Abstract
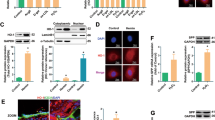
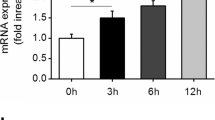
Apurinic/apyrimidinic endonuclease1/redox factor-1 (APE1/Ref-1) is a multifunctional protein involved in base excision DNA repair and transcriptional regulation of gene expression. APE1/Ref-1 is mainly localized in the nucleus, but cytoplasmic localization has also been reported. However, the functional role of cytoplasmic APE1/Ref-1 and its redox cysteine residue are still unknown. We investigated the role of cytoplasmic APE1/Ref-1 on tumor necrosis factor-α (TNF-α)-induced vascular cell adhesion molecule-1 (VCAM-1) expressions in endothelial cells. Endogenous APE1/Ref-1 was mainly observed in the nucleus, however, cytoplasmic APE1/Ref-1 was increased by TNF-α. Cytoplasmic APE1/Ref-1 expression was not blunted by cycloheximide, a protein synthesis inhibitor, suggesting cytoplasmic translocation of APE1/Ref-1. Transfection of an N-terminus deletion mutant APE1/Ref-1(29-318) inhibited TNF-α-induced VCAM-1 expression, indicating an anti-inflammatory role for APE1/Ref-1 in the cytoplasm. In contrast, redox mutant of APE1/Ref-1 (C65A/C93A) transfection led to increased TNF-α-induced VCAM-1 expression. Our findings suggest cytoplasmic APE1/Ref-1 localization and redox cysteine residues of APE1/Ref-1 are associated with its anti-inflammatory activity in endothelial cells.
Similar content being viewed by others
References
Angkeow, P., Deshpande, S.S., Qi, B., Liu, Y.X., Park, Y.C., Jeon, B.H., Ozaki, M., and Irani, K. (2002). Redox factor-1: an extranuclear role in the regulation of endothelial oxidative stress and apoptosis. Cell Death Differ. 9, 717–725.
Bae, Y.S., Oh, H., Rhee, S.G., and Yoo, Y.D. (2011). Regulation of reactive oxygen species generation in cell signaling. Mol. Cells 32, 491–509.
Bhakat, K.K., Izumi, T., Yang, S.H., Hazra, T.K., and Mitra, S. (2003). Role of acetylated human AP-endonuclease (APE1/Ref-1) in regulation of the parathyroid hormone gene. EMBO J. 22, 6299–6309.
Chen, Z., Gibson, T.B., Robinson, F., Silvestro, L., Pearson, G., Xu, B., Wright, A., Vanderbilt, C., and Cobb, M.H. (2001). MAP kinases. Chem. Rev. 101, 2449–2476.
Choi, S., Lee, Y.R., Park, M.S., Joo, H.K., Cho, E.J., Kim, H.S., Kim, C.S., Park, J.B., Irani, K., and Jeon, B.H. (2013). Histone deacetylases inhibitor trichostatin A modulates the extracellular release of APE1/Ref-1. Biochem. Biophys. Res. Commun. 435, 403–407.
Duguid, J.R., Eble, J.N., Wilson, T.M., and Kelley, M.R. (1995). Differential cellular and subcellular expression of the human multifunctional apurinic/apyrimidinic endonuclease (APE/ref-1) DNA repair enzyme. Cancer Res. 55, 6097–6102.
Evans, A.R., Limp-Foster, M., and Kelley, M.R. (2000). Going APE over ref-1. Mutat. Res. 461, 83–108.
Guo, Y., Chen, J., Zhao, T., and Fan, Z. (2008). Granzyme K degrades the redox/DNA repair enzyme Ape1 to trigger oxidative stress of target cells leading to cytotoxicity. Mol. Immunol. 45, 2225–2235.
Jackson, E.B., Theriot, C.A., Chattopadhyay, R., Mitra, S., and Izumi, T. (2005). Analysis of nuclear transport signals in the human apurinic/apyrimidinic endonuclease (APE1/Ref1). Nucleic Acids Res. 33, 3303–3312.
Jeon, B.H., and Irani, K. (2009). APE1/Ref-1: versatility in progress. Antioxid. Redox Signal. 11, 571–573.
Jeon, B.H., Gupta, G., Park, Y.C., Qi, B., Haile, A., Khanday, F.A., Liu, Y.X., Kim, J.M., Ozaki, M., White, A.R., et al. (2004). Apurinic/apyrmidinic endonuclease 1 regulates endothelial NO production and vascular tone. Circ. Res. 95, 902–910.
Joo, H.K., Oh, S.C., Cho, E.J., Park, K.S., Lee, J.Y., Lee, E.J., Lee, S.K., Kim, H.S., Park, J.B., and Jeon, B.H. (2009). Midazolam inhibits tumor necrosis factor-alpha-induced endothelial activation involvement of the peripheral benzodiazepine receptor. Anesthesiology 110, 106–112.
Kakolyris, S., Kaklamanis, L., Giatromanolaki, A., Koukourakis, M., Hickson, I.D., Barzilay, G., Turley, H., Leek, R.D., Kanavaros, P., Georgoulias, V., et al. (1998). Expression and subcellular localization of human AP endonuclease 1 (HAP1/Ref-1) protein: a basis for its role in human disease. Histopathology 33, 561–569.
Kim, C.S., Son, S.J., Kim, E.K., Kim, S.N., Yoo, D.G., Kim, H.S., Ryoo, S.W., Lee, S.D., Irani, K., and Jeon, B.H. (2006). Apurinic/apyrimidinic endonuclease1/redox factor-1 inhibits monocyte adhesion in endothelial cells. Cardiovasc. Res. 69, 520–526.
Lee, H.M., Jeon, B.H., Won, K.J., Lee, C.K., Park, T.K., Choi, W.S., Bae, Y.M., Kim, H.S., Lee, S.K., Park, S.H., et al. (2009). Gene transfer of redox factor-1 inhibits neointimal formation involvement of platelet-derived growth factor-beta receptor signaling via the inhibition of the reactive oxygen species-mediated syk pathway. Circ. Res. 104, 219–U162.
Lee, S.K., Chung, J.I., Park, M.S., Joo, H.K., Lee, E.J., Cho, E.J., Park, J.B., Ryoo, S., Irani, K., and Jeon, B.H. (2011). Apurinic/apyrimidinic endonuclease 1 inhibits protein kinase C-mediated p66shc phosphorylation and vasoconstriction. Cardiovasc. Res. 91, 502–509.
Lee, S.K., Lee, J.Y., Joo, H.K., Cho, E.J., Kim, C.S., Lee, S.D., Park, J.B., and Jeon, B.H. (2012). Tat-mediated p66shc transduction decreased phosphorylation of endothelial nitric oxide synthase in endothelial cells. Korean J. Physiol. Pharmacol. 16, 199–204.
Lee, E.J., Lee, Y.R., Joo, H.K., Cho, E.J., Choi, S., Sohn, K.C., Lee, S.D., Park, J.B., and Jeon, B.H. (2013). Arginase II inhibited lipopolysaccharide-induced cell death by regulation of iNOS and Bcl-2 family proteins in macrophages. Mol. Cells 35, 396–401.
Ozaki, M., Suzuki, S., and Irani, K. (2002). Redox factor-1/APE suppresses oxidative stress by inhibiting activity of the rac1 GTPase. FASEB J. 16, 889–890.
Qu, J., Liu, G.H., Huang, B., and Chen, C. (2007). Nitric oxide controls nuclear export of APE1/Ref-1 through S-nitrosation of Cysteines 93 and 310. Nucleic Acids Res. 35, 2522–2532.
Tell, G., Pellizzari, L., Pucillo, C., Puglisi, F., Cesselli, D., Kelley, M. R., Di Loreto, C., and Damante, G. (2000a). TSH controls Ref-1 nuclear translocation in thyroid cells. J. Mol. Endocrinol. 24, 383–390.
Tell, G., Zecca, A., Pellizzari, L., Spessotto, P., Colombatti, A., Kelley, M.R., Damante, G., and Pucillo, C. (2000b). An ‘environment to nucleus’ signaling system operates in B lymphocytes: redox status modulates BSAP/Pax-5 activation through Ref-1 nuclear translocation. Nucleic Acids Res. 28, 1099–1105.
Tell, G., Damante, G., Caldwell, D., and Kelley, M.R. (2005). The intracellular localization of APE1/Ref-1: more than a passive phenomenon? Antioxid. Redox Signal. 7, 367–384.
Tell, G., Quadrifoglio, F., Tiribelli, C., and Kelley, M.R. (2009). The many functions of APE1/Ref-1: not only a DNA repair enzyme. Antioxid. Redox Signal. 11, 601–619.
Xanthoudakis, S., Miao, G., Wang, F., Pan, Y.C.E., and Curran, T. (1992) Redox activation of Fos Jun DNA-binding activity is mediated by a DNA-repair enzyme. EMBO J. 11, 3323–3335.
Xanthoudakis, S., Miao, G.G., and Curran, T. (1994) The redox and DNA-repair activities of Ref-1 are encoded by nonover-lapping domains. Proc. Natl. Acad. Sci. USA 91, 23–27.
Yu, J.H., Kim, CS., Yoo, D.G., Song, Y.J., Joo, H.K., Kang, G., Jo, J.Y., Park, J.B., and Jeon, B.H. (2006) NADPH oxidase and mitochondrial ROS are involved in the TNF-alpha-induced vascular cell adhesion molecule-1 and monocyte adhesion in cultured endothelial cells. Korean J. Physiol. Pharmacol. 10, 217–222.
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
About this article
Cite this article
Park, M.S., Kim, CS., Joo, H.K. et al. Cytoplasmic localization and redox cysteine residue of APE1/Ref-1 are associated with its anti-inflammatory activity in cultured endothelial cells. Mol Cells 36, 439–445 (2013). https://doi.org/10.1007/s10059-013-0195-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10059-013-0195-6