Abstract
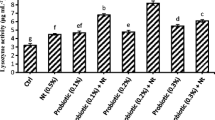
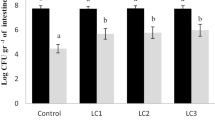
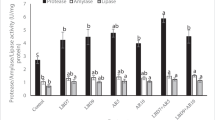
The dietary supplementation of acidifier in Cirrhinus mrigala juvenile was evaluated in terms of change in growth, autochthonous intestinal microbiota and haemato-biochemical parameters for period of 60 days. Acidifier was added at a level of 0 (control diet), 0.5, 1 and 1.5% to the practical pellet diet. At the end of the experiment, growth parameters, survival, bacillus bacterial population, haematological and immunological parameters were analysed. Higher WG%, SGR and PER and lower FCR (P < 0.05) was found in group fed 1.5% OA. Lysozyme activity and respiratory burst activity (nitroblue tetrazolium) were significantly affected by dietary 1.5% acidifier (P < 0.05). In fish fed on the diet with 1.5% acidifier showed a significant increase of total heterotrophic autochthonous bacterial and Bacillus levels (P < 0.05) compared with those fed on the diets supplemented with organic acid. In addition to increase in WBC, RBC, haemoglobin and platelets count levels were observed in this group. Relative % survival rate was found to be higher in the fish fed on 1.5% acidifier after challenging with Aeromonas hydrophilla bacteria. These results indicated that dietary supplementation of acidifier at a dose of 1.5% improved growth performance and enhanced the beneficial intestinal microbiota and stimulate immune response of Cirrhinus mrigala juvenile.

Similar content being viewed by others
References
Alexander C, Sahu NP, Pal AK, Akhtar MS (2011) Haemato-immunological and stress responses of Labeo rohita (Hamilton) fingerlings: effect of rearing temperature and dietary gelatinized carbohydrate. J Anim Physiol Anim Nutr 95:653–663
APHA– AWWA – WEF (1998) Standard methods for the examination of water and wastewater, 20th edn. (Clesceri LS, Greenberg AE, Eaton AD. Ed.). American Public Health Association, American Water Works Association, Water Environment Federation, Washington DC
Balca’zar J, De Blas I, Ruiz-Zarzuela I, Vendrell D, Girone’s O, Muzquiz J (2007) Enhancement of the immune response and protection induced by probiotic lactic acid bacteria against furunculosis in rainbow trout (Oncorhynchus mykiss). FEMS Immunol Med Microbiol 51:185–193
Ballas ZK, Rasmuseen WL, Krieg AM (1996) Induction of natural killer activity in murine and human cells by CpG motifs in oligodeoxy nucleotides and bacterial DNA. J Immunol 157:1840–1850
Baruah K, Sahu NP, Pal AK, Jain KK, Debnath D, Mukherjee SC (2007) Dietary microbial phytase and citric acid synergistically enhances nutrient digestibility and growth performance of Labeo rohita (Hamilton) juveniles at sub-optimal protein level. Aquac Res 38:109–120
Bergy DH (1986) Bergey’s manual of systemic bacteriology. Vol. 2 (Holt JH, Sneath PH, Krieg NR. eds) pp. 636. Lipponcott Williams and Wilkins, ISBN 0683078933
Booth IR, Stratford M (2003) Acidulants and low pH. In: Russell NJ, Gould GW (eds) Food Preservatives. Kluwer Academic/Plenum Publishers, New York, pp 25–47
Chace HJ, Hooker NA, Mildestein KL, Krieg AM, Cowdery JS (1997) Bacterial DNA-induced NK cell IFN-g production is dependent on macrophage secretion of IL-12. Clin Immunol Immunopathol 84:185–193
Christiansen R, Lückstädt C (2008) Effects of different dosages of potassium diformate in fishmeal on the performance of Atlantic salmon Salmo salar. Abstract CD-Rom, World Aquacult. Soc. 19–23 may 2008, Busan, Korea
Da Saliva BC, Vieira FN, Mourino JP, Ferrira GS, Seiffert WQ (2013) Salts of organic acids selection by multiple characteristics for marine shrimp nutrition. Aquaculture:107–110
De Wet L (2005) Can organic acids effectively replace antibiotic growth promotants in diets for rainbow trout Oncorhynchus mykiss raised under sub-optimal water temperatures? Abstract CD-Rom, J. World Aquacult. Soc. 9–13 may 2005; Bali, Indonesia
Defoirdt T, Boon N, Sorgeloos P, Verstraete W, Bossier P (2009) Short-chain fatty acids and poly-b-hydroxyalkanoates: (new) biocontrol agents for a sustainable animal production. Biotechnol Adv 27:680–685
Denev S, Staykov Y, Moutafchieva R, Beev G (2009) Rev. microbial ecology of gastrointestinal tract of fish and the potential application of probiotics and prebiotics in finfish aquaculture. In Aquacult Res 1:1–29
deWet L (2005) Organic acids as performance enhancers. Aqua Feeds: Formulation and Beyond 2:12–14
Doumas BT, Watson W, Biggs HG (1971) Albumin standards and measurement of serum albumin with bromocresol green. Clin Chim Acta 31:87–96
Ellis AE (1990) Lysozyme assays. In: Stolen JS, Fletcher TC, Anderson BS, Van Muiswinkel WB (eds) Techniques in fish immunology. SOS Publications, Fair Haven, pp 101–103
Fange R, Lundblad G, Lind J (1976) Lysozyme and chitinase in blood and lymphomyeloid tissues of marine fish. Mar Biol 36:277–282
Fletcher TC, White A (1973) Lysozyme activity in the plaice (Pleuronectes platessa L). Experientia 29:1283–1295
Guimaraes TM, Moriel DG, Machado IP, Fadel Picheth CMT, Bonfim TMB (2006) Isolation and characterization of Saccharomyces cerevisiae strain of winery interest. Brazilian J Pharm Sci 42:119–126
Halver JE (1976) The nutritional requirements of cultivated warmwater and coldwater fish species. Paper No: 31. FAO Technical conference on Aquaculture, Kyoto, 26 May–2 June, p 9
Hossain MA, Pandey A, Satoh S (2007) Effects of organic acids on growth and phosphorus utilization in red sea bream Pagrus major. Fish Sci 73:1309–1317
Jhingran VG (1991) Fish culture in fresh water pond. Fish and fisheries of India (ed. V. G. Jhingran), III edn. Hindustan Publishing Corporation, New Delhi, p 276
Jun-sheng L, Jian-lin L, Ting-ting W (2006) Ontogeny of protease, amylase and lipase in the alimentary tract of hybrid juvenile tilapia Oreochromis niloticus X Oreochromis aureus. Fish Physiol Biotech 32:295–303
Lie Ø, Evensen Ø, Sørensen A, Frøysadal E (1989) Study on lysozyme activity in some fish species. Dis Aquat Org 6:1–5
Lückstädt C (2007) Effect of organic acid containing additives in worldwide aquaculture: sustainable production the non-antibiotic way. In: Lückstädt C (ed) Acidifiers in animal nutrition: a guide for feed preservation and acidification to promote animal performance. Nottingham University Press, Nottingham, pp 71–79
Lückstädt C (2008) Dietary organic acids as feed additive for tilapia (Oreochromis niloticus) culture. Program and Abstracts, 6th conference of the Society of Ichthyology, 13–15 March 2008, Munich, Germany. p. 34–35
Magnadottir B (2006) Innate immunity of fish (overview). Fish Shellfish Immunol 20:137–151
Misra S, Sahu NP, Pal AK, Xavier B, Kumar S, Mukherjee SC (2006) Pre- and postchallenge immuno-haematological changes in Labeo rohita juveniles fed gelatinised or non-gelatinised carbohydrate with n-3 PUFA. Fish Shellfish Immunol 21:346–356
Ng WK, Koh CB, Sudesh K, Siti-Zahrah A (2009) Effects of dietary organic acids on growth, nutrient digestibility and gut microflora of red hybrid tilapia, Oreochromis sp., and subsequent survival during a challenge test with Streptococcus agalactiae. Aquac Res 40:1490–1500
NRC (2011) Nutrient requirements of fish and shrimp. National Academies Press, Washington, DC
Owen MAG, Waines P, Bradley G, Davies S (2006) The effect of dietary supplementation of sodium butyrate on the growth and microflora of Clarias gariepinus (Burchell 1822). In: proceedings of the XII international symposium fish nutrition and feeding; 28 May–1 June 2006. p. 149
Petkam R, Lückstädt C, Nittayachit P, Sadao S, Encarnacao P (2008) Evaluation of a dietary organic acid blend on tilapia Oreochromis niloticus growth performance. Abstract CD-rom, world Aquacult Soc 19–23 may 2008, Busan, Korea
Ramli N, Heindl U, Sunanto S (2005) Effect of potassium-diformate on growth performance of tilapia challenged with Vibrio anguillarum. Abstract CD-rom J. World Aquac. Soc. 9–13 May 2005; Bali, Indonesia
Reinhold JG (1953) Manual determination of serum total protein, albumin and globulin fractions by biuret method. In: Reiner M (ed) Standard method of clinical chemistry. Academic Press, New York, p 88
Ringo E (1991) Effects of dietary lactate and propionate on growth and digesta in Artic charr, Salvelinus alpinus (L.) Aquaculture 96:321–333
Ringø E (1992) Effects of dietary formate and acetate on growth and lipid digestibility in Arctic charr, Salvelinus alpinus (L.). Fiskeridirektoratets Skrifter. Serie Ernaering 5:17–24
Roberts RJ (1978) The pathophysiology and systemic pathology of teleosts. In: Roberts RJ (ed) Fish pathology. Bailliere Tindal, London, pp 55–91
Russell JB, Diez-Gonzales F (1998) The effects of fermentation acids on bacterial growth. Adv Microb Physiol 39:205–234
Secombes CJ (1990) Isolation of salmonid macrophage and analysis of their killing activity. In: Stolen JSTC, Fletcher DP, Anderson BS, Van Muiswinkel WB (eds) Techniques in fish immunology. SOS Publication, Fair Haven, pp 137–152
Secombes CJ (1996) The nonspecific immune system: cellular defences. In: Iwama G, Nakanishi T (eds) The fish immune system, organism, pathogen and environment. Academic Press, Toronto, pp 63–103
Sparwasser T, Miethe T, Lipford G, Erdmann A, Hawker H, Heeg K (1997) Macrophage sense pathogens via DNA motifs: induction of tumor necrosis factor-a-mediated shock. Eur J Immunol 27:1671–1179
Stasiack AS, Bauman CP (1996) Neutrophil activity as a potent indicator concomitant analysis. Fish Shellfish Immunol 37:539
Tung HT, Koshio S, Teshima S, Ishikawa M, Ren T, Phuong NDT (2006) Effects of heat-killed lactobacillus plantarum on Kuruma shrimp Masurpenaeus japonicas juveniles. XII international symposium fish Nutrition and Feeding, May 28—June 1, 2006. Book of abstracts. p. 46
Vielma J, Lall S (1997) Dietary formic acid enhances apparent digestiblity of minerals in rainbow trout, Oncorhynchus mykiss (Walbaum). Aquac Nutr 3:265–278
Vladimrov VL (1968) Immunity in fish. Bull Off Int Epizoot 69:1365–1372
Wiegertjes GF, Stet RJM, Parmentier HK, Muiswinkle WB (1996) Immunogenetics and disease resistance in fish: a comparative approach. Dev Comp Immunol 20:365–381
Williams MJ, Bell JD, Gupta MV, Dey M, Ahmed M, Prein M, Child S, Gardiner PR, Brummet R, Jamu D (2000) Responsible aquaculture can aid food problems. Nature 406:673
Zhou Z, Liu Y, Wang P, He S, Yao B, Gao X, Wang X (2008) The effects of dietary potassium diformate on growth performance, feed conversion and intestinal microbiota of hybrid tilapia. Abstract, XIII international symposium on fish nutrition and feeding, Florianopolis, Brazil
Zhou Z, Liu Y, He S, Shi P, Gao X, Yao B, Ringo E (2009) Effects of dietary potassium diformate (KDF) on growth diformate (KDF) on growth performance, feed conversion and intestinal bacterial community of hybrid tilapia (Oreochromis niloticus x O. aureus). Aquaculture 291:89–94
Acknowledgements
The authors are grateful to Dr. W.S. Lakra, Director, Central Institute of Fisheries Education, Mumbai, for providing facilities for carrying out the work. The first author is grateful to the Indian Council of Agricultural Research, New Delhi for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, P., Jain, K.K., Sardar, P. et al. Dietary supplementation of acidifier: effect on growth performance and haemato-biochemical parameters in the diet of Cirrhinus mrigala juvenile. Aquacult Int 25, 2101–2116 (2017). https://doi.org/10.1007/s10499-017-0176-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-017-0176-4