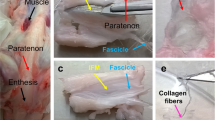
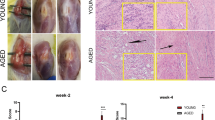
Abstract
Tendon injuries positively correlate with patient age, as aging has significant effects on tendon homeostatic maintenance and healing potential after injury. Vascularity is also influenced by age, with both clinical and animal studies demonstrating reduced blood flow in aged tissues. However, it is unknown how aging effects vascularity following tendon injury, and if this vascular response can be modulated through the delivery of angiogenic factors. Therefore, the objective of this study is to evaluate the vascular response following Achilles tendon injury in adult and aged rats, and to define the alterations to tendon healing in an aged model following injection of angiogenic factors. It was determined that aged rat Achilles tendons have a reduced angiogenesis following injury. Further, the delivery of vascular endothelial growth factor, VEGF, caused an increase in vascular response to tendon injury and improved mechanical outcome in this aged population. This work suggests that reduced angiogenic potential with aging may be contributing to impaired tendon healing response and that the delivery of angiogenic factors can rescue this impaired response. This study was also the first to relate changes in vascular response in an aged model using in vivo measures of blood perfusion to alterations in healing properties.








Similar content being viewed by others
References
Ackerman, J. E., I. Bah, J. H. Jonason, M. R. Buckley, and A. E. Loiselle. Aging does not alter tendon mechanical properties during homeostasis, but does impair flexor tendon healing. J. Orthop. Res. 35(12):2716–2724, 2017
Ahluwalia, A., J. Narula, M. K. Jones, X. Deng, and A. S. Tarnawski. Impaired angiogenesis in aging myocardial microvascular endothelial cells is associated with reduced importin alpha and decreased nuclear transport of HIF1 alpha: mechanistic implications. J. Physiol. Pharmacol. 61:133–139, 2010
Arnesen, S. M., and M. A. Lawson. Age-related changes in focal adhesions lead to altered cell behavior in tendon fibroblasts. Mech. Ageing Dev. 127:726–732, 2006
Barros, L. F., and R. Belfort Jr. The effects of the subconjunctival injection of bevacizumab (Avastin) on angiogenesis in the rat cornea. Anais da Academia Brasileira de Ciencias. 79:389–394, 2007
Basu, G., H. Downey, S. Guo, A. Israel, A. Asmar, B. Hargrave, and R. Heller. Prevention of distal flap necrosis in a rat random skin flap model by gene electro transfer delivering VEGF(165) plasmid. J. Gene Med. 16:55–65, 2014
Bouis, D., Y. Kusumanto, C. Meijer, N. H. Mulder, and G. A. Hospers. A review on pro- and anti-angiogenic factors as targets of clinical intervention. Pharmacol. Res. 53:89–103, 2006
Chereddy, K. K., A. Lopes, S. Koussoroplis, V. Payen, C. Moia, H. Zhu, P. Sonveaux, P. Carmeliet, A. Des Rieux, G. Vandermeulen, and V. Preat. Combined effects of PLGA and vascular endothelial growth factor promote the healing of non-diabetic and diabetic wounds. Nanomedicine. 11:1975–1984, 2015
Corral, C. J., A. Siddiqui, L. Wu, C. L. Farrell, D. Lyons, and T. A. Mustoe. Vascular endothelial growth factor is more important than basic fibroblastic growth factor during ischemic wound healing. Arch. Surg. 134:200–205, 1999
Emami, M. J., F. M. Jaberi, N. Azarpira, A. R. Vosoughi, and N. Tanideh. Prevention of arthrofibrosis by monoclonal antibody against vascular endothelial growth factor: a novel use of bevacizumab in rabbits. Orthop. Traumatol. Surg. Res. 98:759–764, 2012
Falcon, B. L., H. Hashizume, P. Koumoutsakos, J. Chou, J. V. Bready, A. Coxon, J. D. Oliner, and D. M. McDonald. Contrasting actions of selective inhibitors of angiopoietin-1 and angiopoietin-2 on the normalization of tumor blood vessels. Am. J. Pathol. 175:2159–2170, 2009
Fevata, M. Scarless Healing in the Fetus: Implications and Strategies for Postnatal Tendon Repair. Philadelphia: University of Pennsylvania, 2006
Frontera, W. R. Physiologic changes of the musculoskeletal system with aging: a brief review. Phys. Med. Rehabil. Clin. N. Am. 28:705–711, 2017
Funakoshi, T., N. Iwasaki, T. Kamishima, M. Nishida, Y. Ito, M. Kondo, and A. Minami. In vivo visualization of vascular patterns of rotator cuff tears using contrast-enhanced ultrasound. Am. J. Sports Med. 38:2464–2471, 2010
Garcia, T., W. J. Hornof, and M. F. Insana. On the ultrasonic properties of tendon. Ultrasound Med. Biol. 29:1787–1797, 2003
Gennaro, G., C. Menard, S. E. Michaud, and A. Rivard. Age-dependent impairment of reendothelialization after arterial injury: role of vascular endothelial growth factor. Circulation. 107:230–233, 2003
Ju, Y. J., H. Tohyama, E. Kondo, T. Yoshikawa, T. Muneta, K. Shinomiya, and K. Yasuda. Effects of local administration of vascular endothelial growth factor on properties of the in situ frozen-thawed anterior cruciate ligament in rabbits. Am. J. Sports Med. 34:84–91, 2006
Kaux, J. F., L. Janssen, P. Drion, B. Nusgens, V. Libertiaux, F. Pascon, A. Heyeres, A. Hoffmann, C. Lambert, C. Le Goff, V. Denoel, J. O. Defraigne, M. Rickert, J. M. Crielaard, and A. Colige. Vascular endothelial growth factor-111 (VEGF-111) and tendon healing: preliminary results in a rat model of tendon injury. Muscles Ligaments Tendons J. 4:24–28, 2014
Kostrominova, T. Y., and S. V. Brooks. Age-related changes in structure and extracellular matrix protein expression levels in rat tendons. Age (Dordr). 35:2203–2214, 2013
Kryger, Z., F. Zhang, T. Dogan, C. Cheng, W. C. Lineaweaver, and H. J. Buncke. The effects of VEGF on survival of a random flap in the rat: examination of various routes of administration. Br. J. Plast. Surg. 53:234–239, 2000
LaCroix, A. S., S. E. Duenwald-Kuehl, S. Brickson, T. L. Akins, G. Diffee, J. Aiken, R. Vanderby Jr., and R. S. Lakes. Effect of age and exercise on the viscoelastic properties of rat tail tendon. Ann. Biomed. Eng. 41:1120–1128, 2013
Langberg, H., J. Olesen, D. Skovgaard, and M. Kjaer. Age related blood flow around the Achilles tendon during exercise in humans. Eur. J. Appl. Physiol. 84:246–248, 2001
Manzano, R. P., G. A. Peyman, P. Khan, and M. Kivilcim. Testing intravitreal toxicity of bevacizumab (Avastin). Retina. 26:257–261, 2006
Marqueti, R. C., J. L. Q. Durigan, A. J. S. Oliveira, M. S. Mekaro, V. Guzzoni, A. A. Aro, E. R. Pimentel, and H. S. Selistre-de-Araujo. Effects of aging and resistance training in rat tendon remodeling. FASEB J. 32(1):353–368, 2017
Marquez-Arabia, W. H., J. Gomez-Hoyos, M. Gomez, I. Florez, J. A. Gallo, F. Monsalve, L. F. Arias, and H. D. Martin. Influence of aging on microvascular supply of the gluteus medius tendon: a cadaveric and histologic study. Arthroscopy. 33:1354–1360, 2017
McCarthy, M. M., and J. A. Hannafin. The mature athlete: aging tendon and ligament. Sports Health. 6:41–48, 2014
Needles, A., M. Arditi, N. G. Rognin, J. Mehi, T. Coulthard, C. Bilan-Tracey, E. Gaud, P. Frinking, D. Hirson, and F. S. Foster. Nonlinear contrast imaging with an array-based micro-ultrasound system. Ultrasound Med. Biol. 36:2097–2106, 2010
Needles, A., A. Heinmiller, J. Sun, C. Theodoropoulos, D. Bates, D. Hirson, M. Yin, and F. S. Foster. Development and initial application of a fully integrated photoacoustic micro-ultrasound system. IEEE Trans. Ultrason. Ferroelectr. Freq. Control. 60:888–897, 2013
Okada, Y., T. Akisue, H. Hara, K. Kishimoto, T. Kawamoto, M. Imabori, S. Kishimoto, N. Fukase, Y. Onishi, and M. Kurosaka. The effect of bevacizumab on tumour growth of malignant fibrous histiocytoma in an animal model. Anticancer Res. 30:3391–3395, 2010
Plate, J. F., P. J. Brown, J. Walters, J. A. Clark, T. L. Smith, M. T. Freehill, C. J. Tuohy, J. D. Stitzel, and S. Mannava. Advanced age diminishes tendon-to-bone healing in a rat model of rotator cuff repair. Am. J. Sports Med. 42:859–868, 2014
Plate, J. F., W. F. Wiggins, P. Haubruck, A. T. Scott, T. L. Smith, K. R. Saul, and S. Mannava. Normal aging alters in vivo passive biomechanical response of the rat gastrocnemius-Achilles muscle-tendon unit. J. Biomech. 46:450–455, 2013
Riggin, C. N., M. Chen, J. A. Gordon, S. M. Schultz, L. J. Soslowsky, and V. Khoury. Ultrasound-guided dry needling of the healthy rat supraspinatus tendon elicits early healing without causing permanent damage. J. Orthop. Res. 37:2035–2042, 2019
Riggin, C. N., A. B. Rodriguez, S. N. Weiss, H. A. Raja, M. Chen, S. M. Schultz, C. M. Sehgal, and L. J. Soslowsky. Modulation of vascular response after injury in the rat Achilles tendon alters healing capacity. J. Orthop. Res. 39:2000–2016, 2021
Riggin, C. N., J. J. Sarver, B. R. Freedman, S. J. Thomas, and L. J. Soslowsky. Analysis of collagen organization in mouse achilles tendon using high-frequency ultrasound imaging. J. Biomech. Eng. 136:021029, 2014
Riggin, C. N., S. M. Schultz, C. M. Sehgal, and L. J. Soslowsky. Ultrasound evaluation of anti-vascular endothelial growth factor-induced changes in vascular response following tendon injury. Ultrasound Med. Biol. 45:1841–1849, 2019
Rivard, A., L. Berthou-Soulie, N. Principe, M. Kearney, C. Curry, D. Branellec, G. L. Semenza, and J. M. Isner. Age-dependent defect in vascular endothelial growth factor expression is associated with reduced hypoxia-inducible factor 1 activity. J. Biol. Chem. 275:29643–29647, 2000
Rivard, A., J. E. Fabre, M. Silver, D. Chen, T. Murohara, M. Kearney, M. Magner, T. Asahara, and J. M. Isner. Age-dependent impairment of angiogenesis. Circulation. 99:111–120, 1999
Rudzki, J. R., R. S. Adler, R. F. Warren, W. R. Kadrmas, N. Verma, A. D. Pearle, S. Lyman, and S. Fealy. Contrast-enhanced ultrasound characterization of the vascularity of the rotator cuff tendon: age- and activity-related changes in the intact asymptomatic rotator cuff. J. Shoulder Elbow Surg. 17:96S-100S, 2008
Sadoun, E., and M. J. Reed. Impaired angiogenesis in aging is associated with alterations in vessel density, matrix composition, inflammatory response, and growth factor expression. J. Histochem. Cytochem. 51:1119–1130, 2003
Sehgal, C. M., P. H. Arger, A. C. Silver, J. A. Patton, H. M. Saunders, A. Bhattacharyya, and C. P. Bell. Renal blood flow changes induced with endothelin-1 and fenoldopam mesylate at quantitative Doppler US: initial results in a canine study. Radiology. 219:419–426, 2001
Takayama, K., Y. Kawakami, Y. Mifune, T. Matsumoto, Y. Tang, J. H. Cummins, N. Greco, R. Kuroda, M. Kurosaka, B. Wang, F. H. Fu, and J. Huard. The effect of blocking angiogenesis on anterior cruciate ligament healing following stem cell transplantation. Biomaterials. 60:9–19, 2015
Tempfer, H., A. Kaser-Eichberger, C. Lehner, R. Gehwolf, S. Korntner, N. Kunkel, A. Wagner, M. Gruetz, L. M. Heindl, F. Schroedl, and A. Traweger. Bevacizumab improves achilles tendon repair in a rat model. Cell Physiol. Biochem. 46:1148–1158, 2018
Torricelli, P., F. Veronesi, S. Pagani, N. Maffulli, S. Masiero, A. Frizziero, and M. Fini. In vitro tenocyte metabolism in aging and oestrogen deficiency. Age (Dordr). 35:2125–2136, 2013
Turan, A., M. A. Teber, Z. I. Yakut, H. A. Unlu, and B. Hekimoglu. Sonoelastographic assessment of the age-related changes of the Achilles tendon. Med. Ultrason. 17:58–61, 2015
Uefuji, A., T. Matsumoto, T. Matsushita, T. Ueha, S. Zhang, M. Kurosaka, and R. Kuroda. Age-related differences in anterior cruciate ligament remnant vascular-derived cells. Am. J. Sports Med. 42:1478–1486, 2014
Wang, H., J. A. Keiser, B. Olszewski, W. Rosebury, A. Robertson, I. Kovesdi, and D. Gordon. Delayed angiogenesis in aging rats and therapeutic effect of adenoviral gene transfer of VEGF. Int. J. Mol. Med. 13:581–587, 2004
World Health Organization. World Report on Ageing and Health. Geneva: World Health Organization, 2015
Yu, T. Y., J. H. Pang, K. P. Wu, M. J. Chen, C. H. Chen, and W. C. Tsai. Aging is associated with increased activities of matrix metalloproteinase-2 and -9 in tenocytes. BMC Musculoskelet. Disord. 14:2, 2013
Zhang, F., H. Liu, F. Stile, M. P. Lei, Y. Pang, T. M. Oswald, J. Beck, W. Dorsett-Martin, and W. C. Lineaweaver. Effect of vascular endothelial growth factor on rat Achilles tendon healing. Plast. Reconstr. Surg. 112:1613–1619, 2003
Acknowledgments
This study was funded by an NIH/NIAMS (Grant No. P30AR069619) supported Penn Center for Musculoskeletal Disorders Imaging Seed Grant, an NIH T32 Rheumatology Training Grant (Grant No. 4T32AR007442-29) and an NSF Graduate Research Fellowship. The aged Fischer 344 rats were provided by the NIH National Institute on Aging. There are no other conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Shayn Peirce-Cottler oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Riggin, C.N., Weiss, S.N., Rodriguez, A.B. et al. Increasing Vascular Response to Injury Improves Tendon Early Healing Outcome in Aged Rats. Ann Biomed Eng 50, 587–600 (2022). https://doi.org/10.1007/s10439-022-02948-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-022-02948-7