Abstract
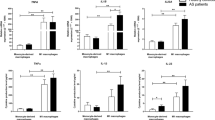
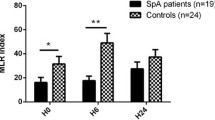
Macrophages play an important role in the ankylosing spondylitis (AS) auto-inflammatory responses and fibrocartilage destruction. Adenosine is a key modulator of inflammatory conditions. The various effects of adenosine are mediated by its interaction with adenosine receptors (AR). In this study, we investigated the mRNA expression of A1, A2A, A2B, and A3 adenosine receptors, ectonucleoside triphosphate diphosphohydrolase-1 (CD39), and ecto-5′-nucleotidase (CD73) in the monocyte-derived macrophages from AS patients in comparison to healthy controls. We also explored the correlation between analyzed gene expression and patients’ clinical manifestations. Whole blood-separated monocytes from 23 healthy controls and 23 active AS patients were stimulated by macrophage colony-stimulating factor (M-CSF) for 7 days and differentiated to macrophages. Monocyte and macrophage markers were analyzed by flow cytometry. Analysis of adenosine receptors (ADORA1، ADORA2A، ADORA2B، ADORA3), CD39 and CD73 gene expression was performed by SYBR green real-time PCR. Our results demonstrated monocyte-derived macrophages from AS patients expressed increased level of A2AAR and reduced level of A1, A2BAR, and CD39 mRNA compared to healthy controls. We found an inverse correlation between A2AAR mRNA expression and Bath Ankylosing Spondylitis Functional Index (BASFI) score in AS patients. According to our results, altered expression level of adenosine-relying system would be involved in AS macrophage dysfunction and inflammation and correlated with functional status in AS patients.





Similar content being viewed by others
References
Braun J, Sieper J (2007) Ankylosing spondylitis. Lancet 369:1379–1390. https://doi.org/10.1016/S0140-6736(07)60635-7
Tam LS, Gu J, Yu D (2010) Pathogenesis of ankylosing spondylitis. Nat Rev Rheumatol 6:399–405. https://doi.org/10.1038/nrrheum.2010.79
Rudwaleit M, Baeten D (2006) Ankylosing spondylitis and bowel disease. Best Pract Res Clin Rheumatol 20:451–471. https://doi.org/10.1016/j.berh.2006.03.010
Heuft-Dorenbosch L, Spoorenberg A, van Tubergen A, Landewe R, van ver Tempel H, Mielants H, Dougados M, van der Heijde D (2003) Assessment of enthesitis in ankylosing spondylitis. Ann Rheum Dis 62:127–132
Henderson C, Davis JC (2006) Drug insight: anti-tumor-necrosis-factor therapy for ankylosing spondylitis. Nat Clin Pract Rheumatol 2:211–218. https://doi.org/10.1038/ncprheum0157
Coates LC, Marzo-Ortega H, Bennett AN, Emery P (2010) Anti-TNF therapy in ankylosing spondylitis: insights for the clinician. Ther Adv Musculoskelet Dis 2:37–43. https://doi.org/10.1177/1759720X09359728
Fujiwara N, Kobayashi K (2005) Macrophages in inflammation. Curr Drug Targets Inflamm Allergy 4:281–286
Laria A, Lurati A, Marrazza M, Mazzocchi D, Re KA, Scarpellini M (2016) The macrophages in rheumatic diseases. J Inflamm Res 9:1–11. https://doi.org/10.2147/JIR.S82320
Melis L, Elewaut D (2009) Progress in spondylarthritis. Immunopathogenesis of spondyloarthritis: which cells drive disease? Arthritis Res Ther 11:233. https://doi.org/10.1186/ar2722
McGonagle D, Marzo-Ortega H, O'Connor P, Gibbon W, Hawkey P, Henshaw K, Emery P (2002) Histological assessment of the early enthesitis lesion in spondyloarthropathy. Ann Rheum Dis 61:534–537
Baeten D, De Keyser F (2004) The histopathology of spondyloarthropathy. Curr Mol Med 4:1–12
Bollow M, Fischer T, Reisshauer H, Backhaus M, Sieper J, Hamm B, Braun J (2000) Quantitative analyses of sacroiliac biopsies in spondyloarthropathies: T cells and macrophages predominate in early and active sacroiliitis—cellularity correlates with the degree of enhancement detected by magnetic resonance imaging. Ann Rheum Dis 59:135–140
Hasko G, Cronstein BN (2004) Adenosine: an endogenous regulator of innate immunity. Trends Immunol 25:33–39
Cronstein BN, Sitkovsky M (2017) Adenosine and adenosine receptors in the pathogenesis and treatment of rheumatic diseases. Nat Rev Rheumatol 13:41–51. https://doi.org/10.1038/nrrheum.2016.178
Hasko G, Pacher P (2012) Regulation of macrophage function by adenosine. Arterioscler Thromb Vasc Biol 32:865–869. https://doi.org/10.1161/ATVBAHA.111.226852
Buenestado A, Grassin Delyle S, Arnould I, Besnard F, Naline E, Blouquit-Laye S, Chapelier A, Bellamy JF, Devillier P (2010) The role of adenosine receptors in regulating production of tumour necrosis factor-alpha and chemokines by human lung macrophages. Br J Pharmacol 159:1304–1311. https://doi.org/10.1111/j.1476-5381.2009.00614.x
Hasko G, Deitch EA, Szabo C, Nemeth ZH, Vizi ES (2002) Adenosine: a potential mediator of immunosuppression in multiple organ failure. Curr Opin Pharmacol 2:440–444
Majumdar S, Aggarwal BB (2003) Adenosine suppresses activation of nuclear factor-kappaB selectively induced by tumor necrosis factor in different cell types. Oncogene 22:1206–1218. https://doi.org/10.1038/sj.onc.1206184
Hasko G, Linden J, Cronstein B, Pacher P (2008) Adenosine receptors: therapeutic aspects for inflammatory and immune diseases. Nat Rev Drug Discov 7:759–770. https://doi.org/10.1038/nrd2638
Hasko G, Kuhel DG, Chen JF, Schwarzschild MA, Deitch EA, Mabley JG, Marton A, Szabo C (2000) Adenosine inhibits IL-12 and TNF-[alpha] production via adenosine A2a receptor-dependent and independent mechanisms. FASEB J 14:2065–2074. https://doi.org/10.1096/fj.99-0508com
Kreckler LM, Wan TC, Ge ZD, Auchampach JA (2006) Adenosine inhibits tumor necrosis factor-alpha release from mouse peritoneal macrophages via A2A and A2B but not the A3 adenosine receptor. J Pharmacol Exp Ther 317:172–180. https://doi.org/10.1124/jpet.105.096016
Ryzhov S, Zaynagetdinov R, Goldstein AE, Novitskiy SV, Blackburn MR, Biaggioni I, Feoktistov I (2008) Effect of A2B adenosine receptor gene ablation on adenosine-dependent regulation of proinflammatory cytokines. J Pharmacol Exp Ther 324:694–700. https://doi.org/10.1124/jpet.107.131540
Le Vraux V, Chen YL, Masson I, De Sousa M, Giroud JP, Florentin I, Chauvelot-Moachon L (1993) Inhibition of human monocyte TNF production by adenosine receptor agonists. Life Sci 52:1917–1924
Sajjadi FG, Takabayashi K, Foster AC, Domingo RC, Firestein GS (1996) Inhibition of TNF-alpha expression by adenosine: role of A3 adenosine receptors. J Immunol 156:3435–3442
Blackburn MR, Vance CO, Morschl E, Wilson CN (2009) Adenosine receptors and inflammation. Handb Exp Pharmacol:215–269. https://doi.org/10.1007/978-3-540-89615-9_8
Fredholm BB (2007) Adenosine, an endogenous distress signal, modulates tissue damage and repair. Cell Death Differ 14:1315–1323. https://doi.org/10.1038/sj.cdd.4402132
Hasko G, Cronstein B (2013) Regulation of inflammation by adenosine. Front Immunol 4:85. https://doi.org/10.3389/fimmu.2013.00085
Yegutkin GG (2008) Nucleotide- and nucleoside-converting ectoenzymes: important modulators of purinergic signalling cascade. Biochim Biophys Acta 1783:673–694. https://doi.org/10.1016/j.bbamcr.2008.01.024
Jacob F, Perez Novo C, Bachert C, Van Crombruggen K (2013) Purinergic signaling in inflammatory cells: P2 receptor expression, functional effects, and modulation of inflammatory responses. Purinergic Signal 9:285–306. https://doi.org/10.1007/s11302-013-9357-4
Antonioli L, Pacher P, Vizi ES, Hasko G (2013) CD39 and CD73 in immunity and inflammation. Trends Mol Med 19:355–367. https://doi.org/10.1016/j.molmed.2013.03.005
Cekic C, Linden J (2016) Purinergic regulation of the immune system. Nat Rev Immunol 16:177–192. https://doi.org/10.1038/nri.2016.4
Ravani A, Vincenzi F, Bortoluzzi A, Padovan M, Pasquini S, Gessi S, Merighi S, Borea PA, Govoni M, Varani K (2017) Role and function of A2A and A(3) adenosine receptors in patients with ankylosing spondylitis, psoriatic arthritis and rheumatoid arthritis. Int J Mol Sci 18. https://doi.org/10.3390/ijms18040697
van der Linden S, Valkenburg HA, Cats A (1984) Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria Arthritis Rheum 27:361–368
Bortoluzzi A, Vincenzi F, Govoni M, Padovan M, Ravani A, Borea PA, Varani K (2016) A2A adenosine receptor upregulation correlates with disease activity in patients with systemic lupus erythematosus. Arthritis Res Ther 18:192. https://doi.org/10.1186/s13075-016-1089-8
Ochaion A, Bar-Yehuda S, Cohen S, Barer F, Patoka R, Amital H, Reitblat T, Reitblat A, Ophir J, Konfino I, Chowers Y, Ben-Horin S, Fishman P (2009) The anti-inflammatory target A(3) adenosine receptor is over-expressed in rheumatoid arthritis, psoriasis and Crohn's disease. Cell Immunol 258:115–122. https://doi.org/10.1016/j.cellimm.2009.03.020
Varani K, Padovan M, Vincenzi F, Targa M, Trotta F, Govoni M, Borea PA (2011) A2A and A3 adenosine receptor expression in rheumatoid arthritis: upregulation, inverse correlation with disease activity score and suppression of inflammatory cytokine and metalloproteinase release. Arthritis Res Ther 13:R197. https://doi.org/10.1186/ar3527
Mediero A, Perez-Aso M, Cronstein BN (2013) Activation of adenosine A(2A) receptor reduces osteoclast formation via PKA- and ERK1/2-mediated suppression of NFkappaB nuclear translocation. Br J Pharmacol 169:1372–1388. https://doi.org/10.1111/bph.12227
Matyash M, Zabiegalov O, Wendt S, Matyash V, Kettenmann H (2017) The adenosine generating enzymes CD39/CD73 control microglial processes ramification in the mouse brain. PLoS One 12:e0175012. https://doi.org/10.1371/journal.pone.0175012
Loza MJ, Anderson AS, O'Rourke KS, Wood J, Khan IU (2011) T-cell specific defect in expression of the NTPDase CD39 as a biomarker for lupus. Cell Immunol 271:110–117. https://doi.org/10.1016/j.cellimm.2011.06.010
Herrath J, Chemin K, Albrecht I, Catrina AI, Malmstrom V (2014) Surface expression of CD39 identifies an enriched Treg-cell subset in the rheumatic joint, which does not suppress IL-17A secretion. Eur J Immunol 44:2979–2989. https://doi.org/10.1002/eji.201344140
Guo H, Zheng M, Zhang K, Yang F, Zhang X, Han Q, Chen ZN, Zhu P (2016) Functional defects in CD4+ CD25high FoxP3+ regulatory cells in ankylosing spondylitis. Sci Rep 6:37559. https://doi.org/10.1038/srep37559
Moncrieffe H, Nistala K, Kamhieh Y, Evans J, Eddaoudi A, Eaton S, Wedderburn LR (2010) High expression of the ectonucleotidase CD39 on T cells from the inflamed site identifies two distinct populations, one regulatory and one memory T cell population. J Immunol 185:134–143. https://doi.org/10.4049/jimmunol.0803474
Acknowledgements
We would like to thank all participants for their collaboration.
Funding
This work has been supported by a research grant from Tehran University of Medical Sciences (TUMS); grant no. 95-04-41-33869.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Disclosures
None.
Electronic supplementary material
Supplementary Fig. 1
Flow cytometry analysis of CD14 expression on the cell surface of isolated monocyte. Histograms of PBMC-isolated monocyte staining with PE-conjugated anti-CD14 and isotype control antibodies. The results showed that monocytes were isolated by positive selection with 92–95% purity (GIF 21 kb)
Supplementary Fig. 2
Flow cytometry analysis of the cell surface macrophage markers CD163 and CD206. Histograms of monocyte-derived macrophage staining with a) FITC-conjugated anti-CD163 b) PE-conjugated anti-CD206, and appropriate isotype control antibodies. Results showed that Monocyte-derived macrophages with 7 days’ stimulation by M-CSF were a) 97% CD163 positive and b) 95% CD206 positive (GIF 17 kb)
Supplementary Fig. 3
There was not significant differences in a) A3AR and b) CD73 enzyme mRNA expression in monocyte-derived macrophages between AS patients and healthy controls. Isolated monocytes from 23 AS patients and 23 healthy donors were differentiated to macrophages with M-CSF. The expression was analyzed in monocyte-generated macrophages by real-time PCR and normalized to GAPDH. Data are expressed as the means ± SD (GIF 22 kb)
Rights and permissions
About this article
Cite this article
Akhtari, M., Zargar, S.J., Mahmoudi, M. et al. Ankylosing spondylitis monocyte-derived macrophages express increased level of A2A adenosine receptor and decreased level of ectonucleoside triphosphate diphosphohydrolase-1 (CD39), A1 and A2B adenosine receptors. Clin Rheumatol 37, 1589–1595 (2018). https://doi.org/10.1007/s10067-018-4055-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-018-4055-9