Abstract
Spruce budworm (Choristoneura fumiferana Clem.) is an important and recurrent disturbance throughout spruce (Picea sp.) and balsam fir (Abies balsamea L.) dominated forests of North America. Forest carbon (C) dynamics in these ecosystems are affected during insect outbreaks because millions of square kilometers of forest suffer growth loss and mortality. We tested the hypothesis that a spruce budworm outbreak similar to those in the past could switch a forest from a C sink to a source in the near future. We used a model of ecosystem C to integrate past spruce budworm impact sequences with current forest management data on 106,000 km2 of forest in eastern Québec. Spruce budworm-caused mortality decreased stand-level merchantable C stocks by 11–90% and decreased ecosystem C stocks by 2–10% by the end of the simulation. For the first 13 years (2011–2024), adding spruce budworm significantly reduced ecosystem C stock change for the landscape from a sink (4.6 ± 2.7 g C m−2 y−1 in 2018) to a source (−16.8 ± 3.0 g C m−2 y−1 in 2018). This result was mostly due to reduced net primary production. The ecosystem stock change was reduced on average by 2 Tg C y−1 for the entire simulated area. This study provides the first estimate that spruce budworm can significantly affect the C sink or source status of a large landscape. These results indicate that reducing spruce budworm impacts on timber may also provide an opportunity to mitigate a C source.
Similar content being viewed by others
Introduction
Spruce budworm (Choristoneura fumiferana (Clem.), Lepidoptera: Tortricidae) is a native insect in spruce (Picea sp.) and balsam fir (Abies balsamea L.) forests of eastern North America. Spruce budworm outbreaks have occurred periodically in these forests for centuries (Blais 1983; Royama 1984). These outbreaks play an important role in the natural history of these forest ecosystems, and have strong influence on stand succession and landscape vegetation dynamics (Baskerville 1975; MacLean 1984). Repeated defoliation of host trees during budworm outbreak periods results in growth loss and mortality of severely defoliated trees. Mortality of dominant or co-dominant canopy trees creates openings for succession by the younger trees. Spruce budworm outbreaks have occurred across tens of millions of square kilometers in eastern North America several times during the twentieth century (Williams and Birdsey 2003), and are anticipated to do so again in the future (Candau and Fleming 2005; Gray 2008). The spruce budworm primarily attacks balsam fir, white spruce (Picea glauca Moench. Voss.), and red spruce (Picea rubens Sarg.). Budworm larvae feed predominantly on current-year foliage, so the loss in leaf area only becomes severe after several consecutive years of defoliation (MacLean and Ostaff 1989). Outbreaks generally end while there is still mature spruce and fir foliage available in the landscape (Royama 1984).
Growth loss and tree mortality during past outbreaks have resulted in significant reductions in timber supply (Sterner and Davidson 1982). It was estimated that Canada lost 44 Mm3 of timber in just 5 years (1977–1981) at the peak of the last budworm outbreak, which affected as much as 200,000 to 500,000 km2 y−1 (Kettela 1983; Simpson and Coy 1999; NFDP 2008). As a consequence, considerable investment has since been made in scientific research to develop a sound biological and ecological understanding of spruce budworm population dynamics and associated impacts on forest dynamics. Tools such as the Spruce Budworm Decision Support System (SBWDSS) were developed to synthesize and make scientific information accessible to forest managers, helping them integrate budworm considerations directly into forest management planning processes (MacLean 1996; MacLean and others 2001).
Insect disturbance agents that have eruptive outbreak dynamics and widespread impacts in terms of reduced growth and increased mortality may exert considerable influence on the forest carbon (C) and other nutrient cycles (see overview in Lovett and others 2006). One recent study investigated the impact of a defoliator (gypsy moth, Lymantria dispar L.) on net ecosystem exchange. Clark and others (2009) found stands that were net C sinks without the defoliator became net sources when there was severe defoliation of the canopy trees. Across the 1600 km2 landscape, they estimated a reduction in C uptake of 41%. Mountain pine beetle (Dendroctonus ponderosae Hopk.), more severe because it kills host trees in a single year, has been forecast to reduce forest C stocks by 270 Tg C over 20 years as a result of decreased uptake and increased decomposition from beetle-killed trees (Kurz and others 2008a). We anticipate that a spruce budworm outbreak of similar magnitude to those that have occurred in the past will also have significant impacts on the forest C budget in affected regions.
Large insect outbreaks can have considerable impact not just regionally, but also at the scale of Canada’s National Forest Carbon Budget (Kurz and Apps 1999; Kurz and others 2008b). These previous analyses took into account the impact of spruce budworm outbreaks as one of several processes ongoing simultaneously on the landscape. We tested the hypothesis that a spruce budworm outbreak similar to those in the past could switch affected forests from a C sink to a source in the near future. Our objectives were to (1) use historically derived, spatially explicit defoliation sequences and the SBWDSS to generate estimates of budworm impacts; (2) apply these in the Carbon Budget Model of the Canadian Forest Sector (CBM-CFS3) (Kurz and others 2009) to estimate C dynamics in the boreal forest of eastern Canada from 2000 to 2024; and (3) compare both stand- and landscape-level model outputs to independent data from the literature. Our study was relatively short-term and therefore did not explicitly take into account global change factors on forest growth, decomposition, or disturbances. Given the uncertainty of post-outbreak dynamics, we terminated the landscape-scale simulations in 2024, even though impacts were projected to continue somewhat beyond this time. We also did not assess the probability of a spruce budworm outbreak although the insect tends to display cyclical population dynamics (for example, Boulanger and Arsenault 2004) and budworm defoliation in Québec has increased from 3000 ha in 2003 to 338,000 ha in 2009 (QMRNF 2009).
Materials and Methods
Study Area
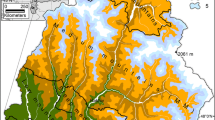
The study focused on 106,000 km2 within a 146,000 km2 region of eastern Québec, Canada (Figure 1). The forest was composed of about 60,000 km2 of spruce-fir dominated stand types, 35,000 km2 of hardwoods, mostly sugar maple (Acer saccharum Marsh.), with the balance composed of jack pine (Pinus banksiana Lamb.), or a mixture of hardwood and softwood species. White birch (Betula papyrifera Ehrh.), larch (Larix laricina Kock.), and poplar (Populus sp.) are also important species in the region. The area is characterized by rolling hills with hardwoods on the more well-drained sites. Black spruce (Picea mariana Mill. BSP) and eastern white cedar (Thuja occidentalis L.) tend to grow in areas of organic soils and poorly drained sites (Boulanger and Arsenault 2004), whereas balsam fir is often found with yellow birch (Betula alleghaniensis Britton) on the more mesic sites. The anthropological influence on forest structure through wood harvesting began in the early nineteenth century and has continued ever since (Boulanger and Arsenault 2004). The region suffered a severe spruce budworm outbreak from 1970 to 1981 (Gray and others 2000), and it is generally assumed that another outbreak in eastern Canada, including this study area, will occur (Candau and Fleming 2005; Gray 2008). The maximum stand age in the forest inventory was 175 years. The distribution was bi-modal with about 40% of the stands between 41 and 80 years of age and about 30% of stands between 140 and 160 years old.
Modeling
The CBM-CFS3 is an empirically driven, stand-, and landscape-level model for the simulation of forest C dynamics (Kull and others 2006; Kurz and others 2009). Stand-level C dynamics are represented in the CBM-CFS3 using a system of pools that allow the model to represent key ecological processes and allow users to compare estimates of stocks with field measurements. CBM-CFS3 simulates annual changes in C stocks of each pool due to growth, litter fall, turnover, decomposition, natural disturbances, and forest management (Figure 2). Where a direct comparison has been done, the basic model outputs of net primary productivity (NPP) and heterotrophic respiration (Rh) were found to be generally similar to those derived from flux tower estimates (Trofymow and others 2008).
Simple schematic of CBM-CFS3. Simulation of growth causes C to enter the forest ecosystem as living biomass. Simulation of turnover and disturbance processes causes the transfers of C from biomass to woody debris, litter, and soil pools. Natural disturbances can also cause the loss of C from the ecosystem as gaseous emissions (for example, combustion during wildfire). Harvesting causes the loss of C from the ecosystem and the transfer to the forest product sector. C is also lost from the ecosystem due to decay of the DOM and soil (Kurz and others 2009, ©Her Majesty the Queen in right of Canada, Natural Resources Canada, Canadian Forest Service as originally published in Ecological Modelling).
Forest inventory data for the region were extracted from Sylva II, the Québec forest management planning system (Lessard and others 2000). These data included a complete circa 2000 inventory of all forests in the study area and associated merchantable volume yield tables. For incorporation into the CBM-CFS3 modeling framework, the study area was stratified into 50 spatial analysis units defined by Québec’s forest management unit boundaries (Common Areas) that were in use at the time of the inventory compilation. All data loaded into the CBM-CFS3 were spatially referenced to these units. The 8919 inventory records, called stands for convenience, averaged 5.2 km2. Each one was associated with a merchantable volume yield table extracted from Sylva II. In total, the inventory was stratified into about 5600 different growth strata. Yield tables are used in CBM-CFS3 to drive tree productivity at the stand level, as a function of stand age and stand type (growth stratum), as described by Kurz and others (2009). The model was run with 8919 records assigned to 50 spatial analysis units. Model output reported on 417 aggregated records (summed) that did not contain age or growth strata information but did vary by spatial analysis unit and forest type. We used these aggregated records to calculate landscape averages and standard errors, and to conduct t-tests comparing a simulation with spruce budworm to one without spruce budworm.
Simulation initialization of the model’s dead organic matter (DOM) and soil pools was based on a historic natural disturbance regime of stand-replacing disturbances every 125 years, with fire used for simplicity (Blais 1983; Bergeron and others 2004). The model started the initialization process with all pools containing zero C stocks. The model simulated each stand through repeated iterations of growth followed by a stand-replacing fire until the sum of the above- and belowground slow C pools at the end of two successive rotations met a difference tolerance of less than 1.00%. Once the quasi-steady state was reached, the model simulated one more rotation terminated by a clear-cut. The model then grew each stand to its current age as defined in the inventory. More information about initialization is available in Kurz and others (2009).
Spruce budworm host species were defined for the purposes of this study as any forest stand greater than 4 years old and composed predominantly of spruce, balsam fir, or a spruce–fir mixture. In past outbreaks, the age and species of trees infested dictated the intensity of mortality and growth loss at the stand and landscape levels (Erdle and MacLean 1999; Bouchard and others 2007). Stand types composed mainly of fir typically have more defoliation than spruce stand types (MacLean and MacKinnon 1997; Hennigar and others 2008). In addition, mature stands have sustained substantially higher budworm-caused mortality than immature stands, on average 85% versus 35–40% (MacLean 1980). Therefore, eligible host was partitioned into four types: young spruce, old spruce, young fir, and old fir. Young host included stands between 5 and 60 years of age, whereas old host included stands greater than 60 years old. We identified 58,000 km2 of host stands in the study area, including 15,000 km2 of young spruce, 20,000 km2 of old spruce, 14,000 km2 of young fir, and 9,000 km2 of old fir. Recent cutovers and tolerant or intolerant hardwood stand types were ineligible for budworm infestation in the model simulations. During the 1970–1981 outbreak in the region, 95% of the host area experienced some level of defoliation (Gray and others 2000). We therefore simulated impacts in 95% of the host area for the future outbreak scenario.
The budworm outbreak was simulated at the landscape scale because population dynamics in large regions tend to cycle in synchrony (for example, Williams and Liebhold 2000). We simulated an outbreak start year of 2011 by applying an assumed interval of 41 years to the last outbreak start date of 1970. Boulanger and Arsenault (2004) found the mean interval of outbreaks in Québec to be 40 years, whereas others have found shorter outbreak intervals between 30 and 36 years (Candau and others 1998; Jardon and others 2003). At the time that this manuscript was written, the anticipated severe outbreak had not yet begun (39 years after the 1970 outbreak started) but, the area defoliated by budworm in Québec has increased from near zero in 2003 to 338,000 ha in 2009 (QMRNF 2009). Within the study area, the outbreak start years for individual spatial analysis units were assumed to follow dynamics of the previous 1970–1981 epidemic, because those were the only spatial data available (Gray and others 2000) (Figure 1).
Gray and MacKinnon (2006) analyzed spruce budworm defoliation from 1941 to 1998 in Canada, and described 27 patterns representing unique temporal sequences of annual percentage defoliation per year. Eighteen of these defoliation patterns occurred in Québec; spatial locations of these defoliation patterns were used in the SBWDSS to estimate impact sequences (annual percent growth loss and percent mortality) for each of the four host stand types (Figure 3). (Details on the impact sequences are provided in Supplementary material). MacLean and others (2001) described how the SBWDSS determines annual impacts expressed as percent growth loss and mortality in affected stands. Impact intensities varied, with some defoliation patterns resulting in only 7 years of growth loss, whereas others had up to 24 years of growth loss and elevated mortality rates.
Area affected by each level of percent defoliation-caused mortality per year for each host type: A old fir, B young fir, C old spruce, D young spruce. Levels of mortality were determined by the SBWDSS as a function of multi-year defoliation sequences. E Area affected by each level of percent growth loss per year.
The spruce budworm impact sequences generated by the SBWDSS were used in the CBM-CFS3 to simulate C impacts within the forest ecosystem during the outbreak period (2011–2024). Impact sequences were structured in the CBM-CFS3 as multi-year sequences of growth reduction multipliers and mortality disturbance matrices (see Kurz and others 1992 for a complete description of disturbance matrices). The amount of area affected by a particular impact sequence depended on the amount of different types of stands within the area (details in Supplementary material). The model was provided areas of each host type, impact sequence, and spatial analysis unit for each year of the simulation. If required, the model would split an inventory record into smaller areas to meet the required host type areas. Overall, area affected by mortality peaked in 2016 (Figure 3). Area affected by growth reduction increased until 2015 and remained at 55,500 km2 through the rest of the simulation. We terminated the landscape-scale simulations in 2024, even though impacts were projected to continue somewhat beyond this time, and thus did not fully estimate impacts of the entire outbreak cycle. The longer simulations are extended in the future, the more sensitive estimates become to assumptions about post-outbreak stand dynamics, including succession and competitive release in severely defoliated stands.
The model applied the estimated growth loss and mortality rates to infested stands as a function of host type and year of outbreak. Stands remained on their assigned mortality and growth loss sequence for the duration of that outbreak pattern unless interrupted by another disturbance, such as fire or harvesting. Mortality was simulated as percentage decrease in biomass pools, such as stemwood or foliage, with the C transferred to one or more of the DOM pools in each simulated year. The disturbance matrices simply move C from one pool within the CBM-CFS3 to another. In subsequent years, the added DOM decays, as defined by decay parameters associated with each pool (Kurz and others 2009). Growth reduction multipliers resulted in decreased accumulation rates of biomass within stands and did not result in additional input to the DOM pools.
During the simulated outbreak, infested stands remained eligible for other disturbances in the model. In the event of stand-replacing disturbance (that is, clear-cut harvesting), the spruce budworm outbreak was terminated in the model and the regenerating stand was not re-infested by budworm. Furthermore, as stands aged, some crossed the threshold used to distinguish between young and old budworm hosts (that is, age = 60 years); however, they remained on their original impact sequence.
In the CBM-CFS3, disturbances and forest management activities cause transfers of C between pools and removals from the ecosystem. Based on input from Québec forestry experts, clear-cut harvest events were simulated with 97% of merchantable stemwood and 50% of snag stemwood C transferred out of the ecosystem to the forest products sector. Commercial thinning activities were also simulated, with stemwood mortality due to thinning ranging from 20 to 40%. Transfers of C from the ecosystem to forest products were reported by the model as losses from the ecosystem, in accordance with current international C accounting guidelines and good practice guidance provided by the Intergovernmental Panel on Climate Change (IPCC 2006). Disturbances can affect stand age and subsequent biomass and DOM C dynamics in the disturbed stand. Following stand-replacing disturbance events, disturbed stands were regrown from age zero on the original yield curve. The simulation period of forest C dynamics was from 2000 to 2024, with the spruce budworm outbreak occurring from 2011 to 2024.
Harvest statistics for 2000–2005 and harvest projections for 2006–2024 were provided by the Government of Québec. No changes in harvest rates were simulated in response to the spruce budworm outbreak. Clear-cut harvesting was simulated, as were selection cutting, eight different intensities of commercial thinning, and silvicultural treatments such as pre-commercial thinning. Harvest projections were generated using Sylva II, which calculates sustainable harvest rates for 5-year periods. These were converted into annual harvest instructions and formatted for use in the CBM-CFS3 over the 2000–2024 simulation. Furthermore, we instructed the model to achieve the greatest possible proportion of total harvest by salvaging standing dead trees rather than cutting live trees. This was accomplished by assigning preference to stands with higher quantities of salvageable standing dead timber when selecting stands for harvest during model simulations.
Sylva II harvest projections were expressed in terms of both areas and merchantable volumes to be harvested. Area targets can be used directly in the CBM-CFS3, but volume targets must be converted from cubic m to tonnes of C. We converted the volume targets into merchantable biomass C targets, multiplying volume by three terms: (i) wood density, (ii) C density, and (iii) bark adjustment factor. We assumed an average wood density in the study area of 0.5 t m−3 for hardwood and 0.45 t m−3 for softwood species (the weighted average wood density of leading hardwood and softwood species in the study area, respectively) and a C density of 0.5 t C t−1 biomass. A bark adjustment factor of 1.19 was applied to account for the fact that volumes in Sylva II represent only the merchantable stemwood (that is, inside-bark volume), whereas the CBM-CFS3 harvests merchantable stems including both wood and bark. The bark factor was calculated by dividing outside-bark biomass (merchantable stemwood plus merchantable stem bark) by the inside-bark biomass (merchantable stemwood only), both estimated using models from Boudewyn and others (2007), for all stands in the forest inventory, and calculating an area-weighted mean ratio for the study area.
Forest fire is a naturally occurring disturbance agent in the eastern Boreal Shield ecozone. In the study area, however, fire has been far less dominant than in other boreal regions. From 1959 to 1999 the average area burned was 1.1 km2 y−1, with the maximum of 530 km2 in 1962 (Stocks and others 2002). Given the relatively small area and C impacts and to simplify the modeling, we excluded fire from the simulations of the study area.
Results
Stand-Level C Impacts
At the stand level, simulated C impacts of the spruce budworm outbreak varied from mild to very severe. The C consequences of the range of impact sequences (based on defoliation patterns from Gray and MacKinnon 2006) simulated are illustrated in Figure 4. This graph shows stand-level C dynamics in an example young balsam fir stand simulated with four contrasting intensities of budworm impact sequences, which resulted in 3, 29, 66, and 93% cumulative merchantable softwood mortality over the course of the outbreak. The spruce budworm outbreaks were simulated from year 2011 to 2024 (stand age 20–33). Merchantable softwood C stocks were reduced during the outbreak relative to the no-outbreak scenario, as tree growth was reduced and trees were killed (Figure 4A). The C in killed trees was transferred by the CBM-CFS3 to the DOM pools, for example, merchantable stemwood is transferred to the standing dead tree (snags) pools. This resulted in a large increase in standing dead wood C stocks relative to the control, particularly in the most severely affected stands (Figure 4B). Gradually, snag C stocks declined as standing dead trees were modeled to fall over and decay, transferring C into other DOM and soil pools. As expected, greater transfers of C from biomass to snags occurred in stands suffering higher mortality. Total DOM (including snags) and soil C pools were higher than the control for all but the mildest impact simulated (Figure 4C). The net impact on total ecosystem C at the stand level was a gradual loss of C relative to the no-outbreak scenario, as trees were killed and the C released gradually to the atmosphere via decomposition in the model (Figure 4D).
C stocks in a young balsam fir stand with example spruce budworm scenarios relative to a control (no-outbreak) scenario. The four spruce budworm impact sequences resulted in 3, 29, 66, and 93% cumulative mortality. A Wood and bark in the merchantable portion of stems of softwood trees, B wood and bark in standing dead softwood trees, C all DOM and soil C, and D all tree biomass, DOM and soil C. Note y-axes scales vary.
Landscape-Level C Impacts
At the landscape scale, there was no apparent trend in C stocks before the onset of the outbreak (Figure 5). The simulated impacts of spruce budworm caused biomass C stocks to start declining about 4 years after the outbreak started in 2011. At the same time, DOM and soil C stocks started increasing as the repeated defoliations caused trees to die. The simulated total ecosystem C stocks remained fairly constant until declining later during the 2011–2024 simulated outbreak, namely after 2018.
Our simulations estimated that, pre-budworm, from 2000 to 2010, forests in the study area acted as a net sink of 13.0 ± 3 g C m−2 y−1 (ecosystem stock change—ESC—also sometimes referred to as net biome production, Chapin and others 2006) (Figure 6A). As the simulated spruce budworm outbreak started in 2011, C uptake began to decline, emissions increased, and the landscape transitioned from a net C sink to a net C source by 2014. In 2018, the source was estimated at −16.8 ± 3.0 g C m−2 y−1. This was significantly different from the simulation without spruce budworm, in which the study landscape was a sink in 2018 (ESC of 4.6 + 2.7 g C m−2 y−1; two sample t-tests, P = 0.0, df = 940). The ESC from the two simulations remained significantly different in 2024 (two sample t-tests, P = 0.0, df = 940). In the absence of a spruce budworm outbreak, the study area remained a net C sink throughout the 2000–2024 simulation. From this comparison, we observed that the simulated spruce budworm outbreak was responsible for the landscape switching from a net C sink to a net C source to the atmosphere.
Simulated average landscape fluxes with (circle) and without (x) a possible spruce budworm outbreak starting in 2011, for A ESC, B NPP, C Rh, and D net growth (Net Grow). The difference in total landscape fluxes between simulations with or without spruce budworm, for E ESC F NPP, G Rh, and H net growth.
The average NPP values for the two simulations were also significantly different in 2018 and 2024 (two sample t-tests, t 2018 = −2.593, t 2024 = −2.511 P = 0.01, df = 940) (Figure 6B). Rh in the study area increased due to mortality caused by the spruce budworm (Figure 6C). Mortality increased the mass of snags, coarse woody debris, and other DOM C in the landscape. However, the landscape average Rh values with and without spruce budworm were not significantly different (two sample t-tests, t = 1.449, P = 0.15 in 2018, df = 940). Net growth (NPP minus litterfall) was reduced significantly in 2018 (two sample t-tests, t = −2.261, P = 0.02, df = 940) but not in 2024 (two sample t-tests, t = −0.153, P = 0.88, df = 940). These results indicate that the difference in ESC was largely due to the decrease in NPP. A smaller contributing factor was the shift of about 10% of the logging from living biomass to dead trees.
Spruce budworm was estimated to reduce annual change in ecosystem C stock integrated over the entire study area by up to 3.4 Tg y−1 and it continued to be reduced for the duration of the simulation period (Figure 6E). The reduction in NPP over the entire area reached a maximum of 2.5 Tg C in 2018 due to the combined impact of growth loss and mortality of hosts, and remained below the no-outbreak levels throughout the simulation period (Figure 6F). Net growth followed a similar pattern although reaching nadir earlier in 2016 at −1.2 Tg C and with a stronger recovery.
Sensitivity of ecosystem C fluxes to harvest rates was evident in the step changes in 2005 and 2010 (Figure 6A). In the simulations, projected harvest levels change every 5 years because these projections were obtained from Sylva II, which uses 5-year time steps. Projected harvest levels from Sylva’s second simulation period (2005–2009) were 10% lower than harvest levels during 2000–2004, resulting in an 18% increase in the total landscape sink of C. Removals of C from the ecosystem during harvesting averaged about 4.4 Tg C y−1 over the simulation period; these were accounted for in this study as a loss from the ecosystem (reduction in ESC), in accordance with current international accounting rules (IPCC 2006).
Validation
We have no direct way to verify the C fluxes estimated by CBM-CFS3 over 23 years and large areas of forest. However, the literature provides an opportunity for cross-model validation of NPP and net ecosystem productivity (NEP). The average NPP of trees in the CBM-CFS3 simulations from 2000 to 2010 was 434 ± 4.3 g C m−2 y−1. Jenkins and others (1999) used the TEM and PnET models to estimate regional NPP in the north-eastern US. Their modeling estimated 323 or 463 g C m−2 y−1 for the spruce–fir forests most similar to our study area. Although the three models are quite different in how they estimate growth, the NPP estimates were of similar scale. A more recent study estimated NPP for spruce–fir forests in the north-eastern US at approximately 717 ± 200 g C m−2 y−1 from FIA plots, approximately 750 g C m−2 y−1 using the PnET model and approximately 800 ± 200 g C m−2 y−1 using MODIS (Moderate Resolution Imaging Spectroradiometer) to drive a model (Pan and others 2006). Note however that spruce–fir forest comprised only 1% of their study area in a climate considerably warmer than our study area, so the higher NPP rates are not surprising. An estimate of 4 months of NEP in a black spruce site using the TRIPLEX model is approximately 180 g C m−2 (Sun and others 2008). These results were somewhat higher than our study where the CBM-CFS3 estimated the mean NEP of trees from 2000 to 2010 was 56.3 ± 51 g C m−2 y−1. The considerable variability in the standard error was due to the diversity of forest types across the landscape. The NEP is higher than the ESC reported above (13.0 ± 3 g C m−2 y−1) because it does not include disturbance emissions and harvesting losses from the ecosystem.
The FLUXNET network includes one eddy covariance flux tower located in a black spruce stand in the boreal forest of Québec. The published data from that tower reported for a single year an NEP of 37 g C m−2 y−1 observed with data gaps filled, and 77 g C m−2 y−1 modeled with CN-CLASS (Yuan and others 2008). Two other flux towers that are also located in Canadian black spruce stands estimated NEP values from 32 to 81 g C m−2 y−1(Yuan and others 2008). Our estimates of NEP values (56.3 ± 51 g C m−2 y−1) were of a similar magnitude to those estimated by flux towers in similar forests and substantially lower than temperate forest estimates from flux towers (Yuan and others 2008). Somewhat south of the study area but still in a similar spruce, fir and eastern hemlock forest is the Howland Ameriflux site (Hollinger and others 2004). That site reported a 7-year average NEP of 174 + 46 g C m−2 y−1.
To validate our modeled impacts of spruce budworm we compared results with field and aerial survey-based impact studies from outbreaks in the 1950s and 1970s. These data were not used in calibrating the SBWDSS or the CBM-CFS3. At the stand level, we selected four example impact sequences resulting in the wide range of 3, 29, 66, and 93% cumulative tree mortality over the course of the outbreak. In the 1970s outbreak in the study area, cumulative mortality rates were measured in 1979 at an average of 91% for balsam fir and 52% for white spruce stands (Blais 1981).
At the landscape scale, Blais (1964) observed impacts amounting to 7.6 m3 ha−1 y−1 killed by budworm during the period 1950–1960 in an area occupying about 25% of the study area. In a province-wide 1981 survey, Sterner and Davidson (1982) estimated that volume killed from 1977 to 1981 was 4.5 m3 ha−1 y−1. Our simulation had an overall volume killed of 2.9 m3 ha−1 y−1 from 2012 to 2024; the peak mortality rate during our simulation period was 4.9 m3 ha−1 y−1 in 2016 (Figure 7). Individual disturbance events had mortality rates as high as 32 m3 ha−1 y−1 (Figure 3). All of these per hectare values are based only on the forest area moderately to severely defoliated by spruce budworm. These comparisons suggest that our results may be conservative, indicating that the true C impacts of a future spruce budworm outbreak could exceed those estimated here.
Discussion
Our model projections indicate that a spruce budworm outbreak similar to one in the past could cause the forest, which has been a net C sink, to become a net C source for over 10 years. The magnitude of these impacts suggests that a spruce budworm outbreak represents a significant risk factor for forest C management, although some of the loss in net uptake could be mitigated by enhanced C-uptake by non-tree species in the ecosystem. If an outbreak of this magnitude does take place, then it will become difficult, if not impossible, to maintain a net C sink in the forests of the study area and the larger outbreak area not included here. Quantifying the impacts of a future outbreak would require ongoing monitoring programs of defoliation level and tree mortality.
Comparisons of spruce budworm impacts estimated in this study against those observed during previous outbreaks suggest that our impact projections may be conservative (Blais 1964, 1981; Sterner and Davidson 1982). Our estimates of volume killed per hectare were somewhat lower than estimates reported in the literature. There are two possible reasons for this: first, our estimates include the beginning and end of the outbreak, whereas the literature values focused on the outbreak peak; and second, species composition of the forest may have changed since the 1970–1981 outbreak, resulting in less vulnerable stands becoming a larger component of the forest landscape. Less vulnerable stands include younger stands and those with a smaller fir component (MacLean 1980; MacLean and MacKinnon 1997; Hennigar and others 2008). We made considerable effort in simulating the outbreak in the context of the current forest condition because the extent of the most susceptible forest types is likely lower today than it was prior to the previous outbreak (Bouchard and others 2007).
The maximum impact on simulations of ESC was less from spruce budworm (−21.4 g C m−2 y−1) compared to mountain pine beetle (−53 g C m−2 y−1) as reported by Kurz and others (2008a). The impact of the insects on NPP was part of the difference (−3.5% for spruce budworm and −10% for mountain pine beetle). However, a greater difference was due to Rh (+0.5% for spruce budworm and +6% for mountain pine beetle). These differences can be explained by the interaction between the insect and its host, in that mountain pine beetle kills the host within a year, whereas spruce budworm kills the host only after severe defoliation over multiple years. Our results were similar, although somewhat more severe than a study of gypsy moth impacts on oak–pine forests (Clark and others 2009). Our simulation showed a reduction in net ecosystem exchange of up to 48% (in 2018), whereas Clark and others reported a reduction of 41%.
There are a number of important sources of uncertainty in our simulations. One is the timing of the start of the next spruce budworm outbreak. There is evidence described in the literature of spatial synchrony across large areas (for example, Peltonen and others 2002). However, the degree of spatial synchrony may not be constant over multiple outbreaks. Moreover, periodicity of outbreaks in Québec has been suggested to have a stable oscillation of about 39 years (Royama 1984) or to be unstable, ranging from 26 to 79 years within the study area (Blais 1983). We assumed an interval of 41 years based on information provided in these references and current larval sampling results indicating that an outbreak had not started as of 2009. Finally, the actual start of the outbreak may be later than predicted. The main effect of this would be a shift in the timing of outbreak effects, not necessarily in their magnitude.
In our simulations, the ESC did not recover from the outbreak relative to the no-outbreak scenario during the simulation period. This result is somewhat uncertain because of assumptions about non-host and post-outbreak stand dynamics. We did not explicitly model competitive release following severe defoliation; the hardwoods in those stands remained on their pre-infestation yield expectations and softwoods continued to have reduced growth rates throughout the simulation. Our limited ability to model non-host and post-outbreak dynamics was the key factor in ending the simulations in 2024—the highest impact of the spruce budworm would be included but the simulation ended while growth reduction data were still available from the SBWDSS. If competitive release of non-host trees results in greater productivity, such as in aspen (Nealis and Régnière 2004), then actual future species composition will differ from the simulated condition, and growth increments provided in the yield tables would most likely be poor estimators of post-outbreak growth. However, such release is probably limited in most spruce–fir stands, based on long-term stand recovery results of Baskerville and MacLean (1979) and MacLean and Andersen (2008). Hennigar and others (2007) found in simulations that non-host volume increases in response to budworm-caused mortality averaged 14 ± 2.7 and 20 ± 4.2% for 53 stand types for moderate and severe outbreaks, versus 5 ± 1% when there was 60% foliage protection. Although a regeneration pulse likely occurs in response to substantial mortality, the regeneration is small enough for the first 10 years or so that it would have limited biomass and C in the 13-year period of our simulated outbreaks.
The simulation modeling approach used in this study only provides the capability to project ESC for a limited period in the future. The longer simulations are extended in the future, the more sensitive estimates become to assumptions about post-outbreak stand dynamics, including succession and competitive release in severely defoliated stands. We terminated landscape-scale simulations in 2024, even though impacts were projected to continue somewhat beyond this time, and thus did not fully estimate impacts over the entire outbreak cycle. Longer-term simulations would be further complicated by changes in forest management response and by impacts of global change, such as effects of elevated atmospheric CO2, increased atmospheric N deposition, and climate warming on tree productivity, insect lifecycle development, natural enemies, or host–insect interactions. All of these factors were beyond the scope of this study and represent sources of uncertainty.
An abundance of literature highlights the effect of temperature and/or precipitation on factors that influence spruce budworm outbreak dynamics: developmental rates (Régnière and You 1991; Weber and others 1999), dispersal (Greenbank and others 1980), feeding (Régnière and You 1991), fecundity (Sanders and others 1978; Harvey 1983), and survival (Régnière and Duval 1998); microsporidian parasite development (Wilson 1974); parasitoid developmental rates (Lysyk and Nealis 1988; Nealis and Fraser 1988; Thireau and Régnière 1995), flight activity (and presumably search rates) (Elliott and others 1986; Nyrop and Simmons 1986), longevity (Nealis and Fraser 1988), and oviposition rate (Nealis 1988); host abundance phenology and growth (Lekas and others 1990; Deslauriers and others 2003); and epizootiology of forest insect pathogens (Smitley and others 1995). Unfortunately, it is not known how these factors will interact in an altered climatic environment. The analysis of Candau and Fleming (2005) suggests that defoliation frequency in Ontario will increase with rising winter minimum and maximum temperatures, lower spring minimum, May maximum and August minimum temperatures, and lower June precipitation. They did not estimate the net effect under any climate change scenario. Gray (2008) predicted an average increase in outbreak duration of approximately 6 years, and an average increase in approximately 15% in defoliation levels in eastern Canada under the IPCC SRES-B1 climate scenario. Neither Candau and Fleming (2005) nor Gray (2008) estimated how climate change would affect forest composition, a significant factor in outbreak dynamics.
The future forest composition and C balance will depend on growth, mortality, decomposition, regeneration, competition (succession), and migration. Each of these processes is affected by change in climate, as well as other factors including disturbances, ozone concentrations, acid and nitrogen deposition, and CO2 concentration effects on growth rates. Recent reviews by Mohan and others (2009) and Campbell and others’ (2009) report on the known complexity and remaining data gaps for northeastern North America. Campbell and others’ (2009) projections show a range of potential outcomes including both increasing and decreasing NPP. In some parts of the boreal forest, even increasing NPP may be overwhelmed by increases in burned area, resulting in a net C source (Kurz and others 2007). However, fire plays a smaller role in our study area and is expected to decrease (Bergeron and others 2004).
Management response to budworm outbreak also represents a source of uncertainty for this study. Several management options are available to mitigate the impacts of spruce budworm outbreak on forest C stocks and timber supply (MacLean 1996; MacLean and others 2001; Hennigar and MacLean 2010). During the outbreak, insecticides can and have been used to reduce foliage loss in infested stands. Our simulations were conducted with the assumption that no insecticide-treatment efforts would be applied: they represent a baseline without any spruce budworm suppression. During and after an outbreak for a limited time, harvesting can be redirected toward salvage of spruce and fir killed by the budworm as was assumed in our study. Long-term silvicultural strategies can also reduce landscape susceptibility to large-scale outbreaks by managing for a broader mix of forest types and wider range of stand ages on the landscape, much of which has already taken place in New Brunswick (MacLean 1996).
In this study, C removed from the forest during harvesting is accounted for as a direct emission to the atmosphere. Although the international accounting rules treat removals of C from forests as direct losses to the atmosphere (IPCC 2006), some of the harvested C accumulates in harvested wood products and landfills (Apps and others 1999). Therefore, although harvest removals reduce ecosystem C stocks, they do not necessarily result in increased greenhouse gas emissions to the atmosphere. Moreover, in addition to the C storage, the impacts of substitution effects (that is, reduced fossil fuel emissions from the use of wood products instead of more energy-intensive products) should be considered when comparing alternative management scenarios (Stinson and Freedman 2001; Neilson and others 2008).
In regions suffering high mortality from insect outbreaks, a common policy response is to increase harvest rates to provide the forest industry with more latitude to recover standing dead timber. The provincial governments may choose to raise harvest levels in the event of a major budworm outbreak. This strategy would not provide any C benefits under the current international accounting rules, but could provide benefits to the atmosphere if forest products are used to substitute for more GHG-intensive products. For example, wood products in the construction industry have been shown to reduce CO2 emissions by 93–1062 kg CO2 equivalent per m3 when replacing concrete and 36–442 kg CO2 equivalent per m3 when substituted for steel (Petersen and Solberg 2005).
Conclusion
In this study, we estimated that a possible future spruce budworm outbreak could cause the managed forests in the 106,000 km2 study area in Québec to transition from a net C sink to net C source during the outbreak. Impacts of a single spruce budworm scenario in this region were projected to be a reduction in NPP of about 25 Tg C over 13 years and an increase in Rh of 6.5 Tg C over the same period. Impacts of this magnitude are substantial, even in the context of the national forest C budget of Canada. Expressed in units of carbon dioxide, the average annual impact in the study area was estimated to be 8.5 Tg CO2 y−1. Given the magnitude of the projected impacts of the spruce budworm outbreak and the geographic extent of the next outbreak, which will undoubtedly be much larger than our study area, efforts to suppress spruce budworm that achieve even relatively minor reductions in impact could make a substantial contribution to climate mitigation efforts. Slaney and others (2009) demonstrated low costs per tonne of CO2 mitigated. Furthermore, emissions created by the suppression efforts were deemed negligible compared to the magnitude of the net CO2 protected. Clearly managers should also consider other ecological impacts of budworm suppression; however, using the naturally occurring biological insecticide Bacillus thuringiensis (B.t.), ecological impacts are low because the insecticide has a limited active period and a narrow range of insects affected. Where mortality does occur, a shift of sustainable logging to focus on stands with a large proportion of dead trees will ensure that instead of decomposing, the wood could be used to meet society’s demands. Any salvage logging must still be done within the context of the wide variety of sustainable forest management objectives including biodiversity, habitat, water quality, and reforestation.
References
Apps MJ, Kurz WA, Beukema SJ, Bhatti JS. 1999. Carbon budget of the Canadian forest product sector. Environ Sci Policy 2:25–41.
Baskerville GL. 1975. Spruce budworm: super silviculturist. For Chron 51:138–40.
Baskerville GL, MacLean DA. 1979. Budworm-caused mortality and 20-year recovery in immature balsam fir stands. Canadian Forest Service Information Report M-X-102.
Bergeron Y, Flannigan M, Gauthier S, Leduc A, Lefort P. 2004. Past, current and future fire frequency in the Canadian boreal forest: implications for sustainable forest management. AMBIO 33:356–60.
Blais JR. 1964. Account of a recent spruce budworm outbreak in the Laurentide Park region of Quebec and measures for reducing damage in future outbreaks. For Chron 40:313–23.
Blais JR. 1981. Mortality of balsam fir and white spruce following a spruce budworm outbreak in the Ottawa River watershed in Quebec. Can J For Res 11:620–9.
Blais JR. 1983. Trends in the frequency, extent, and severity of spruce budworm outbreaks in eastern Canada. Can J For Res 13:539–47.
Bouchard M, Kneeshaw D, Messier C. 2007. Forest dynamics following spruce budworm outbreaks in the northern and southern mixedwoods of central Quebec. Can J For Res 37:763–72.
Boudewyn P, Song X, Magnussen S, Gillis MD. 2007. Model-based, volume-to-biomass conversion for forested and vegetated land in Canada. Canadian Forest Service, Victoria, Canada, Information Report BC-X-411.
Boulanger Y, Arsenault D. 2004. Spruce budworm outbreaks in eastern Quebec over the last 450 years. Can J For Res 34:1035–43.
Campbell JL, Rustad LE, Boyer EW, Christopher SF, Driscoll CT, Fernandez IJ, Groffman PM, Houle D, Kiekbusch J, Magill AH, Mitchell MJ, Ollinger SV. 2009. Consequences of climate change for biogeochemical cycling in forests of northeastern North America. Can J For Res 39:264–84.
Candau J-N, Fleming RA. 2005. Landscape-scale spatial distribution of spruce budworm defoliation in relation to bioclimatic conditions. Can J For Res 35:2218–32.
Candau J-N, Fleming RA, Hopkin A. 1998. Spatiotemporal patterns of large-scale defoliation caused by the spruce budworm in Ontario since 1941. Can J For Res 28:1733–41.
Chapin FS, Woodwell GM, Randerson JT, Rastetter EB, Lovett GM, Baldocchi DD, Clark DA, Harmon ME, Schimel DS, Valentini R, Wirth C, Aber JD, Cole JJ, Goulden ML, Harden JW, Heimann M, Howarth RW, Matson PA, McGuire AD, Melillo JM, Mooney HA, Neff JC, Houghton RA, Pace ML, Ryan MG, Running SW, Sala OE, Schlesinger WH, Schulze E-D. 2006. Reconciling carbon-cycle concepts, terminology, and methods. Ecosystems 9:1041–50.
Clark KL, Skowronski N, Hom J. 2009 Invasive insects impact forest carbon dynamics. Glob Change Biol. doi:10.1111/j.1365-2486.2009.01983.x.
Deslauriers A, Morin H, Urbinati C, Carrer M. 2003. Daily weather responses of balsam fir (Abies balsamea (L.) Mill.) stem radius increment from dendrometer analysis in the boreal forests of Québec (Canada). Trees 17:477–84.
Elliott NC, Simmons GA, Draper RJ. 1986. Adult emergence and activity patterns of parasites of early instar jack pine budworm (Lepidoptera: Tortricidae). Environ Entomol 15:409–16.
Erdle TA, MacLean DA. 1999. Stand growth model calibration for use in forest pest impact assessment. For Chron 75:141–52.
Gray DR. 2008. The relationship between climate and outbreak characteristics of the spruce budworm in eastern Canada. Clim Change 87:361–83.
Gray DR, Régnière J, Boulet B. 2000. Analysis and use of historical patterns of spruce budworm defoliation to forecast outbreak patterns in Quebec. For Ecol Manage 127:217–31.
Gray DR, MacKinnon WE. 2006. Outbreak patterns of the spruce budworm and their impacts in Canada. For Chron 82:550–61.
Greenbank DO, Schaefer GW, Rainey RC. 1980. Spruce budworm (Lepidoptera: Tortricidae) moth flight and dispersal: new understanding from canopy observations, radar, and aircraft. Mem Entomol Soc Can 110:1–49.
Harvey GT. 1983. Environmental and genetic effects on mean egg weight in spruce budworm (Lepidoptera: Tortricidae). Can Entomol 115:1109–17.
Hennigar CR, MacLean DA. 2010. Spruce budworm and management effects on forest and wood product carbon for an intensively managed forest. Can J For Res (in press).
Hennigar CR, MacLean DA, Porter KB, Quiring DT. 2007. Optimized harvest planning under alternative foliage-protection scenarios to reduce volume losses to spruce budworm. Can J For Res 37:1755–69.
Hennigar CR, MacLean DA, Quiring DT, Kershaw JA Jr. 2008. Differences in spruce budworm defoliation among balsam fir and white, red, and black spruce. For Sci 54:150–66.
Hollinger DY, Aber J, Dail B, Davidson EA, Goltz SM, Hughes H, Leclerc MY, Lee JT, Richardson AD, Rodrigues C, Scott NA, Achuatavarier D, Walsh J. 2004. Spatial and temporal variability in forest-atmosphere CO2 exchange. Glob Change Biol 10(10):1689–706.
IPCC. 2006. In: Eggleston HS, Buendia L, Miwa K, Ngara T, and Tanabe K, Eds. 2006 IPCC guidelines for national greenhouse gas inventories. Japan: Institute for Global Environmental Strategies. www.ipcc-nggip.iges.or.jp/public/2006gl/vol4.html.
Jardon Y, Morin H, Dutilleul P. 2003. Périodicité et synchronisme des épidémies de la tordeuse des bourgeons de l’épinette au Québec. Can J For Res 25:902–11.
Jenkins JC, Kicklighter DW, Ollinger SV, Aber JD, Melillo JM. 1999. Sources of variability in net primary production predictions at a regional scale: a comparison using PnET-II and TEM 40 in northeastern US forests. Ecosystems 2:555–70.
Kettela EG. 1983. A cartographic history of spruce budworm defoliation 1967 to 1981 in eastern North America. Canadian Forest Service Information Report DPC-X-14.
Kull SJ, Kurz WA, Rampley GJ, Banfield GE, Schivatcheva RK, Apps MJ. 2006. Operational-Scale Carbon Budget Model off the Canadian Forest Sector (CBM-CFS3), Version 1.0: user’s guide. Edmonton: Natural Resources Canada, Canadian Forest Service.
Kurz WA, Apps MJ, Webb TM, McNamee PJ. 1992. Carbon Budget of the Canadian Forest Sector Phase I. Forestry Canada, Information Report NOR-X-326.
Kurz WA, Apps MJ. 1999. A 70-year retrospective analysis of carbon fluxes in the Canadian forest sector. Ecol Appl 9:526–47.
Kurz WA, Stinson G, Rampley G. 2007. Could increased boreal forest ecosystem productivity offset carbon losses from increased disturbances? Philos Trans R Soc B 363:1–9.
Kurz WA, Dymond CC, Stinson G, Rampley GJ, Neilson ET, Carroll AL, Ebata T, Safranyik L. 2008a. Mountain pine beetle and forest carbon feedback to climate change. Nature 452:987–90.
Kurz WA, Stinson G, Rampley GJ, Dymond CC, Neilson ET. 2008b. Risk of natural disturbances makes future contribution of Canada’s forests to the global carbon cycle highly uncertain. Proc Natl Acad Sci USA 105:1551–5.
Kurz WA, Dymond CC, White TM, Stinson G, Shaw CH, Rampley GJ, Smyth C, Simpson BN, Neilson ET, Trofymow JA, Metsaranta J, Apps MJ. 2009. CBM-CFS3: a model of C-dynamics in forestry and land-use change implementing IPCC standards. Ecol Model 220:480–504. doi:10.1016/j.ecolmodel.2008.10.018.
Lekas TM, MacDougal RG, MacLean DA, Thompson RG. 1990. Seasonal trends and effects of temperature and rainfall on stem electrical capacitance of spruce and fir trees. Can J For Res 20:970–7.
Lessard G, Pelletier F, Champagne M. 2000. Bilan de l’implantation de Sylva II au Québec. Centre d’enseignement et de recherche de Sainte-Foy inc. (CERFO). Rapport 2000-2008. 12 pp. 6 annexes. www.cerfo.qc.ca/gabarit.asp?nompage=down/sylva.htm.
Lovett GM, Canham CD, Authur MA, Weathers KC, Fitzhugh RD. 2006. Forest ecosystem responses to exotic pests and pathogens in Eastern North America. Bioscience 56:395–405.
Lysyk TJ, Nealis VG. 1988. Temperature requirements for development for the jack pine budworm (Lepidoptera: Tortricidae) and two of its parasitoids (Hymenoptera). J Econ Entomol 81:1045–51.
MacLean DA. 1980. Vulnerability of fir-spruce stands during uncontrolled spruce budworm outbreaks: a review and discussion. For Chron 56:213–20.
MacLean DA. 1984. Effects of spruce budworm outbreaks on the productivity and stability of balsam fir forests. For Chron 60:273–9.
MacLean DA. 1996. Forest management strategies to reduce spruce budworm damage in the Fundy Model Forest. For Chron 72:399–405.
MacLean DA, Ostaff DP. 1989. Patterns of balsam fir mortality caused by uncontrolled spruce budworm outbreak. Can J For Res 19:1087–95.
MacLean DA, MacKinnon WE. 1997. Effects of stand and site characteristics on susceptibility and vulnerability of balsam fir and spruce to spruce budworm in New Brunswick. Can J For Res 27:1859–71.
MacLean DA, Andersen AR. 2008. Impact of a spruce budworm outbreak in balsam fir and subsequent stand development over a 40-year period. For Chron 84:60–9.
MacLean DA, Erdle TA, MacKinnon WE, Porter KB, Beaton KP, Cormier G, Morehouse S, Budd M. 2001. The spruce budworm decision support system: Forest protection planning to sustain long-term wood supply. Can J For Res 31:1742–57.
Mohan JE, Cox RM, Iverson LR. 2009. Composition and carbon dynamics of forests in northeastern North America in a future, warmer world. Can J For Res 39:213–30.
Nealis VG. 1988. Weather and the ecology of Apanteles fumiferanae Vier. (Hymenoptera: Braconidae). Mem Entomol Soc Can 146:57–70.
Nealis VG, Fraser S. 1988. Rate of development, reproduction, and mass-rearing of Apanteles fumiferanae Vier. (Hymenoptera: Braconidae) under controlled conditions. Can Entomol 120:197–204.
Nealis VG, Régnière J. 2004. Insect-host relationships influencing disturbance by the spruce budworm in a boreal mixedwood forest. Can J For Res 34:1870–82.
Neilson ET, MacLean DA, Meng F-R, Hennigar CR, Arp PR. 2008. Optimal on- and off-site forest carbon sequestration under existing timber supply constraints in northern New Brunswick. Can J For Res 38:2784–96.
NFDP (National Forestry Database Program). 2008. Compendium of Canadian forestry statistics [online]. Canadian Council of Forest Ministers. www.nfdp.ccfm.org. Accessed February 2008.
Nyrop JP, Simmons GA. 1986. Temporal and spatial activity patterns of an adult parasitoid, Glypta fumiferanae (Hymenoptera: Ichneumonidae), and their influence on parasitism. Environ Entomol 15:481–7.
Pan Y, Birdsey R, Hom J, McCullough K, Clark K. 2006. Improved estimates of net primary productivity from MODIS satellite data at regional and local scales. Ecol Appl 16(1):125–32. doi:10.1890/05-0247.
Peltonen M, Liebhold AM, Bjørnstad ON, Williams DW. 2002. Spatial synchrony in forest insect outbreaks: roles of regional stochasticity and dispersal. Ecology 83:3120–9.
Petersen AK, Solberg B. 2005. Environmental and economic impacts of substitution between wood products and alternative materials: a review of micro-level analyses from Norway and Sweden. For Policy Econ 7:249–59.
QMRNF (Quebec Ministère des Ressources naturelles et de la Faune du Québec). 2009. Aires défoliées par la tordeuse des bourgeons de l’épinette au Québec en 2009. Direction de l’environnement et de la protection des forêts, Québec, Québec.
Régnière J, Duval P. 1998. Overwintering mortality of spruce budworm, Choristoneura fumiferana (Clem.) (Lepidoptera: Tortricidae), populations under field conditions. Can Entomol 130:13–26.
Régnière J, You M. 1991. A simulation model of spruce budworm (Lepidoptera: Tortricidae) feeding on balsam fir and white spruce. Ecol Model 54:277–97.
Royama T. 1984. Population dynamics of the spruce budworm Choristoneura fumiferana. Ecol Monogr 54:429–62.
Sanders CJ, Wallace DR, Lucuik GS. 1978. Flight activity of female eastern spruce budworm (Lepidoptera: Tortricidae) at constant temperatures in the laboratory. Can Entomol 110:627–32.
Simpson R, Coy D. 1999. An ecological atlas of forest insect defoliation in Canada 1980–1996. Canadian Forest Service Information Report M-X-206E.
Slaney GL, Lantz VA, MacLean DA. 2009. The economics of carbon sequestration through pest management: application to forested landbases in New Brunswick and Saskatchewan, Canada. For Policy Econ 11:525–34.
Smitley DR, Bauer LS, Hajek AE, Sapio FJ, Humber RA. 1995. Introduction and establishment of Entomophaga maimaiga, a fungal pathogen of gypsy moth (Lepidoptera: Lymantriidae) in Michigan. Environ Entomol 24:1685–95.
Sterner TE, Davidson AG, Eds. 1982. Forest insect and disease conditions in Canada 1981. Canadian Forest Service FIDS Report. 46 p.
Stinson G, Freedman B. 2001. Potential for carbon sequestration in Canadian forests and agroecosystems. Mitig Adapt Strateg Glob Change 6:1–23.
Stocks BJ, Mason JA, Todd JB, Bosch EM, Wotton BM, Amiro BD, Flannigan MD, Hirsch KG, Logan KA, Martell DL, Skinner WR. 2002. Large forest fires in Canada, 1959–1997. J Geophys Res 108(D1 8149):FFR5.1–FFR5.12.
Sun J, Peng C, McCaughey H, Zhou X, Thomas V, Berninger F, St. Onge B, Hua D. 2008. Simulating carbon exchange of Canadian boreal forests: II. Comparing the carbon budgets of a boreal mixedwood stand to a black spruce forest stand. Ecol Model 219(3–4):276–86.
Thireau J-C, Régnière J. 1995. Development, reproduction, voltinism and host synchrony of Meteorus trachynotus with its host Choristoneura fumiferana and C. rosaceana. Entomologia Experimentalis et Applicata 76:67–82.
Trofymow JA, Stinson G, Kurz WA. 2008. Derivation of a spatially explicit 86-year retrospective carbon budget for a landscape undergoing conversion from old-growth to managed forests on Vancouver Island, BC. For Ecol Manage 256(10):1677–91.
Weber JD, Volney WJA, Spence JR. 1999. Intrinsic developmental rate of spruce budworm (Lepidoptera: Tortricidae) across a gradient of latitude. Environ Entomol 28:224–32.
Williams D, Birdsey R. 2003. Historical patterns of spruce budworm defoliation and bark beetle outbreaks in North American conifer forests: an atlas and description of digital maps. U.S.D.A Forest Service General Technical Report NE-308.
Williams DW, Liebhold AM. 2000. Spatial synchrony of spruce budworm outbreaks in eastern North America. Ecology 81:2753–66.
Wilson GG. 1974. The effects of temperature and ultraviolet radiation on the infection of Choristoneura fumiferana and Malacosoma pluviale by a microsporidian parasite, Nosema (Perezia) fumiferanae (Thom.). Can J Zool 52:59–63.
Yuan F, Arain MA, Barr A, Black TA, Bourque P-A, Coursolle C, Margolis H, McCaughey H, Wofsy SC. 2008. Modeling analysis of primary controls on net ecosystem productivity of seven boreal and temperate coniferous forests across an east-west continental transect in Canada. Glob Change Biol 14:1–20. doi:10.1111/j.1365-2486.2008.01612.x.
Acknowledgements
We would like to acknowledge the contributions of Greg Rampley for CBM-CFS3 model code development, Mark Budd for SBWDSS model parameterization, Kathy Beaton for SBWDSS model parameterization and expertise, and Juha Metsaranta and Kevin Belanger for assistance structuring SBWDSS output for input into the CBM-CFS3. We would also like to thank three anonymous reviewers and editor Dr. Gary Lovett for their helpful suggestions and revisions on a previous version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author Contributions
CD led writing of the article and performed research. EN performed research and contributed to the article. GS performed research, contributed to the study design study, and contributed to the article. KP, DM, and DG contributed to the study design, models, and contributed to the article. MC analyzed data, and WK conceived of the study and contributed to the article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dymond, C.C., Neilson, E.T., Stinson, G. et al. Future Spruce Budworm Outbreak May Create a Carbon Source in Eastern Canadian Forests. Ecosystems 13, 917–931 (2010). https://doi.org/10.1007/s10021-010-9364-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-010-9364-z