Abstract
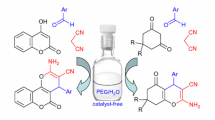
A domino catalytic reaction between terminal alkynes, isocyanates, and malonates has been developed. Copper acetylides attack on isocyanates to form a propargylic amide species which further reacted with malonates by the action of t-BuOLi to form dihydropyridine-3-carboxylates. The regioselectivity of the reaction has been examined with unsymmetrical malonate. The procedure is easy to implement, has a broad scope and allows the construction of the scaffolds by creating three covalent bonds in one pot.
Graphic abstract

Similar content being viewed by others
References
Liu JQ, Ma YG, Zang MM, Wang XS (2017) J Org Chem 82:4918
Fujino D, Yorimitsu H, Osuka A (2014) J Am Chem Soc 136:6255
Echavarren AM, Jiaoc N, Gevorgyand V (2016) Chem Soc Rev 45:4445
Zhou B, Chen H, Wang C (2013) J Am Chem Soc 135:1264
Yang X, Jin X, Wang C (2016) Adv Synth Catal 358:2436
Knöpfel TF, Carreira EM (2003) J Am Chem Soc 125:6054
Varmazyar A, Nozad Goli-Kand A, Sedaghat S, Khalaj M, Arab-Salmanabadi S (2019) J Heterocycl Chem 56:1850
Guo M, Li D, Zhang Z (2003) J Org Chem 68:10172
Xu X, Gao J, Cheng D, Li J, Qiang G, Guoa H (2008) Adv Synth Catal 350:61
Yoo WJ, Lia CJ (2008) Adv Synth Catal 350:1503
Wang H, Pesciaioli F, Oliveira JC, Warratz S, Ackermann L (2017) Angew Chem Int Ed 56:15063
Lu QQ, Cembellin S, Greßies S, Singha S, Daniliuc C, Glorius F (2018) Angew Chem Int Ed 57:1399
Ranjan A, Mandal A, Yerande SG, Dethe DH (2015) Chem Commun 51:14215
Patil NT, Raut VS (2010) J Org Chem 75:6961
Meldal M, Tornøe CW (2008) Chem Rev 108:2952
Ghazanfarpour-Darjani M, Babapour-Kooshalshahi M, Mousavi-Safavi SM, Akbari-Neyestani J, Khalaj M (2016) Synlett 27:259
Samzadeh-Kermani A, Ghasemi S (2019) J Heterocycl Chem 56:2202
Samzadeh-Kermani A (2016) Tetrahedron 72:5301
Samzadeh-Kermani A (2019) J Heterocycl Chem 56:450
Beigi-Somar V, Homami SS, Ghazanfarpour-Darjani M, Monzavi AH (2019) J Heterocycl Chem. https://doi.org/10.1002/jhet.3756
Potuganti GR, Indukuri DR, Nanubolu JB, Alla M (2018) J Org Chem 83:15186
Svensson A, Larsson A, Emtenäs H, Hedenström M, Fex T, Hultgren SJ, Pinkner JS, Almqvist F, Kihlberg J (2001) ChemBioChem 2:915
Joachim G (1994) Angew Chem Int Ed 33:1699
Dragovich PS, Prins TJ, Ru Z, Brown EL, Maldonado FC, Fuhrman SA, Zalman LS, Iuniland L, Lee CA, Worland ST (2002) J Med Chem 45:1607
Cocco MT, Congiu C, Onnis V (2000) Eur J Med Chem 35:545
Yavari I, Souri S (2007) Synlett 2007:2969
Samzadeh-Kermani A (2015) Synlett 26:643
Kibou Z, Cheikh N, Villemin D, Choukchou-Braham N, Mostefa-Kara B, Benabdallah M (2011) Int J Org Chem 1:242
Abadi AH, Ibrahim TM, Abouzid KM, Lehmann J, Tinsley HN, Gary BD, Piazza GA (2009) Bioorg Med Chem 17:5974
Imase H, Noguchi K, Hirano M, Tanaka K (2008) Org Lett 10:3563
Chun YS, Ryu KY, Ko YO, Hong JY, Hong J, Shin H, Lee S (2009) J Org Chem 74:7556
Cheng G, Xue L, Weng Y, Cui X (2017) J Org Chem 82:9515
Jahanshada M, Manafia MR, Mousavi-Safavi SM, Homamia SS, Ghazanfarpour-Darjani M (2019) Tetrahedron Lett 60:151216
Ghazanfarpour-Darjani M, Barat-Seftejani F, Khalaj M, Mousavi-Safavi SM (2017) Helv Chim Acta 100:1700082
Yang CT, Fu Y, Huang YB, Yi J, Guo QX, Liu L (2009) Angew Chem Int Ed 48:7398
Varmazyar A, Sedaghat S, Nozad Goli-Kand A, Khalaj M, Arab-Salmanabadi S (2019) Mol Divers. https://doi.org/10.1007/s11030-019-09978-9
Tang JS, Xie YX, Wang ZQ, Li JH (2011) Synthesis 2011:2789
Acknowledgements
We wish to thank for a grant from Islamic Azad University, South Tehran Branch.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Beigi-Somar, V., Homami, S.S., Ghazanfarpour-Darjani, M. et al. A catalytic multicomponent protocol to dihydropyridine-3-carboxylate from terminal alkynes, isocyanates, and malonates. Monatsh Chem 151, 231–241 (2020). https://doi.org/10.1007/s00706-020-02548-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-020-02548-y