Abstract
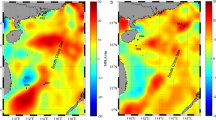
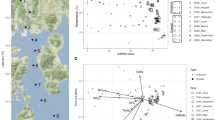
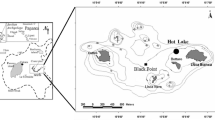
Cold seeps are unique ecosystems that are generally characterized by high salinity and reducing solutions. Seepage fluid, the major water influx of this system, contains hypersaline water, sediment pore water, and other components. The Thuwal cold seeps were recently discovered on the continental margin of the Red Sea. Using 16S rRNA gene pyro-sequencing technology, microbial communities were investigated by comparing samples collected in 2011 and 2013. The results revealed differences in the microbial communities between the two sampling times. In particular, a significantly higher abundance of Marine Group I (MGI) Thaumarchaeota was coupled with lower salinity in 2013. In the brine pool, the dominance of Desulfobacterales in 2011 was supplanted by MGI Thaumarchaeota in 2013, perhaps due to a reduced supply of hydrogen sulfide from the seepage fluid. Collectively, this study revealed a difference in water components in this ecosystem between two sampling times. The results indicated that the seawater in this cold seep displayed a greater number of characteristics of normal seawater in 2013 than in 2011, which might represent the dominant driving force for changes in microbial community structures. This is the first study to provide a temporal comparison of the microbial biodiversity of a cold seep ecosystem in the Red Sea.




Similar content being viewed by others
References
Batang ZB et al (2012) First discovery of a cold seep on the continental margin of the central Red Sea. J Mar Syst 94:247–253. doi:10.1016/j.jmarsys.2011.12.004
Berg IA, Kockelkorn D, Buckel W, Fuchs G (2007) A 3-hydroxypropionate/4-hydroxybutyrate autotrophic carbon dioxide assimilation pathway in Archaea. Science 318:1782–1786. doi:10.1126/science.1149976
Bower AS (2009) R/V Oceanus Voyage 449-6 Red Sea Atlantis II Deep Complex Area 19 October–1 November 2008
Brochier-Armanet C, Boussau B, Gribaldo S, Forterre P (2008) Mesophilic crenarchaeota: proposal for a third archaeal phylum, the Thaumarchaeota. Nat Rev Microbiol 6:245–252. doi:10.1038/Nrmicro1852
Caporaso JG, Bittinger K, Bushman FD, DeSantis TZ, Andersen GL, Knight R (2010a) PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatics 26:266–267. doi:10.1093/bioinformatics/btp636
Caporaso JG et al (2010b) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. doi:10.1038/nmeth.f.303
Delong EF (1992) Archaea In Coastal Marine Environments. Proc Natl Acad Sci USA 89:5685–5689. doi:10.1073/pnas.89.12.5685
Eder W, Schmidt M, Koch M, Garbe-Schonberg D, Huber R (2002) Prokaryotic phylogenetic diversity and corresponding geochemical data of the brine-seawater interface of the Shaban Deep. Red Sea Environ Microbiol 4:758–763
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461. doi:10.1093/bioinformatics/btq461
Fuhrman JA, Davis AA (1997) Widespread archaea and novel bacteria from the deep sea as shown by 16S rRNA gene sequences. Mar Ecol Prog Ser 150:275–285. doi:10.3354/Meps150275
Glover AG et al (2010) Temporal change in deep-sea benthic ecosystems: a review of the evidence from recent time-series studies. Adv Mar Biol 58:1–95. doi:10.1016/B978-0-12-381015-1.00001-0
Greene AC, Patel BK, Sheehy AJ (1997) Deferribacter thermophilus gen. nov., sp. nov., a novel thermophilic manganese- and iron-reducing bacterium isolated from a petroleum reservoir. Int J Syst Bacteriol 47:505–509
Haas BJ et al (2011) Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res 21:494–504. doi:10.1101/gr.112730.110
Hallam SJ, Mincer TJ, Schleper C, Preston CM, Roberts K, Richardson PM, DeLong EF (2006) Pathways of carbon assimilation and ammonia oxidation suggested by environmental genomic analyses of marine Crenarchaeota. Plos Biol 4:520–536. doi:10.1371/Journal.Pbio.0040095
Hammer Ø, Harper D, Ryan P (2001) Past: Paleontological Statistics Software Package for education and data analysis. Paleontología Electrónica 4: 1–9 URL: < http://palaeo-electronica.org/2001_1/past/issue1_01.html
Hansman RL, Griffin S, Watson JT, Druffel ER, Ingalls AE, Pearson A, Aluwihare LI (2009) The radiocarbon signature of microorganisms in the mesopelagic ocean. Proc Natl Acad Sci USA 106:6513–6518. doi:10.1073/pnas.0810871106
Hartmann M, Scholten JC, Stoffers P, Wehner F (1998) Hydrographic structure of brine-filled deeps in the Red Sea—new results from the Shaban Kebrit, Atlantis II, and Discovery Deep. Mar Geol 144:311–330. doi:10.1016/S0025-3227(97)00055-8
Hemleben C, Roether W, Stoffers P (1996) Östliches Mittelmeer, Rotes Meer, Arabisches Meer, Cruise No. 31, 30 December 1994–22 March 1995 METEOR-Ber:96-94
Hugler M, Huber H, Stetter KO, Fuchs G (2003) Autotrophic CO2 fixation pathways in archaea (Crenarchaeota). Arch Microbiol 179:160–173. doi:10.1007/s00203-002-0512-5
Karner MB, DeLong EF, Karl DM (2001) Archaeal dominance in the mesopelagic zone of the Pacific Ocean. Nature 409:507–510. doi:10.1038/35054051
Knittel K, Losekann T, Boetius A, Kort R, Amann R (2005) Diversity and distribution of methanotrophic archaea at cold seeps Applied And. Environ Microbiol 71:467–479. doi:10.1128/Aem.71.1.467-479.2005
Kvenvolden KA (1993) Gas Hydrates - Geological Perspective And Global Change. Rev Geophys 31:173–187. doi:10.1029/93rg00268
Lee OO et al (2014) In situ environment rather than substrate type dictates microbial community structure of biofilms in a cold seep system. Sci Rep 4:3587. doi:10.1038/srep03587
Levin LA (2005) Ecology of cold seep sediments: interactions of fauna with flow, chemistry and microbes Oceanogr. Mar Biol 43:1–46. doi:10.1201/9781420037449.Ch1
Miroshnichenko ML et al (2003) Deferribacter abyssi sp. nov., an anaerobic thermophile from deep-sea hydrothermal vents of the Mid-Atlantic Ridge. Int J Syst Evol Microbiol 53:1637–1641
Omoregie EO et al (2009) Microbial methane oxidation and sulfate reduction at cold seeps of the deep Eastern Mediterranean Sea. Mar Geol 261:114–127. doi:10.1016/j.margeo.2009.02.001
Orcutt BN, Sylvan JB, Knab NJ, Edwards KJ (2011) Microbial ecology of the dark ocean above, at, and below the seafloor. Microbiol Mol Biol Rev MMBR 75:361–422. doi:10.1128/MMBR.00039-10
Ouverney CC, Fuhrman JA (2000) Marine planktonic archaea take up amino acids. Appl Environ Microbiol 66:4829–4833
Paull CK et al (1984) Biological Communities at the Florida Escarpment Resemble Hydrothermal Vent Taxa. Science 226:965–967. doi:10.1126/science.226.4677.965
Quast C et al (2013) The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res 41:D590–D596. doi:10.1093/nar/gks1219
Ryan WB, Thorndike EM, Ewing M, Ross DA (1969) Suspended matter in the Red Sea brines and its detection by light scattering. Hot brines and recent heavy metal deposits in the Red Sea. Springer, New york, pp 153–157
Scholten J, Stoffers P, Garbe-Schönberg D, Moammar M (1999) Hydrothermal mineralization in the Red Sea Handbook of marine mineral deposits. CRC Press, Boca Raton, pp 369–395
Sibuet M, Olu K (1998) Biogeography, biodiversity and fluid dependence of deep-sea cold-seep communities at active and passive margins. Deep-Sea Res Part II 45:517. doi:10.1016/S0967-0645(97)00074-X
Slobodkina GB, Kolganova TV, Chernyh NA, Querellou J, Bonch-Osmolovskaya EA, Slobodkin AI (2009) Deferribacter autotrophicus sp. nov., an iron(III)-reducing bacterium from a deep-sea hydrothermal vent. Int J Syst Evol Microbiol 59:1508–1512. doi:10.1099/ijs.0.006767-0
Swan BK et al (2014) Genomic and metabolic diversity of Marine Group I Thaumarchaeota in the mesopelagic of two subtropical gyres. PLoS ONE 9:e95380. doi:10.1371/journal.pone.0095380
Tomlinson GA, Hochstein LI (1976) Halobacterium saccharovorum sp. nov., a carbohydrate-metabolizing, extremely halophilic bacterium. Can J Microbiol 22:587–591
Trefry JH, Presley BJ, Keeney-Kennicutt WL, Trocine RP (1984) Distribution and chemistry of manganese, iron, and suspended particulates in Orca Basin. Geo-Mar Lett 4:125–130
Tunnicliffe V, Juniper SK, Sibuet M (2003) Reducing environments of the deep-sea floor. Ecosyst World 81–110
Walker CB et al (2010) Nitrosopumilus maritimus genome reveals unique mechanisms for nitrification and autotrophy in globally distributed marine crenarchaea. Proc Natl Acad Sci USA 107:8818–8823. doi:10.1073/pnas.0913533107
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267. doi:10.1128/Aem.00062-07
Wang Y et al (2011a) Hydrothermally generated aromatic compounds are consumed by bacteria colonizing in Atlantis II Deep of the Red Sea. ISME J 5:1652–1659. doi:10.1038/ismej.2011.42
Wang Y, Yang JK, Lee OO, Li TG, Al-Suwailem A, Danchin A, Qian P-Y (2011b) Bacterial niche-specific genome expansion is coupled with highly frequent gene disruptions in deep-sea sediments. PLoS ONE 6:e29149. doi:10.1371/journal.pone.0029149
Wang Y, Tian RM, Gao ZM, Bougouffa S, Qian PY (2014a) Optimal Eukaryotic 18S and Universal 16S/18S Ribosomal RNA Primers and Their Application in a Study of Symbiosis. PLoS ONE. doi:10.1371/journal.pone.0090053
Wang Y et al (2014b) Diversity and distribution of eukaryotic microbes in and around a brine pool adjacent to the Thuwal cold seeps in the Red Sea. Front Microbiol 5:37. doi:10.3389/fmicb.2014.00037
Wang Y et al (2014c) Diversity and distribution of eukaryotic microbes in and around a brine pool adjacent to the Thuwal cold seeps in the Red Sea. Front Microbiol. doi:10.3389/Fmicb.2014.00037
Widdel F (1987) New Types Of Acetate-Oxidizing Sulfate-Reducing Desulfobacter Species, D-Hydrogenophilus Sp-Nov, D-Latus Sp-Nov, And D-Curvatus Sp-Nov. Arch Microbiol 148:286–291. doi:10.1007/Bf00456706
Wright JJ, Mewis K, Hanson NW, Konwar KM, Maas KR, Hallam SJ (2014) Genomic properties of Marine Group A bacteria indicate a role in the marine sulfur cycle. ISME J 8:455–468. doi:10.1038/ismej.2013.152
Wuchter C et al (2006) Archaeal nitrification in the ocean. Proc Natl Acad Sci USA 103:12317–12322. doi:10.1073/pnas.0600756103
Zhang W et al (2013) Adaptation of intertidal biofilm communities is driven by metal ion and oxidative stresses. Sci Rep 3:3180. doi:10.1038/srep03180
Zhang WP et al (2014) Species sorting during biofilm assembly by artificial substrates deployed in a cold seep system. Sci Rep 4:6647. doi:10.1038/srep06647
Acknowledgments
The authors are grateful to the crew members of R/V Aegaeo and Dr Abdulaziz Al-Suwailem and his team from Coastal and Marine Resources Core Laboratory from King Abdullah University of Science and Technology (KAUST) for providing technical assistance during the sample collection. This study was supported by grants from the National Basic Research Program of China (973 Program, No: 2012CB417304) and the China Ocean Mineral Resources R & D Association (COMRA) (DY125-15-R-01) as well as by a Global Collaborative Research Award from KAUST to Pei-Yuan Qian.
Author information
Authors and Affiliations
Corresponding author
Additional information
Accession Numbers All the sequence files are available from the NCBI SRA database (accession number SRP040612).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, B., Zhang, W., Tian, R. et al. Changing composition of microbial communities indicates seepage fluid difference of the Thuwal Seeps in the Red Sea. Antonie van Leeuwenhoek 108, 461–471 (2015). https://doi.org/10.1007/s10482-015-0499-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-015-0499-y