Abstract
Background
Navigated transcranial magnetic stimulation (nTMS) is a non-invasive mapping tool to locate functional areas of the brain. While gaining importance in the preoperative planning process in motor eloquent regions, its usefulness for reliably identifying language areas is still being discussed. The aim of this study was to identify biometric factors which might influence and therefore bias the results of repetitive nTMS (rnTMS) over cortex areas relevant for language.
Method
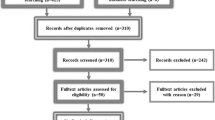
We included data of 101 patients with language eloquent brain lesions who underwent preoperative rnTMS examination bihemispherically. Prior to rnTMS mapping, all patients performed two to three baseline runs of a picture-naming paradigm without stimulation, and only promptly and correctly named objects were retained for TMS mapping. Nine biometric factors (age, gender, baseline dataset, cognitive performance score, aphasia score, histology of lesion, affected hemisphere, location of lesion on the hemisphere, pain caused by examination) were included in the statistical analysis measuring their correlation with the incidence of errors during baseline naming as well as during rnTMS mapping.
Results
The incidence of baseline errors correlated with aphasia (p < 0.0001) and cognitive impairment (p < 0.0001). No significant correlation was observed between most biometric factors and errors during rnTMS mapping. Factors significantly affecting the incidence of errors during rnTMS mapping were again aphasia (p < 0.023) and cognitive impairment (p < 0.038). Patients affected by those factors showed a significantly higher baseline error rate, starting at 28% error rate.
Conclusions
Patients with pre-existing aphasia or severe cognitive impairment did still make significantly more mistakes during rnTMS mapping than non-aphasic patients despite baseline stratification, rendering the question of whether the procedure is reliable in those patient groups. Baseline testing revealed a cut-off point at 28% error rate. Interestingly, age or pain (caused by the examination) did not bias the results.




Similar content being viewed by others
References
Adlington RL, Laws KR, Gale TM (2009) Visual processing in Alzheimer’s disease: surface detail and colour fail to aid object identification. Neuropsychologia 47:2574–2583. https://doi.org/10.1016/j.neuropsychologia.2009.05.004
Chang EF, Wang DD, Perry DW, Barbaro NM, Berger MS (2011) Homotopic organization of essential language sites in right and bilateral cerebral hemispheric dominance. J Neurosurg 114:893–902. https://doi.org/10.3171/2010.11.JNS10888
Corina DP, Loudermilk BC, Detwiler L, Martin RF, Brinkley JF, Ojemann G (2010) Analysis of naming errors during cortical stimulation mapping: implications for models of language representation. Brain Lang 115:101–112. https://doi.org/10.1016/j.bandl.2010.04.001
Cummings JL, Benson F, Hill MA, Read S (1985) Aphasia in dementia of the Alzheimer type. Neurology 35:394–397
Damasio AR (1992) Aphasia. N Engl J Med 326:531–539. https://doi.org/10.1056/NEJM199202203260806
Deng X, Zhang Y, Xu L, Wang B, Wang S, Wu J, Zhang D, Wang R, Wang J, Zhao J (2015) Comparison of language cortex reorganization patterns between cerebral arteriovenous malformations and gliomas: a functional MRI study. J Neurosurg 122:996–1003. https://doi.org/10.3171/2014.12.JNS14629
Desmurget M, Bonnetblanc F, Duffau H (2007) Contrasting acute and slow-growing lesions: a new door to brain plasticity. Brain J Neurol 130:898–914. https://doi.org/10.1093/brain/awl300
DGN S1 Leitlinie: Zerebrale Gefäßmalformationen (arteriovenöse Malformationen, arteriovenöse Fisteln, Kavernome). Leitlinien für Diagnostik und Therapie in der Neurologie, Aufl 5 2012. URL: http://www.dgn.org/images/red_leitlinien/LL_2015/PDFs_Download/030088_LL_Zerebrale_Gef%C3%A4%C3%9Fmalformationen.pdf Accessed May 22 2016
Duffau H, Lopes M, Arthuis F, Bitar A, Sichez JP, Van Effenterre R, Capelle L (2005) Contribution of intraoperative electrical stimulations in surgery of low grade gliomas: a comparative study between two series without (1985-96) and with (1996-2003) functional mapping in the same institution. J Neurol Neurosurg Psychiatry 76:845–851. https://doi.org/10.1136/jnnp.2004.048520
Epstein CM, Lah JJ, Meador K, Weissmann JD, Gaitan LE, Dihenia B (1996) Optimum stimulus parameters for lateralized suppression of speech with magnetic brain stimulation. Neurology 47:1590–1593
Frey D, Schilt S, Strack V, Zdunczyk A, Rosler J, Niraula B, Vajkoczy P, Picht T (2014) Navigated transcranial magnetic stimulation improves the treatment outcome in patients with brain tumors in motor eloquent locations. Neuro-Oncology 16:1365–1372. https://doi.org/10.1093/neuonc/nou110
Groppa S, Oliviero A, Eisen A, Quartarone A, Cohen LG, Mall V, Kaelin-Lang A, Mima T, Rossi S, Thickbroom GW, Rossini PM, Ziemann U, Valls-Sole J, Siebner HR (2012) A practical guide to diagnostic transcranial magnetic stimulation: report of an IFCN committee. Clin Neurophysiol 123:858–882. https://doi.org/10.1016/j.clinph.2012.01.010
Hauck T, Tanigawa N, Probst M, Wohlschlaeger A, Ille S, Sollmann N, Maurer S, Zimmer C, Ringel F, Meyer B, Krieg SM (2015) Stimulation frequency determines the distribution of language positive cortical regions during navigated transcranial magnetic brain stimulation. BMC Neurosci 16:5. https://doi.org/10.1186/s12868-015-0143-9
Hodges JR, Salmon DP, Butters N (1992) Semantic memory impairment in Alzheimer’s disease: failure of access or degraded knowledge? Neuropsychologia 30:301–314
Huber W, Poeck K, Willmes K (1984) The Aachen aphasia test. Adv Neurol 42:291–303
Indefrey P (2011) The spatial and temporal signatures of word production components: a critical update. Front Psychol 2:255. https://doi.org/10.3389/fpsyg.2011.00255
Jennum P, Friberg L, Fuglsang-Frederiksen A, Dam M (1994) Speech localization using repetitive transcranial magnetic stimulation. Neurology 44:269–273
Johnson CJ, Paivio A, Clark JM (1996) Cognitive components of picture naming. Psychol Bull 120:113–139
Kalbe E, Kessler J, Calabrese P, Smith R, Passmore AP, Brand M, Bullock R (2004) DemTect: a new, sensitive cognitive screening test to support the diagnosis of mild cognitive impairment and early dementia. Int J Geriatr Psychiatry 19:136–143. https://doi.org/10.1002/gps.1042
Kohn SE, Goodglass H (1985) Picture-naming in aphasia. Brain Lang 24:266–283
Kosla K, Pfajfer L, Bryszewski B, Jaskolski D, Stefanczyk L, Majos A (2012) Functional rearrangement of language areas in patients with tumors of the central nervous system using functional magnetic resonance imaging. Pol J Radiol 77:39–45
Krieg SM, Lioumis P, Makela JP, Wilenius J, Karhu J, Hannula H, Savolainen P, Lucas CW, Seidel K, Laakso A, Islam M, Vaalto S, Lehtinen H, Vitikainen AM, Tarapore PE, Picht T (2017) Protocol for motor and language mapping by navigated TMS in patients and healthy volunteers; workshop report. Acta Neurochir 159:1187–1195. https://doi.org/10.1007/s00701-017-3187-z
Lioumis P, Zhdanov A, Makela N, Lehtinen H, Wilenius J, Neuvonen T, Hannula H, Deletis V, Picht T, Makela JP (2012) A novel approach for documenting naming errors induced by navigated transcranial magnetic stimulation. J Neurosci Methods 204:349–354. https://doi.org/10.1016/j.jneumeth.2011.11.003
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109. https://doi.org/10.1007/s00401-007-0243-4
Martin A, Fedio P (1983) Word production and comprehension in Alzheimer's disease: the breakdown of semantic knowledge. Brain Lang 19:124–141
Moreaud O, David D, Charnallet A, Pellat J (2001) Are semantic errors actually semantic? Evidence from Alzheimer’s disease. Brain Lang 77:176–186. https://doi.org/10.1006/brln.2000.2427
Ojemann G, Ojemann J, Lettich E, Berger M (1989) Cortical language localization in left, dominant hemisphere. An electrical stimulation mapping investigation in 117 patients. J Neurosurg 71:316–326. https://doi.org/10.3171/jns.1989.71.3.0316
Pascual-Leone A, Gates JR, Dhuna A (1991) Induction of speech arrest and counting errors with rapid-rate transcranial magnetic stimulation. Neurology 41:697–702
Picht T, Krieg SM, Sollmann N, Rosler J, Niraula B, Neuvonen T, Savolainen P, Lioumis P, Makela JP, Deletis V, Meyer B, Vajkoczy P, Ringel F (2013) A comparison of language mapping by preoperative navigated transcranial magnetic stimulation and direct cortical stimulation during awake surgery. Neurosurgery 72:808–819. https://doi.org/10.1227/NEU.0b013e3182889e01
Pulvermuller F, Fadiga L (2010) Active perception: sensorimotor circuits as a cortical basis for language. Nat Rev Neurosci 11:351–360. https://doi.org/10.1038/nrn2811
Rosler J, Niraula B, Strack V, Zdunczyk A, Schilt S, Savolainen P, Lioumis P, Makela J, Vajkoczy P, Frey D, Picht T (2014) Language mapping in healthy volunteers and brain tumor patients with a novel navigated TMS system: evidence of tumor-induced plasticity. Clin Neurophysiol 125:526–536. https://doi.org/10.1016/j.clinph.2013.08.015
Rossini PM, Burke D, Chen R, Cohen LG, Daskalakis Z, Di Iorio R, Di Lazzaro V, Ferreri F, Fitzgerald PB, George MS, Hallett M, Lefaucheur JP, Langguth B, Matsumoto H, Miniussi C, Nitsche MA, Pascual-Leone A, Paulus W, Rossi S, Rothwell JC, Siebner HR, Ugawa Y, Walsh V, Ziemann U (2015) Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin Neurophysiol 126:1071–1107. https://doi.org/10.1016/j.clinph.2015.02.001
Ruohonen J, Karhu J (2010) Navigated transcranial magnetic stimulation. Neurophysiol Clin 40:7–17. https://doi.org/10.1016/j.neucli.2010.01.006
Tarapore PE, Findlay AM, Honma SM, Mizuiri D, Houde JF, Berger MS, Nagarajan SS (2013) Language mapping with navigated repetitive TMS: proof of technique and validation. NeuroImage 82:260–272. https://doi.org/10.1016/j.neuroimage.2013.05.018
Thiel A, Herholz K, Koyuncu A, Ghaemi M, Kracht LW, Habedank B, Heiss WD (2001) Plasticity of language networks in patients with brain tumors: a positron emission tomography activation study. Ann Neurol 50:620–629
Thiel A, Schumacher B, Wienhard K, Gairing S, Kracht LW, Wagner R, Haupt WF, Heiss WD (2006) Direct demonstration of transcallosal disinhibition in language networks. J Cereb Blood Flow Metab 26:1122–1127. https://doi.org/10.1038/sj.jcbfm.9600350
Thomas C, Altenmuller E, Marckmann G, Kahrs J, Dichgans J (1997) Language processing in aphasia: changes in lateralization patterns during recovery reflect cerebral plasticity in adults. Electroencephalogr Clin Neurophysiol 102:86–97
Acknowledgements
A special thank you to Heike Schneider for the support in conducting the rnTMS examinations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Thomas Picht has served as a speaker for a TMS manufacturer (Nexstim Oy) but is not a contracted consultant. All other authors certify that they have no affiliations with or involvement in any organisation or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical standards
The study proposal is in accordance with the ethical standards of the Declaration of Helsinki and was approved by the Ethics Commission of the Charité University Hospital (EA4/093/11). All patients gave written informed consent for medical evaluation within the scope of the study.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Schwarzer, V., Bährend, I., Rosenstock, T. et al. Aphasia and cognitive impairment decrease the reliability of rnTMS language mapping. Acta Neurochir 160, 343–356 (2018). https://doi.org/10.1007/s00701-017-3397-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-017-3397-4