Abstract
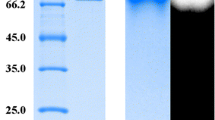
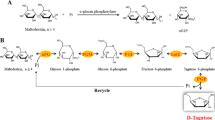
An optimal reaction condition for producing cycloamylose (CA) from sweet potato starch was investigated using a combination of isoamylase (from Pseudomonas sp.) and 4-α-glucanotransferase (from Thermus aquaticus, TAαGT). Starch was debranched by isoamylase for 8 h and subsequently reacted with TAαGT for 12 h. The yield and purity of CA products were determined using HPSEC and MALDI-TOFMS, respectively. Consequently, the maximum yield was 48.56%, exhibiting the highest CA production efficiency ever reported from starch. The CA products showed a wide range of the degree of polymerization (DP) with the minimum DP of 5. CA was also produced by simultaneous treatment of isoamylase and TAαGT. The yield was 3.31%, and the final products were contaminated by multiple branched and linear molecules. This result suggests that a former reaction condition (the sequential addition of isoamylase and TAαGT) is preferable for producing CA from sweet potato starch.
Similar content being viewed by others
References
Woolfe JA. Sweet potato: An untapped food resource. Cambridge University Press, New York, NY, USA. pp. 1–12 (1992)
George NA, Pecota KV, Bowen BD, Schultheis JR, Yencho GC. Root piece planting in sweetpotato—A synthesis of previous research and directions for the future. HortTechnology 21: 703–711 (2011)
Machida S, Ogawa S, Xiaohua S, Takaha T, Fujii K, Hayashi K. Cycloamylose as an efficient artificial chaperone for protein refolding. FEBS Lett. 486: 131–135 (2000)
Toita S, Morimoto N, Akiyoshi K. Functional cycloamylose as a polysaccharidebased biomaterial: application in a gene delivery system. Biomacromolecules 11: 397–401 (2009)
Baek HH, Kim DH, Kwon SY, Rho SJ, Kim DW, Choi HG, Kim YR, Yong CS. Development of novel ibuprofen-loaded solid dispersion with enhanced bioavailability using cycloamylose. Arch. Pharm. Res. 35: 683–689 (2012)
Baek HH, Kwon SY, Rho SJ, Lee WS, Yang HJ, Hah JM, Choi HG, Kim YR, Yong CS. Enhanced solubility and bioavailability of flurbiprofen by cycloamylose. Arch. Pharm. Res. 34: 391–397 (2011)
Kim JH, Wang R, Lee WH, Park CS, Lee S, Yoo SH. One-pot synthesis of cycloamyloses from sucrose by dual enzyme treatment: Combined reaction of amylosucrase and 4-a-glucanotransferase. J. Agr. Food Chem. 59: 5044–5051 (2011)
Lee BH, Oh DK, Yoo SH. Characterization of 4-á-glucanotransferase from Synechocystis sp. PCC 6803 and its application to various corn starches. New Biotechnol. 26: 29–36 (2009)
Takaha T, Yanase M, Takata H, Okada S, Smith SM. Potato D-enzyme catalyzes the cyclization of amylose to produce cycloamylose, a novel cyclic glucan. J. Biol. Chem. 271: 2902–2908 (1996)
Yanase M, Takata H, Takaha T, Kuriki T, Smith SM, Okada S. Cyclization reaction catalyzed by glycogen debranching enzyme (EC 2.4. 1.25/EC 3.2. 1.33) and its potential for cycloamylose production. Appl. Environ. Microb. 68: 4233–4239 (2002)
Xu Y, Zhou X, Bai Y, Wang J, Wu C, Xu X, Jin Z. Cycloamylose production from amylomaize by isoamylase and Thermus aquaticus 4-a-glucanotransferase. Carbohyd. Polym. 102: 66–73 (2014)
Gidley MJ. Molecular mechanisms underlying amylose aggregation and gelation. Macromolecules 22: 351–358 (1989)
Case S, Capitani T, Whaley J, Shi Y, Trzasko P, Jeffcoat R, Goldfarb H. Physical properties and gelation behavior of a low-amylopectin maize starch and other high-amylose maize starches. J. Cereal Sci. 27: 301–314 (1998)
Yamamoto K, Sawada S, Onogaki T. Properties of rice starch prepared by alkali method with various conditions. J. Jpn. Soc Starch Sci. 20: 99–104 (1973)
Park JH, Kim HJ, Kim YH, Cha H, Kim YW, Kim TJ, Kim YR, Park KH. The action mode of Thermus aquaticus YT-1 4-á-glucanotransferase and its chimeric enzymes introduced with starch-binding domain on amylose and amylopectin. Carbohyd. Polym. 67: 164–173 (2007)
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685 (1970)
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254 (1976)
Liebl W, Feil R, Gabelsberger J, Kellermann J, Schleifer KH. Purification and characterization of a novel thermostable 4-a-glucanotransferase of Thermotoga maritima cloned in Escherichia coli. Eur. J. Biochem. 207: 81–88 (1992)
Miller GL. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 31: 426–428 (1959)
Juliano B, Perez C, Blakeney A, Castillo T, Kongseree N, Laignelet B, Lapis E, Murty V, Paule C, Webb B. International cooperative testing on the amylose content of milled rice. Starch-Stärke 33: 157–162 (1981)
Abegunde OK, Mu TH, Chen JW, Deng FM. Physicochemical characterization of sweet potato starches popularly used in Chinese starch industry. Food Hydrocolloid. 33: 169–177 (2013)
Cai L, Shi YC, Rong L, Hsiao BS. Debranching and crystallization of waxy maize starch in relation to enzyme digestibility. Carbohyd. Polym. 81: 385–393 (2010)
Pohu A, Planchot V, Putaux J, Colonna P, Buléon A. Split crystallization during debranching of maltodextrins at high concentration by isoamylase. Biomacromolecules 5: 1792–1798 (2004)
Duan X, Chen S, Chen J, Wu J. Enhancing the cyclodextrin production by synchronous utilization of isoamylase and a-CGTase. Appl. Microbiol. Biot. 97: 3467–3474 (2013)
Fujii K, Minagawa H, Terada Y, Takaha T, Kuriki T, Shimada J, Kaneko H. Protein engineering of amylomaltase from Thermus aquaticus with random and saturation mutagenesis. Biologia 60: 97–102 (2005)
Koizumi K, Sanbe H, Kubota Y, Terada Y, Takaha T. Isolation and characterization of cyclic a-(1-4)-glucans having degrees of polymerization 9–31 and their quantitative analysis by high-performance anion-exchange chromatography with pulsed amperometric detection. J. Chromatogr. A 852: 407–416 (1999)
Taira H, Nagase H, Endo T, Ueda H. Isolation, Purification and Characterization of Large-Ring Cyclodextrins (CD36~~CD39). J. Incl. Phenom. Macro. 56: 23–28 (2006)
Sundararajan P, Rao V. Conformational studies on cycloamyloses. Carbohyd. Res. 13: 351–358 (1970)
Takeda Y, Tokunaga N, Takeda C, Hizukuri S. Physicochemical properties of sweet potato starches. Starch-Stärke 38: 345–350 (1986)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chu, S., Hong, J.S., Rho, SJ. et al. High-yield cycloamylose production from sweet potato starch using Pseudomonas isoamylase and Thermus aquaticus 4-α-glucanotransferase. Food Sci Biotechnol 25, 1413–1419 (2016). https://doi.org/10.1007/s10068-016-0220-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-016-0220-6