Summary
Background
Intestinal anastomotic failure continues to be one of the most relevant surgical incidents. Teduglutide is an enterotrophic long-acting equivalent of glucagon-like peptide-2 (GLP-2) recommended for the treatment of adult patients with short bowel syndrome. This study evaluates the effects of short-term teduglutide administration on histological parameters of intestinal anastomotic healing in an animal model.
Methods
Wistar rats (n = 31) were submitted to ileal resection and standard anastomosis, with or without postoperative teduglutide ministration, and sacrificed at the third or seventh day with tissue and blood sampling. Intestinal anastomotic healing was assessed with the modified Houdart–Hutschenreiter classification after hematoxylin and eosin staining. Collagen content and distribution were evaluated with the Gordon and Sweet technique; neoangiogenesis and epithelial proliferation indexes with anti-CD31 and anti-Ki67 immunostaining, respectively. GLP-2 plasma levels were determined by quantitative enzyme immunoassay.
Results
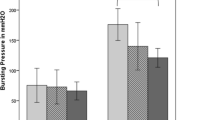
Teduglutide administration was associated with higher levels of reepithelialization (P = 0.022) and a higher neoangiogenesis index (neovessel density = 16.0 ± 10.8/mm2 vs. 5.3 ± 3.4/mm2; P = 0.0001) at the seventh postoperative day. Lower expression of type I collagen (P = 0.015) and higher content of type III collagen (P = 0.007) in the submucosa were also documented at the seventh day in teduglutide-treated animals. Drug administration was not associated with statistically significant differences of GLP-2 plasma levels.
Conclusion
These findings indicate a positive impact of teduglutide on the reepithelialization and neoangiogenesis events of the proliferative phase of intestinal anastomotic repair. Additional studies are necessary to clarify its effects on the fibrogenesis process.






Similar content being viewed by others
References
Hyman NH. Managing anastomotic leaks from intestinal anastomoses. Surgeon. 2009;7(1):31–5.
Thompson SK, Chang EY, Jobe BA. Clinical review: healing in gastrointestinal anastomoses, part I. Microsurgery. 2006;26(3):131–6.
Rijcken E, Sachs L, Fuchs T, et al. Growth factors and gastrointestinal anastomotic healing. J Surg Res. 2014;187(1):202–10.
Øines MN, Krarup P, Jorgensen LN, et al. Pharmacological interventions for improved colonic anastomotic healing: a meta-analysis. World J Gastroenterol. 2014;20(35):12637–48.
Vakalopoulos KA, Daams F, Wu Z, et al. Tissue adhesives in gastrointestinal anastomosis: a systematic review. J Surg Res. 2013;180(2):290–300.
Drucker DJ, Yusta B. Physiology and pharmacology of the enteroendocrine hormone glucagon-like peptide-2. Annu Rev Physiol. 2014;76:561–83.
Janssen P, Rotondo A, Mulé F, et al. Review article: a comparison of glucagon-like peptides 1 and 2. Aliment Pharmacol Ther. 2013;37(1):18–36.
Burness CB, McCormack PL. Teduglutide: a review of its use in the treatment of patients with short bowel syndrome. Drugs. 2013;73(9):935–47.
Van Landeghem L, Blue RE, Dehmer JJ, et al. Localized intestinal radiation and liquid diet enhance survival and permit evaluation of long-term intestinal responses to high dose radiation in mice. PLOS ONE. 2012;7(12):e51310.
Hulka JK, Omran K, Berger GS. Classification of adnexal adhesions: a proposal and evaluation of its prognostic value. Fertil Steril. 1978;30(6):661–5.
Cakmak G, Irkorucu O, Ucan B, et al. Simvastatin improves wound strength after intestinal anastomosis in the rat. J Gastrointest Surg. 2009;13(9):1707–16.
Gordon H, Sweets HH. A simple method for the silver impregnation of reticulum. Am J Pathol. 1936;12(4):545–52.
Saxena R. Special stains in interpretation of liver biopsies. Technical Articles. Connection 2010:92–103. https://pt.scribd.com/document/171923166/28829-2010-Conn14-Special-Stains-Interpretation-Liver-Biopsies-Saxena. Assessed in 6 April 2017.
Mall JW, Schwenk W, Philipp AW, et al. Intraperitoneal administration of the angiogenesis inhibitor thalidomide does not impair anastomotic healing following large bowel resection in a rabbit model. World J Surg. 2003;27(10):1119–23.
Inglin RA, Baumann G, Wagner OJ, et al. Insulin-like growth factor I improves aspects of mycophenolatemofetil-impaired anastomotic healing in an experimental model. Br J Surg. 2008;95(6):793–8.
Rekhtman N, Bishop JA. Quick reference handbook for surgical pathologists. Berlin Heidelberg: Springer; 2011.
Dubé PE, Brubaker PL. Frontiers in glucagon-like peptide-2: multiple actions, multiple mediators. Am J Physiol Endocrinol Metab. 2007;293(2):E460–5.
Gelse K, Pöschl E, Aigner T. Collagens – structure, function, and biosynthesis. Adv Drug Deliv Rev. 2003;55(12):1531–46.
Chen CZ, Raghunath M. Focus on collagen: in vitro systems to study fibrogenesis and antifibrosis – state of the art. Fibrogenesis Tissue Repair. 2009;2:7.
Hendriks T, Maastboom WJ. Healing of experimental intestinal anastomoses. Parameters of repair. Dis Colon Rectum. 1990;33(10):891–901.
Martin GR, Wallace LE, Hartmann B, et al. Nutrient-stimulated GLP-2 release and crypt cell proliferation in experimental short bowel syndrome. Am J Physiol Gastrointest Liver Physiol. 2005;288(3):G431–G438.
Garrison AP, Dekaney CM, von Allmen DC, et al. Early but not late administration of glucagon-like peptide-2 following ileo-cecal resection augments putative intestinal stem cell expansion. Am J Physiol Gastrointest Liver Physiol. 2009;296(3):G643–G650.
Koopmann MC, Liu X, Boehler CJ, et al. Colonic GLP-2 is not sufficient to promote jejunal adaptation in a PN-dependent rat model of human short bowel syndrome. JPEN J Parenter Enteral Nutr. 2009;33(6):629–39.
Muto M, Kaji T, Mukai M, et al. Ghrelin and glucagon-like peptide-2 increase immediately following massive small bowel resection. Peptides. 2013;43:160–6.
Tappenden KA. Intestinal adaptation following resection. JPEN J Parenter Enteral Nutr. 2014;38(1 Suppl):23S–31S.
Pommergaard HC. Experimental evaluation of clinical colon anastomotic leakage. Dan Med J. 2014;61(3):B4821.
Pommergaard HC, Rosenberg J, Schumacher-Petersen C, et al. Choosing the best animal species to mimic clinical colon anastomotic leakage in humans: a qualitative systematic review. Eur Surg Res. 2011;47(3):173–81.
Mantzoros I, Kanellos I, Angelopoulos S, et al. The effect of insulin-like growth factor I on healing of colonic anastomoses in cortisone-treated rats. Dis Colon Rectum. 2006;49(9):1431–8.
Zacharakis E, Demetriades H, Pramateftakis MG, et al. Effect of IGF-I on healing of colonic anastomoses in rats under 5‑FU treatment. J Surg Res. 2008;144(1):138–44.
Rijcken E, Fuchs T, Sachs L, et al. Insulin-like growth factor 1‑coated sutures improve anastomotic healing in an experimental model of colitis. Br J Surg. 2010;97(2):258–65.
Radulescu A, Zhang HY, Chen CL, et al. Heparin-binding EGF-like growth factor promotes intestinal anastomotic healing. J Surg Res. 2011;171(2):540–50.
Egger B, Inglin R, Zeeh J, et al. Insulin-like growth factor I and truncated keratinocyte growth factor accelerate healing of left-sided colonic anastomoses. Br J Surg. 2001;88(1):90–8.
Baldassano S, Amato A, Cappello F, et al. Glucagon-like peptide-2 and mouse intestinal adaptation to a high-fat diet. J Endocrinol. 2013;217(1):11–20.
El-Jamal N, Erdual E, Neunlist M, et al. Glugacon-like peptide-2: broad receptor expression, limited therapeutic effect on intestinal inflammation and novel role in liver regeneration. Am J Physiol Gastrointest Liver Physiol. 2014;307(3):G274–G285.
Martin GR, Wallace LE, Sigalet DL. Glucagon-like peptide-2 induces intestinal adaptation in parenterally fed rats with short bowel syndrome. Am J Physiol Gastrointest Liver Physiol. 2004;286(6):G964–G972.
Kaji T, Tanaka H, Holst JJ, et al. The effects of variations in dose and method of administration on glucagon like peptide-2 activity in the rat. Eur J Pharmacol. 2008;596(1-3):138–45.
Kaji T, Tanaka H, Redstone H, et al. Temporal changes in the intestinal growth promoting effects of glucagon-like peptide 2 following intestinal resection. J Surg Res. 2009;152(2):271–80.
Arda-Pirincci P, Oztay F, Bayrak BB, et al. Teduglutide, a glucagon-like peptide 2 analogue: a novel protective agent with anti-apoptotic and anti-oxidant properties in mice with lung injury. Peptides. 2012;38(2):238–47.
Alters SE, McLaughlin B, Spink B, et al. GLP2-2G-XTEN: a pharmaceutical protein with improved serum half-life and efficacy in a rat Crohn’s disease model. PLOS ONE. 2012;7(11):e50630.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
B.P. Costa, M.A. Cipriano, A.C. Gonçalves, A.M. Abrantes, P. Matafome,R. Seiça, A.B. Sarmento-Ribeiro, M. F. Botelho, and F. Castro-Sousa declare that they have no competing interests, including no financial or personal interests in any product mentioned or discussed in the article that could inappropriately influence their work.
Ethical standards
This experiment was authorized by the Ethics Committee of the Faculty of Medicine, University of Coimbra, Coimbra, Portugal (License nº32-06-2009) and conducted according to institutional and national animal welfare recommendations.
Additional information
Authors’ contributions Study conception and design: B. Costa; acquisition of data: B. Costa, M. A. Cipriano, A. C. Gonçalves, A. M. Abrantes, P. Matafome; analysis and interpretation of data: B. Costa; drafting of manuscript: B. Costa; critical review: M. A. Cipriano, R. Seiça, A. B. Sarmento-Ribeiro, M. F. Botelho, F. Castro Sousa
Original communication This manuscript represents original research that has not been previously published or submitted for publication elsewhere. The research leading to the results presented in this article was performed without external funding.
Rights and permissions
About this article
Cite this article
Costa, B.P., Cipriano, M.A., Gonçalves, A.C. et al. Effects of teduglutide on histological parameters of intestinal anastomotic healing. Eur Surg 49, 218–227 (2017). https://doi.org/10.1007/s10353-017-0478-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10353-017-0478-9