GLMs (Tables 3, 5). At the scale of individual plots, HP model indicated again that the historical connectivity to other grasslands, but also the current connectivity, grassland area, cover of bare rock and cover of trampled ground contributed significantly and independently to the species richness of all vascular plants (Fig. 2b). In the GLMMs, historical connectivity had a significant positive relationship with the species richness of all vascular plants, while the cover of bare rock had a negative relationship with it (Tables 4, 5). The results were somewhat similar regarding grassland specialist plant species richness: at the grassland scale, only the cover of trampled ground had a significant independent contribution to the species richness of grassland specialists according to both HP models and GLMs (Fig. 2a, Tables 3, 5). At the plot scale, also current connectivity, grassland area, cover of bare rock and slope steepness emerged in HP model as significant independent contributors to the species richness of grassland specialist vascular plants (Fig. 2b), while in the GLMMs, only the cover of bare rock and the degree of successional shift had significant and negative relationships with it (Tables 4, 5).
Variance in the species richness of the studied species groups partitioned into proportions explained independently by each of the listed environmental variables at the scale of a grasslands (n = 33) and b vegetation plots (n = 165). Asterisks denote statistically significant effects after 1000 permutations. Historical connectivity here refers to all grassland types ranging from dry to mesic
Bryophyte species richness
According to the HP model, the cover of litter, the cover of bare rock and the steepness of slope contributed significantly and independently to the variation in the richness of bryophyte species at the scale of grasslands (Fig. 2a). As for univariate GLMs, the species richness of bryophytes related positively with the cover of bare rock, and negatively with the grassland area, slope steepness, cover of vascular plants and current connectivity to other dry grasslands (Tables 3, 5).
At the plot scale, current connectivity to other grasslands, and again the cover of litter, the cover of bare rock and the steepness of slope contributed significantly and independently to the variation in the richness of bryophyte species according to the HP models (Fig. 2b). In the univariate GLMMs, the species richness of bryophytes had a significant positive relationship with the cover of bare rock, and a negative relationship with slope steepness and cover of vascular plants (Tables 4, 5).
Discussion
Conservation measures intended to maintain or increase species richness of indigenous species and protect endangered species usually aim at creating and sustaining a network of conservation areas with habitat conditions suitable for the species in question (e.g. Wintle et al. 2019; Correa Ayram et al. 2016). The present study shows that such attempts, when based on data from a single species group, may result in different outcomes regarding the species richness in other species groups. We observed that the most important determinants of species richness of vascular plants and bryophytes in dry grasslands are primarily different and when these drivers overlap their effect on the studied groups may be opposite. For example, the species richness of vascular plants in a given grassland fragment depended strongly on historical connectivity but had no significant relationship with current connectivity. In contrast, there was no strong evidence that the species richness of bryophytes responded to historical context, but it did respond negatively to current habitat connectivity. These results show how the requirements in terms of habitat quality of two distinct lineages of plants, both common in semi-natural grasslands, do not always align. It has been shown previously that alien species inhabit dry grasslands in urban environments (Boscutti et al. 2022; Štajerová et al. 2017). In the present case, 12% of the vascular species richness was composed of alien species, which is higher than the global average of 3.9% (Kleunen et al. 2015), but lower than in e.g. the 20% in NW Poland (Popiela et al 2015). To prohibit the distribution to even more vulnerable environments, conservation measures in grasslands should always include the removal of alien and invasive alien species.
Contrasting responses of vascular plants and bryophytes to structural connectivity in dry grasslands
While we observed current structural connectivity to explain a small, but significant fraction of the variation in the species richness in all studied species groups, the effect of historical connectivity was significant only in vascular plants. Further, the relationship between current connectivity and bryophyte species richness was negative, while that between historical connectivity and vascular plant species richness was positive. We attribute these results to differences in life-history traits between the studied species groups, as dispersal, propagule production rate, and seed and spore bank persistence all influence the responses of species to habitat fragmentation (Kolb and Diekmann 2005) and include important differences between the studied species groups.
Many studies report a positive relationship between the species richness patterns of vascular plants in grasslands and past structural habitat connectivity (e.g. Lindborg and Eriksson 2004; Lampinen et al. 2018). The strength of these relationships has been attributed to life history traits (Lindborg 2007). Well-connected habitats may be expected to accumulate more species with time than isolated ones, and local extinctions in isolated habitats may lead to more long-lasting or even permanent declines in species richness as dispersal from nearby communities is not possible (Helm et al. 2006). Due to time lags at which the species richness of plant communities responds to habitat loss or connectivity, current communities often reflect the past rather than the present habitat configuration in the system in question (Helm et al. 2006). We interpret the observed vascular plant communities to be currently in the process of reaching an equilibrium with the habitat configuration in the network of grasslands in question, as both historical and current connectivity were identified as significant independent contributors to their species richness. This may translate to future declines in species richness, because the proportion of variance explained by historical connectivity (50%) was by far larger than that explained by current connectivity (10%).
Efforts to link bryophyte diversity patterns with measures of past landscape, whether related to functional or structural connectivity, disturbance, or management, are much rarer than those concerning vascular plants. Furthermore, the studies primarily focus on epiphyte species prior to the present work. Similarly to our study, Király et al. (2013) found no relationship between the amount of historical habitat and current species richness of epiphytic bryophytes in forests. On the other hand, McCune et al. (2021) showed that the species richness of perennial forest bryophyte communities is affected by road density several decades earlier, a proxy for habitat availability. We suggest that the observed lack of significant relationships between current bryophyte species richness and historical connectivity stems from the specific ecology and life-history traits of bryophytes as a plant group. Firstly, although there are very few studies on bryophyte spore longevity, the bryophyte spore-bank may not be as persistent as the seed bank of vascular plants, and the longevity of bryophyte spores most likely varies according to the taxa and habitat in question (Glime 2017). For example, Sphagnum spores may remain viable in peatlands for hundreds of years (Bu et al. 2017), while the spores of epiphytic and epilithic bryophytes may remain viable only a few years (Glime 2017). Another possible explanation for the lack of relationship between bryophyte species richness and historical connectivity is the readiness at which bryophytes spores disperse across long distances (Barbé et al. 2016). As a consequence, despite the declining habitat area and quality during the preceding decades in the grassland system in this study, dispersal ability may not have limited bryophytes to the same extent as vascular plants. This would have led the present species richness of bryophytes to be determined mainly by current site characteristics rather than historical connectivity.
Furthermore, in contrast to theories predicting positive relationships between plant species richness and habitat connectivity, we discovered a negative relationship between bryophyte species richness and current connectivity. A possible explanation is that the closer the remaining grassland fragments are, i.e. the more structurally connected they are, the easier it is for competitive species to colonize the fragments and outcompete the more specialist and less competitive bryophyte species. As a form of biotic homogenization (Clavel et al. 2010) this process could contribute to the decline in bryophyte species richness as a function of current structural connectivity. However, more studies are needed to quantify the importance of past and present phenomena in explaining current variation in bryophyte communities.
Edaphic conditions, microclimate and disturbance structure both vascular plant and bryophyte communities in dry grasslands
We discovered that measures related to edaphic conditions and microclimate such as the cover of bare rock or the steepness of slope translated to strong changes in the species richness of either vascular plants, bryophytes, or both. The effect of bushes, cover of bare rock and the cover of trees on vascular plant species richness were significant only at plot level. This may be partly due to the higher number of plots (165) compared to grasslands (33) but it may also demonstrate the importance of scale. Some of factors may only be noticed at an appropriate scale.
Variation in both slope steepness and the cover of bare rock indicate differences in soil moisture that species inhabiting a given grassland experience. In grasslands with steep topography and rocky and shallow soils, periods of extreme drought may contribute to some species facing periodic extinctions (Tilman and Haddi 1992), but they may also help resist successional shift by colonizing woody vegetation (Li et al. 2020). This is because grasslands on productive, moist soils undergo changes in physiognomy and community composition upon abandonment faster than those on dry, steep slopes (Bohner et al. 2012). In addition, the cover of bare rock surfaces reflects variation in habitat availability. Bare rock surfaces are a habitat suitable only for species with no need for soil and no roots, and thus increasing cover of bare rock results in decreases in the richness of vascular plant species but not necessarily in that of bryophytes. On the other hand, we discovered bryophyte species richness to decline the steeper the slope indicating possibly too extreme environmental conditions for a rich bryophyte species community. The excessive dryness on the steep slope may be the main explanatory factor (Tilman and Haddi 1992). The recreational use of the grasslands may on its part intensify the erosion of shallow soil and peel off bryophytes from rock surface (Jägerbrand and Alatalo 2015).
Soil parent material affects soil pH (Vestin et al. 2006), but bare rock may be less acidic than the adjacent soil as decomposition of organic matter may lower soil pH (Johnston et al. 1986). Soil pH is often reported as a key determinant for the diversity patterns of both vascular plants (Gheza et al. 2018; Ewald 2003) and bryophytes (Virtanen et al. 2000; Löbel et al. 2006; Oldén et al. 2016). Soil pH affects the solubility of nutrients (Palpurina et al. 2017) and thus the realized edaphic conditions that species inhabiting a given grassland experience. In this study, neither vascular plants nor bryophytes had significant relationships with soil pH, although it did explain as much as 30–40% of the variation in the species richness of the bryophytes. We attribute the apparent lack of relationship to the relatively low variation in soil pH in the studied grasslands and to the low amount of neutral bedrock in the study region, which may have resulted in few species adapted to such conditions in the regional species pool.
We observed no effects by the extent of successional shift and trampling on the species richness of bryophytes and vascular plants in general. However, the cover of trampled ground had a positive effect, and the extent of successional change had a negative effect on the richness of grassland specialist vascular plants. Grassland specialist species are defined as species that occur primarily in grasslands (Pykälä 2001; Pykälä et al. 2005). The negative relationship between grassland species richness and the extent of successional shift highlights the sensitivity of these species to specific successional stages in grasslands. These specialist species depend on disturbance to deter the progress of secondary succession, usually in the form of traditional dry grassland management such as grazing (Raatikainen et al. 2009). Together with previous studies (Burden and Randerson 1972; Lampinen 2019), our results suggest that trampling may function as a suitable disturbance and benefit grassland specialist vascular plant species in grasslands that are under no other management or disturbance regime. Although we found no evidence of trampling affecting the species richness of bryophytes, they have been shown to respond positively to moderate trampling in other studies. For example, Takala et al. (2012, 2014) observed that the species richness of bryophytes is positively linked with grazing and proposed that the effect is mediated through the decreased dominance of vascular plants in the field layer. Intense trampling, on the other hand, may be directly deleterious for some bryophyte species (Hamberg et al. 2008).
The lack of significant relationships between successional stage shift and the species richness in the other studied species groups likely relates to two alternative explanations. There may be relatively little variation in terms of successional stages between the grassland fragments included in the study. All of them have remained unmanaged for decades. Alternatively, plant species richness is not generally related to the succession stages in the mid-successional range of secondary succession (Lebrija-Trejos et al. 2010). Because the grassland specialist species responded to the succession stage shift, we find the latter explanation more likely. The species pool of the vascular plants in the landscape is likely to facilitate turnover of species in grasslands undergoing successional changes not depicted in species richness.
We found strong negative relationships between increasing cover of vascular plant species and the species richness of bryophytes both at the scale of grasslands and individual study plots. These results agree with similar findings by Virtanen et al. (2000), Peintinger and Bergamini (2006) and Takala et al. (2014) and can be attributed to asymmetrical resource competition between different species groups inhabiting grassland. In the absence of disturbance, secondary succession progresses and tall-growing vascular plants with greater biomass can outcompete the low-growing, small-statured bryophytes (Takala et al. 2014). The speed of the secondary succession and subsequent competitive exclusion depends on the environmental context and is possibly the faster the more productive the soil is (Bohner et al. 2012). In addition, the local species pool may also affect which species colonize grassland fragments and how fast the colonization takes place following abandonment (Brändle et al. 2003). This may explain why antagonistic relationships between vascular plants and bryophytes are not always found in the resource poor, dry semi-natural grasslands where succession may proceed slower than in grasslands on more productive soils (Bohner et al. 2012). For example, Gheza et al. (2018) found no evidence of antagonism between bryophytes and vascular plants in dry grasslands.
Conclusions and implications for practice
According to the present study, vascular plants and bryophytes may respond somewhat differently to the changes in environmental conditions, whether local or landscape-scale, which follow the abandonment and loss of dry grassland habitats. Despite these differences, certain implications are apparent. First, the positive relationship between the species richness of vascular plants and historical structural connectivity indicates that even if the current area of these habitats no longer deteriorates, the species richness of vascular plants may decrease in the future at the grassland scale. Second, reinstated management regimes in grasslands currently optimal for vascular plants and bryophytes and increased restoration efforts in those currently suboptimal for them could counteract decreases in species richness. Third, the restoration of currently suboptimal grasslands would favor especially bryophytes, because they decline in species richness upon increasing cover of vascular plants. Viable methods for restoring and managing dry grasslands include low intensity grazing by e.g. sheep (Dostálek and Frantík 2008; Lehmair et al. 2020). Our results suggest that in the absence of grazing or other restoration measures, moderate trampling may help deter the decline in species richness of at least grassland specialist plants.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Albrecht H, Haider S (2013) Species diversity and life history traits in calcareous grasslands vary along an urbanization gradient. Biodivers Conserv 22:2243–2267. https://doi.org/10.1007/s10531-013-0437-0
Auffret AG, Plue J, Cousins SAO (2015) The spatial and temporal components of functional connectivity in fragmented landscapes. Ambio 44:51–59. https://doi.org/10.1007/s13280-014-0588-6
Barbé M, Fenton NJ, Bergeron Y (2016) So close and yet so far away: long-distance dispersal events govern bryophyte metacommunity reassembly. J Ecol 104:1707–1719. https://doi.org/10.1111/1365-2745.12637
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bedford BL, Walbridge MR, Aldous A (1999) Patterns in nutrient availability and plant diversity of temperate North American wetlands. Ecology 80:2151–2169. https://doi.org/10.1890/0012-9658(1999)080[2151:PINAAP]2.0.CO;2
Bohner A, Starlinger F, Koutecky P (2012) Vegetation changes in an abandoned montane grassland, compared to changes in a habitat with low-intensity sheep grazing-a case study in Styria, Austria. J Protected Mt Areas Res Manage 4:5–12. https://doi.org/10.1553/eco.mont-4-2s5
Boscutti F, Lami F, Pellegrini E, Buccheri M, Busato F, Martini F, Sibella R, Sigura M, Marini L (2022) Urban sprawl facilitates invasions of exotic plants across multiple spatial scales. Biol Invasions 24:1497–1510. https://doi.org/10.1007/s10530-022-02733-6
Brändle M, Durka W, Krug H, Brandl R (2003) The assembly of local communities: plants and birds in non-reclaimed mining sites. Ecography 26:652–660. https://doi.org/10.1034/j.1600-0587.2003.03513.x
Bu Z-J, Sundberg S, Feng L, Li H-K, Zhao H-Y, Li H-Z (2017) The Methuselah of plant diaspores: Sphagnum spores can survive in nature for centuries. New Phyto 214:1398–1402
Burden RF, Randerson PF (1972) Quantitative studies of the effects of human trampling on vegetation as an aid to the management of semi-natural areas. J Appl Ecol 9:439–457. https://doi.org/10.2307/2402445
Carson WP, Peterson CJ (1990) The role of litter in an old-field community: impact of litter quantity in different seasons on plant species richness and abundance. Oecologia 85:8–13. https://doi.org/10.1007/BF00317337
Clavel J, Julliard R, Devictor V (2010) Worldwide decline of specialist species: toward a global functional homogenization? Front Ecol Environ 9:222–228. https://doi.org/10.1890/080216
Correa Ayram CA, Mendoza ME, Etter A, Salicrup DRP (2016) Habitat connectivity in biodiversity conservation: a review of recent studies and applications. Prog Phys Geogr 40:7–37. https://doi.org/10.1177/0309133315598713
Cramp L, Evershed R, Lavento M, Halinen M, Mannermaa K, Oinonen M, Kettunen J, Perola M, Onkamo P, Heyd V (2014) Neolithic dairy farming at the extreme of agriculture in northern Europe. Proc R Soc B 281:20140819. https://doi.org/10.1098/rspb.2014.0819
Crooks KR, Sanjayan M (2006) Connectivity conservation: maintaining connections for nature. Cambridge University Press, Cambridge
Dostálek J, Frantík T (2008) Dry grassland plant diversity conservation using low-intensity sheep and goat grazing management: case study in Prague (Czech Republic). Biodivers Conserv 17:1439–1454. https://doi.org/10.1007/s10531-008-9352-1
Ejrnæs R, Liira J, Poulsen RS, Nygaard B (2008) When has an abandoned field become a semi-natural grassland or heathland? Environ Manage 42:707–716. https://doi.org/10.1007/s00267-008-9183-6
European Commission, Directorate-General for Environment, Tsiripidis I, Piernik A, Janssen J et al (2017) European red list of habitats, part 2. Terr Freshw Habitats. https://doi.org/10.2779/091372
Ewald J (2003) The calcareous riddle: why are there so many calciphilous species in the Central European flora? Folia Geobot 38:357–366. https://doi.org/10.1007/BF02803244
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Finnish Forest Center (2021) Open-access national raster dataset depicting canopy height with 1m2-resolution. License CC BY 4.0. https://www.metsakeskus.fi/fi/avoin-metsa-ja-luontotieto/aineistot-paikkatieto-ohjelmille/paikkatietoaineistot. Accessed 15 Oct 2022
Frey SJK, Strong AM, McFarland KP (2011) The relative contribution of local habitat and landscape context to metapopulation processes: a dynamic occupancy modeling approach. Ecography 35:581–589. https://doi.org/10.1111/j.1600-0587.2011.06936.x
García de León D, Neuenkamp L, Gertz M, Oja E, Zobel M (2016) Secondary succession in alvar grasslands—do changes in vascular plant and cryptogam communities correspond? Folia Geobot 51:285–296. https://doi.org/10.1007/s12224-016-9260-1
Geological Survey of Finland (2017) Bedrock of Finland (map). https://gtkdata.gtk.fi/Kalliopera/. Accessed 15 Oct 2022
Gheza G, Assini S, Marini L, Nascimbene J (2018) Impact of an invasive herbivore and human trampling on lichen-rich dry grasslands: soil-dependent response of multiple taxa. Sci Total Environ 639:633–639. https://doi.org/10.1016/j.scitotenv.2018.05.191
Glime JM (2017) Ecophysiology of development: spore germination. In: Glime JM (ed) Bryophyte Ecology. Volume 1. 5-2-1 Physiological Ecology
Hamberg L, Lehvävirta S, Malmivaara-Lämsä M, Rita H, Kotze J (2008) The effects of habitat edge and trampling on understorey vegetation in urban forests in Helsinki, Finland. Appl Veg Sci 11:83–98. https://doi.org/10.1111/j.1654-109X.2008.tb00207.x
Hanski I (2011) Habitat loss, the dynamics of biodiversity, and a perspective on conservation. Ambio 40:248–255. https://doi.org/10.1007/s13280-011-0147-3
Harrell FE Jr, Dupont C (2019) Hmisc: Harrell Miscellaneous. R package version 4.2-0
Haworth BJ, Ashmore MR, Headley AD (2007) Effects of nitrogen deposition on bryophyte species composition of calcareous grasslands. Water Air Soil Pollut Focus 7:111–117. https://doi.org/10.1007/s11267-006-9071-0
Helm A, Hanski I, Pärtel M (2006) Slow response of plant species richness to habitat loss and fragmentation. Ecol Lett 9:72–77. https://doi.org/10.1111/j.1461-0248.2005.00841.x
Husáková I, Münzbergová Z (2014) Relative importance of current and past landscape structure and local habitat conditions for plant species richness in dry grassland-like forest openings. PLoS ONE 9:e97110. https://doi.org/10.1371/journal.pone.0097110
Hydbom S, Ödman AM, Olsson PA, Cronberg N (2012) The effects of pH and disturbance on the bryophyte flora in calcareous sandy grasslands. Nord J Bot 30:446–452. https://doi.org/10.1111/j.1756-1051.2012.01463.x
Hyvärinen E, Juslén A, Kemppainen E, Uddström A, Liukko UM (2019) The 2019 red list of Finnish species. Ministry of the Environment & Finnish Environment Institute, Helsinki
Ingerpuu N, Liira J, Pärtel M (2005) Vascular plants facilitated bryophytes in a grassland experiment. Plant Ecol 180:69–75. https://doi.org/10.1007/s11258-005-2508-0
Jägerbrand AK, Alatalo JM (2015) Effects of human trampling on abundance and diversity of vascular plants, bryophytes and lichens in alpine heath vegetation, Northern Sweden. Springerplus 4:1–12. https://doi.org/10.1186/s40064-015-0876-z
Janišová M, Michalcová D, Bacaro G, Ghisla A (2014) Landscape effects on diversity of semi-natural grasslands. Agr Ecosyst Environ 182:47–58. https://doi.org/10.1016/j.agee.2013.05.022
Johnston AE, Goulding KWT, Poulton PR (1986) Soil acidification during more than 100 years under permanent grassland and woodland at Rothamsted. Soil Use Manag 2(1):3–10. https://doi.org/10.1111/j.1475-2743.1986.tb00669.x
Jokinen P, Pirinen P, Kaukoranta J-P, Kangas A, Alenius P, Eriksson P, Johansson M, Wilkman S (2021) Tilastoja Suomen ilmastosta ja merestä 1991–2020. Raportteja—Rapporter—Reports 2021:8. Ilmatieteen laitos—Finnish Meteorological Institute. https://doi.org/10.35614/isbn.9789523361485
Kalliola R (1973) Suomen Kasvimaantiede. Porvoo. (In Finnish)
Karlík P, Poschlod P (2019) Identifying plant and environmental indicators of ancient and recent calcareous grasslands. Ecol Ind 104:405–421. https://doi.org/10.1016/j.ecolind.2019.05.016
Király I, Nascimbene J, Tinya F, Ódor P (2013) Factors influencing epiphytic bryophyte and lichen species richness at different spatial scales in managed temperate forests. Biodivers Conserv 22:209–223. https://doi.org/10.1007/s10531-012-0415-y
Kolb A, Diekmann M (2005) Effects of life-history traits on responses of plant species to forest fragmentation. Cons Biol 19:929–938. https://doi.org/10.1111/j.1523-1739.2005.00065.x
Kontula T, Raunio A (2018) Threatened habitat types in Finland 2018, Red List of habitats Part I: Results and basis for assessment. The Finnish Environment 5/2018. http://urn.fi/URN:ISBN:978-952-11-5110-1
Lampinen J (2017) The present size, protection status, threats and restoration requirements of Carex caryophyllea -populations in continental Finland. Memoranda Societatis pro Fauna Et Flora Fennica 93:106–135
Lampinen J (2019) Disturbance, microclimate and historical habitat connectivity determine the population performance of the threatened grassland specialist Carex caryophyllea in remnant grasslands. Nord J Bot 37:e02175. https://doi.org/10.1111/njb.02175
Lampinen J, Ruokolainen K, Huhta AP (2015) Urban power line corridors as novel habitats for grassland and alien plant species in South-Western Finland. PLoS ONE 10(11):e0142236. https://doi.org/10.1371/journal.pone.0142236
Lampinen J, Heikkinen RK, Manninen P, Ryttäri T, Kuussaari M (2018) Importance of local habitat conditions and past and present habitat connectivity for the species richness of grassland plants and butterflies in power line clearings. Biodivers Conserv 27:217–233. https://doi.org/10.1007/s10531-017-1430-9
Lampinen J, Koskela A (2016) Juhannuskukkulan perinnebiotoopin kasvillisuus, luontotyypit ja hoitosuunnitelma. Turun kaupungin ympäristöjulkaisuja 4/2016.
Lazzaro L, Lastrucci L, Viciani D, Benesperi R, Gonnelli V, Coppi A (2020) Patterns of change in α and β taxonomic and phylogenetic diversity in the secondary succession of semi-natural grasslands in the Northern Apennines. PeerJ 8:e8683. https://doi.org/10.7717/peerj.8683
Lebrija-Trejos E, Meave JA, Poorter L, Pérez-García EA, Bongers F (2010) Pathways, mechanisms and predictability of vegetation change during tropical dry forest succession. Perspect Plant Ecol Evol Syst 12:267–275. https://doi.org/10.1016/j.ppees.2010.09.002
Lehmair TA, Pagel E, Poschlod P, Reisch C (2020) Surrounding landscape structures, rather than habitat age, drive genetic variation of typical calcareous grassland plant species. Landscape Ecol 35:2881–2893. https://doi.org/10.1007/s10980-020-01120-7
Lehtomaa L, Ahonen I, Hakamäki H, Häggblom M, Jutila H, Järvinen C, Kemppainen R, Kondelin H, Laitinen T, Lipponen M, Mussaari M, Pessa J, Raatikainen KJ, Raatikainen K, Tuominen S, Vainio M, Vieno M, Vuomajoki M (2018a) Seminatural grasslands. In: Kontula T, Raunio A (eds,) Threatened habitat types in Finland 2018a, Red List of habitats Part I: Results and basis for assessment. The Finnish Environment 5/2018a. http://urn.fi/URN:ISBN:978-952-11-5110-1
Lehtomaa L, Ahonen I, Hakamäki H, Jantunen J, Jutila H, Järvinen C, Kemppainen R, Kondelin H, Laitinen T, Lipponen M, Mussaari M, Pessa J, Raatikainen KJ, Raatikainen K, Tuominen S, Vainio M, Vieno M, Vuomajoki M (2018b) Seminatural grasslands. In: Kontula T, Raunio A (eds) Threatened habitat types in Finland 2018b, Red List of habitats Part II: Descriptions of habitat types. The Finnish Environment 5/2018b. http://urn.fi/URN:ISBN:978-952-361-256-3
Li X, Hajek O, LaRoe J, Wilkins K, Knapp A, Smith MD (2020) Fire frequency influenced grazed grasslands’ resistance and resilience to extreme drought. EGU General Assembly 2020, Online, 4–8 May 2020, EGU2020-12758. https://doi.org/10.1111/j.1365-2745.2007.01232.x
Lindborg R (2007) Evaluating the distribution of plant life-history traits in relation to current and historical landscape configurations. J Ecol 95:555–564. https://doi.org/10.1111/j.1365-2745.2007.01232.x
Lindborg R, Eriksson O (2004) Historical landscape connectivity affects present plant species diversity. Ecology 85:18401845. https://doi.org/10.1890/04-0367
Löbel S, Dengler J, Hobohm C (2006) Species richness of vascular plants, bryophytes and lichens in dry grasslands: the effects of environment, landscape structure and competition. Folia Geobot 41:377–393. https://doi.org/10.1007/BF02806555
Matter SF, Ezzeddine M, Duermit E, Mashburn J, Hamilton R, Lucas T, Roland J (2009) Interactions between habitat quality and connectivity affect immigration, but not abundance of population growth of the butterfly, Parnassius smintheus. Oikos 118:1461–1470. https://doi.org/10.1111/j.1600-0706.2009.17438.x
McCune JL, Frendo CJ, Ramadan M, Baldwin LK (2021) Comparing the effect of landscape context on vascular plant and bryophyte communities in a human-dominated landscape. J Veg Sci 32:e12932. https://doi.org/10.1111/jvs.12932
Müller J, Klaus VH, Kleinebecker T, Prati D, Hölzel N, Fischer M (2012) Impact of land-use intensity and productivity on bryophyte diversity in agricultural grasslands. PLoS ONE 7(12):e51520. https://doi.org/10.1371/journal.pone.0051520
Nally RM, Walsh CJ (2004) Hierarchical partitioning public-domain software. Biodivers Conserv 13:659–660. https://doi.org/10.1023/B:BIOC.0000009515.11717.0b
Oldén A, Raatikainen KJ, Tervonen K, Halme P (2016) Grazing and soil pH are biodiversity drivers of vascular plants and bryophytes in boreal wood-pastures. Agr Ecosyst Environ 222:171–184. https://doi.org/10.1016/j.agee.2016.02.018
Olea PP, Mateo-Tomás P, de Frutos Á (2010) Estimating and modelling bieas of the hierachical partitioning public-domain software: implications in environmental management and conservation. PLoS ONE. https://doi.org/10.1371/journal.pone.0011698
Öster M, Cousins SAO, Eriksson O (2007) Size and heterogeneity rather than landscape context determine plant species richness in semi-natural grasslands. J Veg Sci 18:859–868. https://doi.org/10.1111/j.1654-1103.2007.tb02602.x
Palpurina S, Wagner V, von Wehrden H, Hájek M, Horsák M, Brinkert A, Hölzel N, Wesche K, Kamp J, Hájková P, Danihelka J, Lustyk P, Merunková K, Preislerová Z, Kočí M, Kubešová S, Cherosov M, Ermakov N, German D, Gogoleva P, Lashchinsky N, Martynenko V, Chytrý M (2017) The relationship between plant species richness and soil pH vanishes with increasing aridity across Eurasian dry grasslands. Glob Ecol Biogeogr 26:425–434. https://doi.org/10.1111/geb.12549
Paradis E, Schliep K (2018) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528. https://doi.org/10.1093/bioinformatics/bty633
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci Discuss 11:1633–1644. https://doi.org/10.5194/hess-11-1633-2007
Peintinger M, Bergamini A (2006) Community structure and diversity of bryophytes and vascular plants in abandoned fen meadows. Plant Ecol 185:1–17. https://doi.org/10.1007/s11258-005-9079-y
Popiela A, Łysko A, Sotek Z, Ziarnek K (2015) Preliminary results of studies on the distribution of invasive alien vascular plant species occurring in semi-natural and natural habitats in NW Poland. Biodivers Res Conserv 37:21–35. https://doi.org/10.1515/biorc-2015-0003
Pykälä J (2001) Maintaining biodiversity through traditional animal husbandry. Finnish Environ 495:1–205 (In Finnish)
Pykälä J, Luoto M, Heikkinen RK, Kontula T (2005) Plant species richness and persistence of rare plants in abandoned semi-natural grasslands in northern Europe. Basic Appl Ecol 6:25–33. https://doi.org/10.1016/j.baae.2004.10.002
QGIS Development Team (2018) QGIS Geographic Information System. Open Source Geospatial Foundation Project. 2018. Available: http://qgis.osgeo.org/.
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Raatikainen KM, Heikkinen RK, Luoto M (2009) Relative importance of habitat area, connectivity, management and local factors for vascular plants: spring ephemerals in boreal semi-natural grasslands. Biodivers Conserv 18:1067–1085. https://doi.org/10.1007/s10531-008-9462-9
Redhead JW, Sheail BJM, Ferreruela A, Walker KJ, Pywell RF, Fraser L (2014) The natural regeneration of calcareous grassland at a landscape scale: 150 years of plant community re-assembly on Salisbury Plain, UK. Appl Veg Sci 17:408–418. https://doi.org/10.1111/avsc.12076
Reitalu T, Purschke O, Johansson LJ, Hall K, Sykes MT, Prentice HC (2012) Responses of grassland species richness to local and landscape factors depend on spatial scale and habitat specialization. J Veg Sci 23:41–51. https://doi.org/10.1111/j.1654-1103.2011.01334.x
Ryttäri T, Kalliovirta M, Lampinen R (2012) Suomen uhanalaiset kasvit. Tammi, Helsinki
Schmid BC, Poschlod P, Prentice HC (2017) The contribution of successional grasslands to the conservation of semi-natural grasslands species—a landscape perspective. Biol Conserv 206:112–119. https://doi.org/10.1016/j.biocon.2016.12.002
Simmel J, Karlík P, Poschlod P (2021) Land-use history affects vascular plant composition of calcareous grasslands—does it affect cryptogam composition, too? Ecol Ind 124:e107408. https://doi.org/10.1016/j.ecolind.2021.107408
Snäll T, Hagström A, Rudolphi J, Rydin H (2004) Distribution pattern of the epiphyte Neckera pennata on three spatial scales—importance of past landscape structure, connectivity and local conditions. Ecography 27:757–766. https://doi.org/10.1111/j.0906-7590.2004.04026.x
Štajerová K, Šmilauer P, Brůna J, Pyšek P (2017) Distribution of invasive plants in urban environment is strongly spatially structured. Landscape Ecol 32:681–692. https://doi.org/10.1007/s10980-016-0480-9
Takala T, Tahvanainen T, Kouki J (2012) Can re-establishment of cattle grazing restore bryophyte diversity in abandoned mesic semi-natural grasslands? Biodivers Conserv 21:981–992. https://doi.org/10.1007/s10531-012-0234-1
Takala T, Tahvanainen T, Kouki J (2014) Grazing promotes bryophyte species-richness in semi-natural grasslands. Ann Bot Fenn 51:148–160. https://doi.org/10.5735/085.053.0102
The National Archives Service of Finland (NASF) (2022) Topografiakarttojen kokoelma. Venäläiset topografiakartat 1:21 000 (1870-1907). https://astia.narc.fi/uusiastia/. Accessed 15 Oct 2022
Tilman D, El Haddi A (1992) Drought and biodiversity in grasslands. Oecologia 89:257–264. https://doi.org/10.1007/BF00317226
Tinya F, Márialigeti S, Király I, Németh B, Odor P (2009) The effect of light conditions on herbs, bryophytes and seedlings of temperate mixed forests in Őrség, Western Hungary. Plant Ecol 204:69–81. https://doi.org/10.1007/s11258-008-9566-z
Tyler T, Bengtsson F, Dahlberg CJ, Lönnell N, Hallingbäck T, Reitalu T (2018) Determinants of bryophyte species composition and diversity on the Great Alvar of Öland, Sweden. J Bryol 40:12–30. https://doi.org/10.1080/03736687.2017.1412387
Uroy L, Ernoult A, Mony C (2019) Effect of landscape connectivity on plant communities: a review of response patterns. Landscape Ecol. https://doi.org/10.1007/s10980-019-00771-5
Van Kleunen M, Dawson W, Essl F, Pergl J, Winter M, Weber E, Kreft H, Weigelt P, Kartesz J, Nishino M, Antonova LA, Barcelona JF, Cabezas FJ, Cárdenas D, Cárdenas-Toro J, Castaño N, Chacón E, Chatelain C, Ebel AL, Figueiredo E, Fuentes N, Groom QJ, Henderson L, Inderjit S, Kupriyanov A, Masciadri S, Meerman J, Morozova O, Moser D, Nickrent DL, Patzelt A, Pelser PB, Baptiste MP, Poopath M, Schulze M, Seebens H, Shu W, Thomas J, Velayos M, Wieringa JJ, Pyšek P (2015) Global exchange and accumulation of non-native plants. Nature 525:100–103. https://doi.org/10.1038/nature14910
Verhoeven KJ, Simonsen KL, McIntyre LM (2005) Implementing false discovery rate control: increasing your power. Oikos 108:643–647. https://doi.org/10.1111/j.0030-1299.2005.13727.x
Vestin JLK, Nambu K, van Hees PAW, Bylund LUS (2006) The influence of alkaline and non-alkaline parent material on soil chemistry. Geoderma 135:97–106. https://doi.org/10.1016/j.geoderma.2005.11.013
Virtanen R, Johnston A, Crawley M, Edwards G (2000) Bryophyte biomass and species richness on the Park Grass experiment, Rothamsted, UK. Plant Ecol 151:129–141. https://doi.org/10.1023/A:1026533418357
Wintle BA, Kujala H, Whitehead A, Cameron A, Veloz S, Kukkala A, Moilanen A, Gordon A, Lentinit PE, Cadenhead NCR, Bekessy SA (2019) Global synthesis of conservation studies reveals the importance of small habitat patches for biodiversity. PNAS 116:909–914. https://doi.org/10.1073/pnas.1813051115
Zulka KP, Abensperg-Traun M, Milasowszky N, Bieringer G, Gereben-Krenn BA, Holzinger W, Hölzler G, Rabitsch W, Reischütz A, Querner P, Sauberer N, Scmitzberger I, Willner W, Wrbka T, Zechmeister H (2014) Species richness in dry grassland patches of eastern Austria: a multi-taxon study on the role of local, landscape and habitat quality variables. Agr Ecosyst Environ 182:25–46. https://doi.org/10.1016/j.agee.2013.11.016
Funding
Open Access funding provided by University of Jyväskylä (JYU). Author Järvenpää S. has received grants for this research from Societas Biologica Fennica Vanamo and Societas pro Fauna et Flora Fennica and a one-week residence grant from University of Jyväskylä.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection was performed by SJ and analysis were performed by JL and SJ. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Daniel Sanchez Mata.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Järvenpää, S., Kytöviita, MM., Pitkämäki, T. et al. Contrasting responses of vascular plants and bryophytes to present and past connectivity in unmanaged grasslands. Biodivers Conserv 32, 139–162 (2023). https://doi.org/10.1007/s10531-022-02492-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-022-02492-9