Abstract
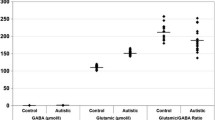
Agmatine is a polyamine endogenously synthesized from arginine and is considered to be a new neurotransmitter. Agmatine has been implicated in the pathophysiology of several diseases such as anxiety disorder, depression, and schizophrenia. Agmatine also possesses anticonvulsant, neuroprotective, antiapoptotic, antioxidant, anxiolytic, and antidepressant effects. Furthermore, agmatine inhibits the nitric oxide synthase enzyme and exerts antagonist effects on NMDA, alpha-2, and imidazoline receptors. Considering these characteristics, the present study investigated whether agmatine plays a role in the pathogenesis of autistic spectrum disorders (ASDs). Therefore, plasma agmatine levels were evaluated in 34 patients with ASD and 28 non-ASD controls. Plasma agmatine levels were measured using the HPLC method. The study found remarkably lower agmatine levels in patients with ASD compared with the non-ASD control group (p < 0.001). These findings support the notion that agmatine might contribute to the pathogenesis of ASD and may serve as a new target for treatment.

Similar content being viewed by others
References
Akyol O, Zoroglu SS, Armutcu F, Sahin S, Gurel A (2004) Nitric oxide as a physiopathological factor in neuropsychiatric disorders. In Vivo 18(3):377–390
American Psychiatry Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatry Association, Washington, DC
Aricioglu F, Kan B, Yillar O, Korcegez E, Berkman K (2003) Effect of agmatine on electrically and chemically induced seizures in mice. Ann N Y Acad Sci 1009(1):141–146
Arndt MA, Battaglia V, Parisi E, Lortie MJ, Isome M, Baskerville C et al (2009) The arginine metabolite agmatine protects mitochondrial function and confers resistance to cellular apoptosis. Am J Physiol Cell Physiol 296(6):C1411–C1419
Carlson GC (2012) Glutamate receptor dysfunction and drug targets across models of autism spectrum disorders. Pharmacol Biochem Behav 100(4):850–854
Çelik VK, Ersan EE, Kilicgun H, Kapancik S, Ersan S (2016) Agmatine mediated hypertonic stress development in schizophrenia: a novel study. Neuropsychiatry 6(5):184–189
Chauhan V, Chauhan A (2009) Chapter 10 abnormalities in membrane lipids, membrane-associated proteins, and signal transduction in autism. In: Chauhan A, Chauhan V, Brown WT (eds) Autism: oxidative stress, inflammation, and immune abnormalities. CRC Press, Boca Raton, pp 177–206
Choudhury PR, Lahiri S, Rajamma U (2012) Glutamate mediated signaling in the pathophysiology of autism spectrum disorders. Pharmacol Biochem Behav 100(4):841–849
Ciccoli L, De Felice C, Paccagnini E, Leoncini S, Pecorelli A, Signorini C, Belmonte G, Guerranti R, Cortelazzo A, Gentile M, Zollo G, Durand T, Valacchi G, Rossi M, Hayek J (2013) Erythrocyte shape abnormalities, membrane oxidative damage, and β-actin alterations: an unrecognized triad in classical autism. Mediat Inflamm 2013:432616-1–432616-11
Condello S, Currò M, Ferlazzo N, Caccamo D, Satriano J, Ientile R (2011) Agmatine effects on mitochondrial membrane potential and NF-κB activation protect against rotenone-induced cell damage in human neuronal-like SH-SY5Y cells. J Neurochem 116(1):67–75
Demehri S, Homayoun H, Honar H, Riazi K, Vafaie K, Roushanzamir F, Dehpour AR (2003) Agmatine exerts anticonvulsant effect in mice: modulation by α 2-adrenoceptors and nitric oxide. Neuropharmacology 45(4):534–542
Erickson CA, Posey DJ, Stigler KA, Mullett J, Katschke AR, McDougle CJ (2007) A retrospective study of memantine in children and adolescents with pervasive developmental disorders. Psychopharmacology 191(1):141–147
Essa MM, Braidy N, Vijayan KR, Subash S, Guillemin GJ (2013) Excitotoxicity in the pathogenesis of autism. Neurotox Res 23(4):393–400
Feng Y, Piletz JE, Leblanc MH (2002) Agmatine suppresses nitric oxide production and attenuates hypoxic-ischemic brain injury in neonatal rats. Pediatr Res 52(4):606–611
Freitas AE, Neis VB, Rodrigues ALS (2016) Agmatine, a potential novel therapeutic strategy for depression. Eur Neuropsychopharmacol 26(12):1885–1899
Gatto CL, Broadie K (2010) Genetic controls balancing excitatory and inhibitory synaptogenesis in neurodevelopmental disorder models. Front Synaptic Neurosci 2:4
Gawali NB, Chowdhury AA, Kothavade PS, Bulani VD, Nagmoti DM, Juvekar AR (2016) Involvement of nitric oxide in anticompulsive-like effect of agmatine on marble-burying behaviour in mice. Eur J Pharmacol 770:165–171
Ghaleiha A, Asadabadi M, Mohammadi MR, Shahei M, Tabrizi M, Hajiaghaee R, Akhondzadeh S (2013) Memantine as adjunctive treatment to risperidone in children with autistic disorder: a randomized, double-blind, placebo-controlled trial. Int J Neuropsychopharmacol 16(4):783–789
Ghezzo A, Visconti P, Abruzzo PM, Bolotta A, Ferreri C, Gobbi G, Pipitone E (2013) Oxidative stress and erythrocyte membrane alterations in children with autism: correlation with clinical features. PLoS One 8(6):e66418
Gong ZH, Li YF, Zhao N, Yang HJ, Su RB, Luo ZP, Li J (2006) Anxiolytic effect of agmatine in rats and mice. Eur J Pharmacol 550(1):112–116
Grillo MA, Colombatto S (2004) Metabolism and function in animal tissues of agmatine, a biogenic amine formed from arginine. Amino Acids 26(1):3–8
Halaris A, Plietz J (2007) Agmatine. CNS Drugs 21(11):885–900
Halaris A, Zhu H, Feng Y, Piletz JE (1999) Plasma agmatine and platelet imidazoline receptors in depression. Ann N Y Acad Sci 881(1):445–451
Hassan TH, Abdelrahman HM, Fattah NRA, El-Masry NM, Hashim HM, El-Gerby KM, Fattah NRA (2013) Blood and brain glutamate levels in children with autistic disorder. Res Autism Spectr Disord 7(4):541–548
Hussman JP (2001) Letters to the editor: suppressed GABAergic inhibition as a common factor in suspected etiologies of autism. J Autism Dev Disord 31(2):247–248
Jansson LC, Åkerman KE (2014) The role of glutamate and its receptors in the proliferation, migration, differentiation and survival of neural progenitor cells. J Neural Transm 121(8):819–836
Kastner T, Walsh K, Shulman L, Alam F, Flood S (2016) Ketamine and the core symptoms of autism. Int J Disabil Hum Dev 15(1):121–123
Kim JW, Seung H, Kim KC, Gonzales ELT, Oh HA, Yang SM, Shin CY (2017) Agmatine rescues autistic behaviors in the valproic acid-induced animal model of autism. Neuropharmacology 113:71–81
Lavinsky D, Arteni NS, Netto CA (2003) Agmatine induces anxiolysis in the elevated plus maze task in adult rats. Behav Brain Res 141(1):19–24
Lee EJ, Choi SY, Kim E (2015) NMDA receptor dysfunction in autism spectrum disorders. Curr Opin Pharmacol 20:8–13
Li G, Regunathan S, Barrow CJ, Eshraghi J, Cooper R, Reis DJ (1994) Agmatine: an endogenous clonidine-displacing substance in the brain. Science 263(5149):966–969
Liu P, Jing Y, Collie ND, Dean B, Bilkey DK, Zhang H (2016) Altered brain arginine metabolism in schizophrenia. Transl Psychiatry 6(8):e871
Manent JB, Represa A (2007) Neurotransmitters and brain maturation: early paracrine actions of GABA and glutamate modulate neuronal migration. Neuroscientist 13(3):268–279
Ming X, Gordon E, Kang N, Wagner GC (2008) Use of clonidine in children with autism spectrum disorders. Brain Dev 30(7):454–460
Moretti M, Matheus FC, de Oliveira PA, Neis VB, Ben J, Walz R, Prediger RD (2014) Role of agmatine in neurodegenerative diseases and epilepsy. Front Biosci (Elite Ed) 6:341–359
Olmos G, DeGregorio-Rocasolano N, Regalado MP, Gasull T, Boronat MA, Trullas R, García-Sevilla JA (1999) Protection by imidazol (ine) drugs and agmatine of glutamate-induced neurotoxicity in cultured cerebellar granule cells through blockade of NMDA receptor. Br J Pharmacol 127(6):1317–1326
Owley T, Salt J, Guter S, Grieve A, Walton L, Ayuyao N, Cook EH Jr (2006) A prospective, Open-Label Trial of memantine in the treatment of cognitive, behavioral, and memory dysfunction in pervasive developmental disorders. J Child Adolesc Psychopharmacol 16(5):517–524
Piletz JE, May PJ, Wang G, Zhu H (2003) Agmatine crosses the blood–brain barrier. Ann N Y Acad Sci 1009(1):64–74
Piletz JE, Aricioglu F, Cheng JT, Fairbanks CA, Gilad VH, Haenisch B, Liu P (2013) Agmatine: clinical applications after 100 years in translation. Drug Discov Today 18(17):880–893
Regunathan S, Dozier D, Takkalapalli R, Phillips WJ (2009) Agmatine levels in the cerebrospinal fluid of normal human volunteers. J Pain Palliat Care Pharmacother 23(1):35–39
Reis DJ, Regunathan S (2000) Is agmatine a novel neurotransmitter in brain? Trends Pharmacol Sci 21(5):187–193
Rojas DC (2014) The role of glutamate and its receptors in autism and the use of glutamate receptor antagonists in treatment. J Neural Transm 121(8):891–905
Schuber F (1989) Influence of polyamines on membrane functions. Biochem J 260(1):1
Shende VM, Patel FR (2014) Agmatine ameliorates social isolation induced obsessive-compulsive behavior in mice. J Glob Trends Pharm Sci 5(4):2048–2051
Shimmura C, Suda S, Tsuchiya KJ, Hashimoto K, Ohno K, Matsuzaki H, Suzuki K (2011) Alteration of plasma glutamate and glutamine levels in children with high-functioning autism. PLoS One 6(10):e25340
Shinohe A, Hashimoto K, Nakamura K, Tsujii M, Iwata Y, Tsuchiya KJ, Matsuzaki H (2006) Increased serum levels of glutamate in adult patients with autism. Prog Neuropsychopharmacol Biol Psychiatry 30(8):1472–1477
Söğüt S, Zoroğlu SS, Özyurt H, Yılmaz HR, Özuğurlu F, Sivaslı E, Tarakçıoğlu M (2003) Changes in nitric oxide levels and antioxidant enzyme activities may have a role in the pathophysiological mechanisms involved in autism. Clin Chim Acta 331(1):111–117
Su RB, Wei XL, Zheng JQ, Liu Y, Lu XQ, Li J (2004) Anticonvulsive effect of agmatine in mice. Pharmacol Biochem Behav 77(2):345–349
Sucuoglu B, Oktem F, Akkok F, Gokler B (1996) A study of the scales for the assessment of the children with autism. Psikiyatri Psikoloji Psikofarmakoloji 4:116–121
Sweeten TL, Posey DJ, Shankar S, McDougle CJ (2004) High nitric oxide production in autistic disorder: a possible role for interferon-γ. Biol Psychiatry 55(4):434–437
Tabor CW, Tabor H (1984) Polyamines. Annu Rev Biochem 53(1):749–790
Terán Y, Ponce O, Betancourt L, Hernández L, Rada P (2012) Amino acid profile of plasma and cerebrospinal fluid in preeclampsia. Pregnancy Hypertens Int J Womens Cardiovasc Health 2(4):416–422
Uzbay IT (2009) New pharmacological approaches to the treatment of schizophrenia. Turk Psikiyatri Derg 20(2):175–182
Uzbay TI (2012a) The pharmacological importance of agmatine in the brain. Neurosci Biobehav Rev 36(1):502–519
Uzbay T (2012b) A new target for diagnosis and treatment of CNS disorders: agmatinergic system. Curr Med Chem 19(30):5116–5121
Uzbay T, Goktalay G, Kayir H, Eker SS, Sarandol A, Oral S, Kirli S (2013) Increased plasma agmatine levels in patients with schizophrenia. J Psychiatr Res 47(8):1054–1060
Uzunova G, Hollander E, Shepherd J (2014) The role of ionotropic glutamate receptors in childhood neurodevelopmental disorders: autism spectrum disorders and fragile × syndrome. Curr Neuropharmacol 12(1):71–98
Wang WP, Iyo AH, Miguel-Hidalgo J, Regunathan S, Zhu MY (2006) Agmatine protects against cell damage induced by NMDA and glutamate in cultured hippocampal neurons. Brain Res 1084(1):210–216
Wei H, Alberts I, Li X (2014) The apoptotic perspective of autism. Int J Dev Neurosci 36:13–18
Yang P, Chang CL (2014) Glutamate-mediated signaling and autism spectrum disorders: emerging treatment targets. Curr Pharm Des 20(32):5186–5193
Yang XC, Reis DJ (1999) Agmatine selectively blocks the N-methyl-d-aspartate subclass of glutamate receptor channels in rat hippocampal neurons. J Pharmacol Exp Ther 288(2):544–549
Zhu MY, Wang WP, Bissette G (2006) Neuroprotective effects of agmatine against cell damage caused by glucocorticoids in cultured rat hippocampal neurons. Neuroscience 141(4):2019–2027
Zhu MY, Wang WP, Cai ZW, Regunathan S, Ordway G (2008) Exogenous agmatine has neuroprotective effects against restraint-induced structural changes in the rat brain. Eur J Neurosci 27(6):1320–1332
Funding
The present study was supported by Ordu University Scientific Research Project (Project no: AR-1520).
Author information
Authors and Affiliations
Contributions
Dr. EE produced the idea and planned the study. In addition, he recruited the subjects, performed clinical and diagnostic evaluation and statistical analyses, and wrote the manuscript. Dr. İİ studied the samples with HPLC and contributed to the manuscript writing process. Both authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no financial interest or potential conflicts of interest.
Rights and permissions
About this article
Cite this article
Esnafoglu, E., İrende, İ. Decreased plasma agmatine levels in autistic subjects. J Neural Transm 125, 735–740 (2018). https://doi.org/10.1007/s00702-017-1836-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-017-1836-2