Abstract
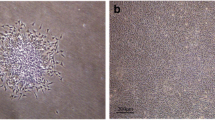
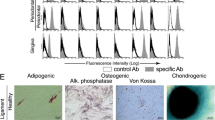
Stem cell therapy is a promising area in regenerative medicine. Periodontal granulation tissues are often discarded during conventional surgery. If stromal stem cells can be isolated from these tissues, they can be used for subsequent surgery on the same patient. Fifteen human periodontal granulation tissue samples were obtained from intrabony defects during surgery. Immunohistochemistry (IHC) was carried out on five of the samples to identify STRO-1, a marker of mesenchymal stem cells. Five samples underwent flow cytometry analysis for the same marker. The remaining five samples were characterized by “colony formation unit-fibroblast” (CFU-f) assay and selected for proliferation assay, flow cytometry of stem cell markers, immunocytochemistry (ICC), multipotent differentiation assays, and repairing critical-size defects in mice. The ratio of STRO-1+ cells detected by IHC was 5.91 ± 1.50%. The analysis of flow cytometry for STRO-1 was 6.70 ± 0.81%. Approximately two thirds of the CFU-f colonies had a strong reaction to STRO-1 in ICC staining. The cells were multipotent both in vitro and in vivo. Mice given bone grafts and stem cells showed significantly better bone healing than those without stem cells. Multipotent stromal stem cells can be isolated from human periodontal granulation tissues. These cells improve new bone formation when transplanted in mouse calvarial defects. Isolating stem cells from relatively accessible sites without extra procedures is clinically advantageous. This study demonstrated that human periodontal granulation tissues contain isolatable multipotent stem cells. The cells may be a good source for autotransplantation in subsequent treatment.






Similar content being viewed by others
References
Persidis A (1999) Tissue engineering. Nat Biotechnol 17:508–509
Bartold PM, Xiao Y, Lyngstaadas SP, Paine ML, Snead ML (2006) Principles and applications of cell delivery systems for periodontal regeneration. Periodontol 200(41):123–135
Tuch BE (2006) Stem cells—a clinical update. Aust Fam Physician 35:719–21
Pozzobon M, Ghionzoli M, De Coppi P (2010) ES, iPS, MSC, and AFS cells. Stem cells exploitation for pediatric surgery: current research and perspective. Pediatr Surg Int 26:3–10
Prockop DJ (1997) Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 276:71–74
Gronthos S, Akintoye SO, Wang C-Y, Shi S (2006) Bone marrow stromal stem cells for tissue engineering. Periodontol 2000(41):188–195
Gronthos S, Mankani M, Brahim J, Robey PG, Shi S (2000) Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci USA 97:13625–13630
Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG, Shi S (2003) SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci USA 100:5807–5812
Seo BM, Miura M, Gronthos S, Bartold PM, Batouli S, Brahim J, Young M, Robey PG, Wang CY, Shi S (2004) Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 364:149–155
Seo BM, Sonoyama W, Yamaza T, Coppe C, Kikuiri T, Akiyama K, Lee JS, Shi S (2008) SHED repair critical-size calvarial defects in mice. Oral Dis 14:428–434
Morsczeck C, Schmalz G, Reichert TE, Völlner F, Galler K, Driemel O (2008) Somatic stem cells for regenerative dentistry. Clin Oral Investig 12:113–118
Wennström JL, Heijl L, Lindhe J (2003) Periodontal surgery: access therapy. In: Lindhe J (ed) Clinical periodontology and implant dentistry, 4th edn. Blackwell Munksgaard, Oxford, pp 519–560
Häkkinen L, Larjava H (1992) Characterization of fibroblast clones from periodontal granulation tissue in vitro. J Dent Res 71:1901–1907
Dennis JE, Carbillet JP, Caplan AI, Charbord P (2002) The STRO-1+ marrow cell population is multipotential. Cells Tissues Organs 170:73–82
Gronthos S, Zannettino AC, Hay SJ, Shi S, Graves SE, Kortesidis A, Simmons PJ (2003) Molecular and cellular characterization of highly purified stromal stem cells derived from human bone marrow. J Cell Sci 116:1827–1835
De Bari C, Dell'Accio F, Luyten FP (2001) Human periosteum-derived cells maintain phenotypic stability and chondrogenic potential throughout expansion regardless of donor age. Arthritis Rheum 44:85–95
Liu Y, Zheng Y, Ding G, Fang D, Fang D, Zhang C, Bartold PM, Gronthos S, Shi S, Wang S (2008) Periodontal ligament stem cell-mediated treatment for periodontitis in miniature swine. Stem cells 26:1065–1073
Yang Y, Rossi FM, Putnins EE (2010) Periodontal regeneration using engineered bone marrow mesenchymal stromal cells. Biomaterials 33:8574–8582
Clarke E, McCann SR (1989) Age dependent in vitro stromal growth. Bone Marrow Transplant 4:596–597
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Mulitilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228
Mostafa SS, Miller WM, Papoutsakis ET (2000) Oxygen tension influences the differentiation, maturation and apoptosis of human megakaryocytes. Br J Haematol 111:879–889
Grayson WL, Zhao F, Izadpanah R, Bunnell B, Ma T (2006) Effects of hypoxia on human mesenchymal stem cell expansion and plasticity in 3D constructs. J Cell Physiol 207:331–339
Semenza GL (2001) HIF-1α, O2, and the 3PHDs: how animal cells signal hypoxia to the nucleus. Cell 107:1–3
Lennon DP, Edmison JM, Caplan A (2001) Cultivation of rat marrow-derived mesenchymal stem cells in reduced oxygen tension: effects on in vitro and in vivo osteochondrogenesis. J Cell Physiol 187:345–355
Zhou S, Lechpammer S, Greenberger JS, Glowacki J (2005) Hypoxia inhibition of adipocytogenesis in human bone marrow stromal cells requires transforming growth factor-β/Smad3 signaling. J Biol Chem 280:22688–22696
Cowan CM, Shi YY, Aalami OO, Chou YF, Mari C, Thomas R, Quarto N, Contag CH, Wu B, Longaker MT (2004) Adipose-derived adult stromal cells heal critical-size mouse calvarial defects. Nat Biotechnol 22:560–567
Oreffo RO, Cooper C, Mason C, Clements M (2005) Mesenchymal stem cells lineage, plasticity, and skeletal therapeutic potential. Stem Cell Rev 1:169–178
Acknowledgements
This study was supported in part by the National Science Council, Taiwan under grant NSC 96–2314-B-006–069. We would like to thank two anonymous (unknown) reviewers and the editor for their comments.
Conflicts of interest
The authors declare that there are no conflicts of interest in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hung, TY., Lin, HC., Chan, YJ. et al. Isolating stromal stem cells from periodontal granulation tissues. Clin Oral Invest 16, 1171–1180 (2012). https://doi.org/10.1007/s00784-011-0600-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-011-0600-5