Abstract
Objective
SB5 showed comparable efficacy and safety profile in respect to adalimumab originator (ADA) in randomized clinical trials of rheumatoid arthritis (RA) and psoriasis. We aimed to describe the efficacy and safety of SB5 after switching from ADA in RA, axial spondyloarthritis (axSpA), psoriatic arthritis (PsA) and juvenile idiopathic arthritis (JIA) patients.
Method
Adult RA, PsA, axSpA, JIA patients treated with ADA for at least 6 months, switched to SB5 in stable clinical conditions, were eligible. Data on safety, activity indexes and patient-reported outcomes were collected at baseline, 3 and 6 months after switching.
Results
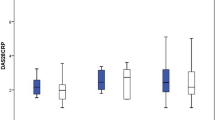
Eighty-two patients (19 RA, 28 PsA, 32 axSpA and 3 JIA; 45 females, mean age 54 ± 14 years, disease duration 13 ± 7 years, ADA duration 6 ± 3 years) were enrolled. RA patients showed stable conditions, while PsA patients showed an increase in both HAQ, DAS28(CRP) and DAPSA and axSpA patients an increase in VAS pain, VAS patient disease activity and ASDAS, both at 3 months. There were changes in the concomitant medications profile, with regression of activity indexes increases at 6 months. Adverse events were reported by 33.7% patients at 3 months and 16.6% patients at 6 months, mostly disease flares and infectious events. Two patients stopped SB5.
Conclusions
Despite temporary changes in the concomitant medication profile for mild disease flares, our real-life data replicate the safety profile of switching from ADA to SB5 in RA, with additional data for its applicability in PsA and axSpA patients, further supporting switching to biosimilars in treating inflammatory rheumatic conditions.
Key Points • Switching from adalimumab originator to SB5 is feasible in real life rheumatic inflammatory joint diseases. • Mild disease flares can present after switching from originator adalimumab to SB5, in particular in axial spondyloarthritis and psoriatic arthritis. • Changes in concomitant medications profile allows the control of minor disease flares presenting after switching from adalimumab originator to SB5. |
Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Scott DL, Wolfe F, Huizinga TW (2010) Rheumatoid arthritis. Lancet 376(9746):1094–1108. https://doi.org/10.1016/S0140-6736(10)60826-4
Ritchlin CT, Colbert RA, Gladman DD (2017) Psoriatic arthritis. N Engl J Med 376(10):957–970. https://doi.org/10.1056/NEJMra1505557
Taurog JD, Chhabra A, Colbert RA (2016) Ankylosing spondylitis and axial spondyloarthritis. N Engl J Med 374(26):2563–2574. https://doi.org/10.1056/NEJMra1406182
Prakken B, Albani S, Martini A (2011) Juvenile idiopathic arthritis. Lancet Lond Engl 377(9783):2138–2149. https://doi.org/10.1016/S0140-6736(11)60244-4
Ravelli A, Martini A (2007) Juvenile idiopathic arthritis. Lancet Lond Engl 369(9563):767–778. https://doi.org/10.1016/S0140-6736(07)60363-8
Smolen JS, Landewé R, Bijlsma J, Burmester G, Chatzidionysiou K, Dougados M, Nam J, Ramiro S, Voshaar M, van Vollenhoven R, Aletaha D, Aringer M, Boers M, Buckley CD, Buttgereit F, Bykerk V, Cardiel M, Combe B, Cutolo M, van Eijk-Hustings Y, Emery P, Finckh A, Gabay C, Gomez-Reino J, Gossec L, Gottenberg JE, Hazes JMW, Huizinga T, Jani M, Karateev D, Kouloumas M, Kvien T, Li Z, Mariette X, McInnes I, Mysler E, Nash P, Pavelka K, Poór G, Richez C, van Riel P, Rubbert-Roth A, Saag K, da Silva J, Stamm T, Takeuchi T, Westhovens R, de Wit M, van der Heijde D (2016) EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis 76(6):960–977. https://doi.org/10.1136/annrheumdis-2016-210715
Mian A, Ibrahim F, Scott DL (2019) A systematic review of guidelines for managing rheumatoid arthritis. BMC Rheumatol 3(1):42. https://doi.org/10.1186/s41927-019-0090-7
Kerschbaumer A, Sepriano A, Smolen JS, van der Heijde D, Dougados M, van Vollenhoven R, McInnes IB, Bijlsma JWJ, Burmester GR, de Wit M, Falzon L, Landewé R (2019) Efficacy of pharmacological treatment in rheumatoid arthritis: a systematic literature research informing the 2019 update of the EULAR recommendations for management of rheumatoid arthritis. Ann Rheum Dis https://doi.org/10.1136/annrheumdis-2019-216656, annrheumdis-2019-216656
Gossec L, Smolen JS, Ramiro S, de Wit M, Cutolo M, Dougados M, Emery P, Landewé R, Oliver S, Aletaha D, Betteridge N, Braun J, Burmester G, Cañete JD, Damjanov N, FitzGerald O, Haglund E, Helliwell P, Kvien TK, Lories R, Luger T, Maccarone M, Marzo-Ortega H, McGonagle D, McInnes IB, Olivieri I, Pavelka K, Schett G, Sieper J, van den Bosch F, Veale DJ, Wollenhaupt J, Zink A, van der Heijde D (2016) European league against rheumatism (EULAR) recommendations for the management of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis 75(3):499–510. https://doi.org/10.1136/annrheumdis-2015-208337
Ringold S, Weiss PF, Beukelman T, DeWitt E, Ilowite NT, Kimura Y, Laxer RM, Lovell DJ, Nigrovic PA, Robinson AB, Vehe RK, American Collge of Rheumatology (2013) 2013 update of the 2011 American College of Rheumatology Recommendations for the treatment of juvenile idiopathic arthritis. Arthritis Rheum 65(10):2499–2512. https://doi.org/10.1002/art.38092
van der Heijde D, Ramiro S, Landewé R, Baraliakos X, van den Bosch F, Sepriano A, Regel A, Ciurea A, Dagfinrud H, Dougados M, van Gaalen F, Géher P, van der Horst-Bruinsma I, Inman RD, Jongkees M, Kiltz U, Kvien TK, Machado PM, Marzo-Ortega H, Molto A, Navarro-Compàn V, Ozgocmen S, Pimentel-Santos FM, Reveille J, Rudwaleit M, Sieper J, Sampaio-Barros P, Wiek D, Braun J (2017) 2016 update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Ann Rheum Dis 76(6):978–991. https://doi.org/10.1136/annrheumdis-2016-210770
Maksymowych WP, Gladman D, Rahman P, Boonen A, Bykerk V, Choquette D, Dimond S, Fortin P, Karsh J, Klinkhoff AV, Mosher D, Mulholland K, Olszynski WP, Russell AS, Savage L, Shanner L, Shojania K, Starr M, Thomson G, Zummer M, Inman R, Canadian Rheumatology Association/ Spondyloarthritis Research Consortium of Canada (2007) The Canadian rheumatology association/ Spondyloarthritis research consortium of Canada treatment recommendations for the management of spondyloarthritis: a national multidisciplinary stakeholder project. J Rheumatol 34(11):2273–2284
Ward MM, Deodhar A, Akl EA, Lui A, Ermann J, Gensler LS, Smith JA, Borenstein D, Hiratzka J, Weiss PF, Inman RD, Majithia V, Haroon N, Maksymowych WP, Joyce J, Clark BM, Colbert RA, Figgie MP, Hallegua DS, Prete PE, Rosenbaum JT, Stebulis JA, van den Bosch F, Yu DTY, Miller AS, Reveille JD, Caplan L (2016) American College of Rheumatology/Spondylitis Association of America/Spondyloarthritis Research and Treatment Network 2015 recommendations for the treatment of Ankylosing spondylitis and nonradiographic axial spondyloarthritis. Arthritis Rheumatol Hoboken NJ 68(2):282–298. https://doi.org/10.1002/art.39298
Ruiz S (2017) Biosimilars in the EU. Biosimilar Drug Prod Dev 395–411
European Medicines Agency (2018). Imraldi. https://www.ema.europa.eu/en/medicines/human/EPAR/imraldi. Accessed 24 February 2020
Weinblatt ME, Baranauskaite A, Dokoupilova E, Zielinska A, Jaworski J, Racewicz A, Pileckyte M, Jedrychowicz-Rosiak K, Baek I, Ghil J (2018) Switching from reference adalimumab to SB5 (adalimumab biosimilar) in patients with rheumatoid arthritis: fifty-two-week phase III randomized study results. Arthritis Rheumatol Hoboken NJ 70(6):832–840. https://doi.org/10.1002/art.40444
Weinblatt ME, Baranauskaite A, Niebrzydowski J, Dokoupilova E, Zielinska A, Jaworski J, Racewicz A, Pileckyte M, Jedrychowicz-Rosiak K, Cheong SY, Ghil J, Sokolovic S, Mekic M, Prodanovic N, Gajic B, Karaselimovic-Dzambasovic E, Pojskic B, Toncheva A, Dimitar P, Rodina L, Geneva-Popova M, Staykov I, Stoilov R, Podrazilova L, Mosterova Z, Simkova G, Kopackova J, Stejfova Z, Vencovsky J, Urbanova Z, Janska L, Galatíkova D, Stropuviene S, Sniuoliene I, Sitek-Ziolkowska K, Rell-Bakalarska M, Kolasa R, Daniluk S, Sliwowska B, Bartosik-Twardowska M, Brzezicki J, Konieczny M, Jeka S, Choe J, Bae S, Kang Y, Prystupa L, Vyacheslav Z, Gasanov I, Yatsyshyn R, Rekalov D, Iaremenko O, Stanislavchuk M, Tseluyko V (2018) Phase III randomized study of SB5, an adalimumab biosimilar, versus reference adalimumab in patients with moderate-to-severe rheumatoid arthritis. Arthritis Rheumatol Hoboken NJ 70(1):40–48. https://doi.org/10.1002/art.40336
Bae SC, Lee YH (2018) Comparative efficacy and safety of biosimilar adalimumab and originator adalimumab in combination with methotrexate in patients with active rheumatoid arthritis: a Bayesian network meta-analysis of randomized controlled trials. Clin Rheumatol 37(5):1199–1205
Di Cesare A, Tronconi G, Fastame TM et al (2020) SB5 adalimumab biosimilar in the treatment of psoriasis and psoriatic arthritis. Dermatol Ther. https://doi.org/10.1111/dth.13435
Wolfe F, Michaud K, Strand V (2005) Expanding the definition of clinical differences: from minimally clinically important differences to really important differences. Analyses in 8931 patients with rheumatoid arthritis. J Rheumatol 32(4):583–589
Anonymous (2019). A Real-World study of Imraldi ® use (PROPER). https://clinicaltrials.gov/ct2/show/NCT04089514. Accessed 05 May 2020
Acknowledgements
We thank the patients who participated to the study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by CB, RB, FN, LC, LT. The first draft of the manuscript was written by CB and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
CB received honoraria from Actelion and Eli-Lilly; RB, FN, LC, LT, FB, GF: none. MMC reports receipt of grant/research support and/or speaker’s bureau attendance from Actelion, Pfizer, GlaxoSmithKline, Bristol-Myers Squibb, Bayer - MSD, Biogen, Eli Lilly.
Ethical approval
Obtained from Comitato Etico Area Vasta Toscana Centro.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Bruni, C., Bitti, R., Nacci, F. et al. Efficacy and safety of switching from reference adalimumab to SB5 in a real-life cohort of inflammatory rheumatic joint diseases. Clin Rheumatol 40, 85–91 (2021). https://doi.org/10.1007/s10067-020-05199-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05199-w