Abstract
Objectives
To present and explore the potential of an animal-based experimental model developed to determine the set of root canal sealers in vivo. The setting of AH Plus, BioC Sealer, TotalFill BC Sealer, and Sealapex was determined using either ISO 6876 or the novel in vivo method proposed in this study.
Material and methods
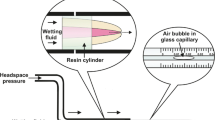
The in vitro setting time of the sealers tested was determined in accordance with ISO 6876:2012. In determining the in vivo set, 24 adult Wistar rats were followed up for two evaluation periods: 1 and 4 weeks. Their upper-right incisor was extracted, and its pulp tissue was removed. The root canal was then filled from retrograde with one of the 4 sealers, and the tooth was re-implanted and fixed with a layer of a flowable composite resin. After 1 or 4 weeks of the surgical procedures, the animals were euthanized, and their incisors were extracted. Two-mm-thick slices of the middle third of the tooth root were obtained and assessed with a Gillmore device, to determine whether or not the sealer had set.
Results
The following in vitro results were obtained by using ISO 6876 methodology: AH Plus set after a mean time of 423 ± 20 min and 476 ± 35 min, in metal and plaster molds, respectively. BioC Sealer set after 7 days (in dental plaster molds), whereas TotalFill BC Sealer and Sealapex did not set even after 25 days in both tested conditions (metal or dental plaster molds). Using the novel in vivo methodology, AH Plus, BioC Sealer, and TotalFill BC Sealer set after both 7 and 30 days. In contrast, Sealapex did not set at either time point.
Conclusions
AH Plus and BioC Sealer set under both in vitro and in vivo test conditions. TotalFill BC Sealer did not set under in vitro conditions but did after 1 week under in vivo conditions. Sealapex did not set under either in vitro or in vivo conditions.
Clinical relevance
The influence of the testing conditions on the setting results is a clear indication that new in vivo experimental models should be useful in future studies on Bioceramics root canal sealers.


Similar content being viewed by others
References
Damas BA, Wheater MA, Bringas JS, Hoen MM (2011) Cytotoxicity comparison of mineral trioxide aggregates and EndoSequence bioceramic root repair materials. J Endod 37:372–375
Leal F, De-Deus G, Brandão C, Luna AS, Fidel SR, Souza EM (2011) Comparison of the root-end seal provided by bioceramic repair cements and White MTA. Int Endod J 44:662–668
Parirokh M, Torabinejad M, Dummer PMH (2018) Mineral trioxide aggregate and other bioactive endodontic cements: an updated overview - part I: vital pulp therapy. Int Endod J 2:177–205
Torabinejad M, Parirokh M, Dummer PMH (2018) Mineral trioxide aggregate and other bioactive endodontic cements: an updated overview - part II: other clinical applications and complications. Int Endod J 3:284–317
Chang SW, Lee SY, Kang SK, Kum KY, Kim EC (2014) In vitro biocompatibility, inflammatory response, and osteogenic potential of 4 root canal sealers: Sealapex, Sankin Apatite Root Sealer, MTA Fillapex, and iRoot SP root canal sealer. J Endod 40:1642–1648
Shokouhinejad N, Nekoofar MH, Razmi H, Sajadi S, Davies TE, Saghiri MA, Gorjestani H, Dummer PM (2012) Bioactivity of EndoSequence root repair material and bioaggregate. Int Endod J 45:1127–1134
Chen I, Salhab I, Setzer FC, Kim S, Nah HD (2016) A new calcium silicate-based bioceramic material promotes human osteo- and odontogenic stem cell proliferation and survival via the extracellular signal-regulated kinase signaling pathway. J Endod 3:480–486
Moinzadeh AT, Aznar Portoles C, Schembri Wismayer P, Camilleri J (2016) Bioactivity potential of EndoSequence BC RRM Putty. J Endod 4:615–621
Lv F, Zhu L, Zhang J, Yu J, Cheng X, Peng B (2017) Evaluation of the in vitro biocompatibility of a new fast-setting ready-to-use root filling and repair material. Int Endod J 6:540–548
Giacomino CM, Wealleans JA, Kuhn N, Diogenes A (2019) Comparative biocompatibility and osteogenic potential of two bioceramic sealers. J Endod 45:51–56
Candeiro GT, Correia FC, Duarte MA, Ribeiro-Siqueira DC, Gavini G (2012) Evaluation of radiopacity, pH, release of calcium ions, and flow of a bioceramic root canal sealer. J Endod 3:842–845
Zhang H, Shen Y, Ruse ND, Haapasalo M (2009) Antibacterial activity of endodontic sealers by modified direct contact test against Enterococcus faecalis. J Endod 35:1051–1055
Lovato KF, Sedgley CM (2011) Antibacterial activity of Endosequence root repair material and proroot MTA against clinical isolates of Enterococcus faecalis. J Endod 37:1542–1546
Fernández R, Restrepo JS, Aristizábal DC, Álvarez LG (2016) Evaluation of the filling ability of artificial lateral canals using calcium silicate-based and epoxy resin-based endodontic sealers and two gutta-percha filling techniques. Int Endod J 4:365–373
International Organization for Standardization (2012) International Standard ISO 6876:2012: dental root canal sealing materials. Geneva: International Organization for Standardization
American Dental Association (2000) ANSI/ADA Specification n° 57 - Endodontic Sealing Material. Chicago: ADA
ASTM International C266–07 (2007) Standard test method for time of setting of hydraulic cement paste by Gillmore needles
Loushine BA, Bryan TE, Looney SW, Gillen BM, Loushine RJ, Weller RN, Pashley DH, Tay FR (2011) Setting properties and cytotoxicity evaluation of a premixed bioceramic root canal sealer. J Endod 5:673–677
Han L, Okiji T (2013) Bioactivity evaluation of three calcium silicate-based endodontic materials. Int Endod J 9:808–814
Zhou HM, Shen Y, Zheng W, Li L, Zheng YF, Haapasalo M (2013) Physical properties of 5 root canal sealers. J Endod 10:1281–1286
Xuereb M, Vella P, Damidot D, Sammut CV, Camilleri J (2015) In situ assessment of the setting of tricalcium silicate-based sealers using a dentin pressure model. J Endod 1:111–124
Duarte MAH, Marciano MA, Vivan RR, Tanomaru Filho M, Tanomaru JMG, Camilleri J (2018) Tricalcium silicate-based cements: properties and modifications. Braz Oral Res 1:e70
Massi S, Tanomaru-Filho M, Silva GF, Duarte MA, Grizzo LT, Buzalaf MA, Guerreiro-Tanomaru JM (2011) pH, calcium ion release, and setting time of an experimental mineral trioxide aggregate-based root canal sealer. J Endod 6:844–846
Viapiana R, Flumignan DL, Guerreiro-Tanomaru JM, Camilleri J, Tanomaru-Filho M (2014) Physicochemical and mechanical properties of zirconium oxide and niobium oxide modified Portland cement-based experimental endodontic sealers. Int Endod J 5:437–448
Mendes AT, Silva PBD, Só BB, Hashizume LN, Vivan RR, Rosa RAD, Duarte MAH, Só MVR (2018) Evaluation of physicochemical properties of new calcium silicate-based sealer. Braz Dent J 6:536–540
Guo YJ, Du TF, Li HB, Shen Y, Mobuchon C, Hieawy A, Wang ZJ, Yang Y, Ma J, Haapasalo M (2016) Physical properties and hydration behavior of a fast-setting bioceramic endodontic material. BMC Oral Health 20:16–23
Lee JK, Kwak SW, Ha JH, Lee W, Kim HC (2017) Physicochemical properties of epoxy resin-based and bioceramic-based root canal sealers. Bioinorg Chem Appl 2582849
Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG (2010) Animal research: reporting in vivo experiments: the ARRIVE guidelines. J Gene Med 12:561–563
Smith AJ, Clutton RE, Lilley E, Hansen KEA, Brattelid T (2018) PREPARE: guidelines for planning animal research and testing. Lab Anim 52:135–141
Versiani MA, Carvalho-Junior JR, Padilha MIAF, Lacey S, Pascon EA, Sousa-Neto MD (2006) A comparative study of physicochemical properties of AH Plus and Epiphany root canal sealants. Int Endod J 6:464–471
Flores DS, Rached FJ Jr, Versiani MA, Guedes DF, Sousa-Neto MD, Pécora JD (2011) Evaluation of physicochemical properties of four root canal sealers. Int Endod J 2:126–135
Marin-Bauza GA, Rached-Junior FJ, Souza-Gabriel AE, Sousa-Neto MD, Miranda CE, Silva-Sousa YT (2010) Physicochemical properties of methacrylate resin-based root canal sealers. J Endod 9:1531–1536
Andolfatto C, Bonetti-Filho I, Carlos IZ, Guerreiro-Tanomaru JM, Kuga MC, Tormin FBC, Faria G (2017) Cytocompatibility, physical properties, and antibiofilm activity of endodontic sealers with amoxiciline. Microsc Res Tech 80:1036–1048
Kaup M, Schäfer E, Dammaschke T (2015) An in vitro study of different material properties of Biodentine compared to ProRoot MTA. Head Face Med 11:16
Sousa-Neto MD, Guimarães LF, Saquy PC, Pécora JD (1999) Effect of different grades of gum resins and hydrogenated resins on the solubility, disintegration, and dimensional alterations of Grossman cement. J Endod 7:477–480
Higginbotham (1967) A comparative study of the physical properties of five commonly used root canal sealers. Oral Surg Oral Med Oral Pathol 24:89–101
Torabinejad M, Kiger RD (1980) Experimentally induced alterations in periapical tissues of the cat. J Dent Res 1:87–96
Walton RE, Ardjmand K (1992) Histological evaluation of the presence of bacteria in induced periapical lesions in monkeys. J Endod 5:216–227
Cohenca N, Romualdo PC, da Silva LA, da Silva RA, de Queiroz AM, De Rossi A, Nelson-Filho P (2015) Tissue response to root canal irrigation systems in dogs’ teeth with apical periodontitis. Clin Oral Investig 5:1147–1156
Alsulaimani RS (2018) Immediate and delayed repair of 2 sizes of furcal perforations in dogs’ teeth using mineral trioxide aggregate cement. J Endod 6:1000–1006
Zmener O (2004) Tissue response to a new methacrylate-based root canal sealer: preliminary observations in the subcutaneous connective tissue of rats. J Endod 5:348–351
Garcia L d F, Marques AA, Roselino LM, Pires-de-Souza FC, Consani S (2010) Biocompatibility evaluation of Epiphany/Resilon root canal filling system in subcutaneous tissue of rats. J Endod 1:110–114
Belladonna FG, Calasans-Maia MD, Novellino Alves AT, de Brito Resende RF, Souza EM, Silva EJ, Fidel SR, De-Deus G (2014) Biocompatibility of a self-adhesive gutta-percha-based material in subcutaneous tissue of mice. J Endod 1:1869–1873
Brandão PM, de Figueiredo JAP, Morgental RD, Scarparo RK, Hartmann RC, Souza RA (2019) Correction to: Influence of foraminal enlargement on the healing of periapical lesions in rat molars. Clin Oral Investig 4:2001–2003
Scarparo RK, Dondoni L, Böttcher DE, Grecca FS, Figueiredo JA, Kantarci A, Van Dyke TE, Batista EL Jr (2014) Intracanal delivery of Resolvin E1 controls inflammation in necrotic immature rat teeth. J Endod 5:678–682
Yoneda N, Noiri Y, Matsui S, Kuremoto K, Maezono H, Ishimoto T, Nakano T, Ebisu S, Hayashi M (2017) Development of a root canal treatment model in the rat. Sci Rep 1:3315
Calasans-Maia MD, Melo BR, Alves AT, Resende RF, Louro RS, Sartoretto SC, Granjeiro JM, Alves GG (2016) Cytocompatibility and biocompatibility of nanostructured carbonated hydroxyapatite spheres for bone repair. J Appl Oral Sci 6:599–608
Hess D, Solomon E, Spears R, He J (2011) Retreatability of a bioceramic root canal sealing material. J Endod 11:1547–1549
Funding
This study was partially founded by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies were in accordance with the ethical standards of the institutional and/or national research committee and based on the welfare of animals.
Informed consent
For this type of study, formal consent is not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Silva, E.J.N.L., Ehrhardt, I.C., Sampaio, G.C. et al. Determining the setting of root canal sealers using an in vivo animal experimental model. Clin Oral Invest 25, 1899–1906 (2021). https://doi.org/10.1007/s00784-020-03496-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03496-x