Abstract
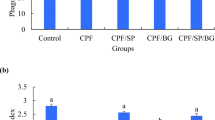
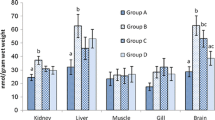
The β-1,3-glucan exerts potential antioxidant effect in experimental animal models, but its role in the protection against fipronil and lead-induced toxicity in fish has not been investigated. In this study, we tested the β-1,3-glucan role against fipronil and lead-induced hepatotoxicity in Cyprinus carpio. Two hundred seventy C. carpio were divided into six groups: control, β-1,3-glucan (0.1 %), fipronil (0.0428 mg/L), lead nitrate (7.3 mg/L), coadministered (fipronil and lead), and coadministered β-1,3-glucan treated. Macrocytic hypochromic anemia with significant decrease in the hepatic antioxidant activities (catalase—CAT, superoxide dismutase—SOD, and glutathione—GSH) and significant increase in the malondialdehyde (MDA) level was observed in fipronil, lead, and the coadministered groups. Moreover, a relative decrease in the level of gene expression (SOD, CAT, glutathione S-transferase—GST, glutathione peroxidase—GPx, and glutathione reductase—GR) was observed. The coadministered β-1,3-glucan-treated group showed an improvement in the erythrocytic pictures, increased hepatic antioxidant activities with a decrease in the MDA level. An improvement in the transcriptional level of SOD, CAT, GST, GPx, and GR was detected. The histopathological finding confirmed the obtained results. It could be concluded that β-1,3-glucan has a hepatoprotective effect against coadministration of fipronil and lead-induced hepatotoxicity in common carp (C. carpio).




Similar content being viewed by others
References
Abdou HM, Hassan MA (2014) Protective role of omega-3 polyunsaturated fatty acid against lead acetate-induced toxicity in liver and kidney of female rats. BioMed Res Int 2014:435857. doi:10.1155/2014/435857
Adegbesan BO, Adenuga GA (2007) Effect of lead exposure on liver lipid peroxidative and antioxidant defense systems of protein-undernourished rats. Biol Trace Elem Res 116:219–225. doi:10.1007/BF02685932
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Agus HH, Sümer S, Erkoç F (2015) Toxicity and molecular effects of di-n-butyl phthalate (DBP) on CYP1A, SOD, and GPx in Cyprinus carpio (common carp). Environ Monit Assess 187:1–8
Asahi M, Kobayashi M, Matsui H, Nakahira K (2015) Differential mechanisms of action of the novel gamma-aminobutyric acid receptor antagonist ectoparasiticides fluralaner (A1443) and fipronil. Pest Manag Sci 71:91–95. doi:10.1002/ps.3768
Blaxhall P, Daisley K (1973) Routine haematological methods for use with fish blood. J Fish Biol 5:771–781
Carriel VS, Aneiros-Fernandez J, Arias-Santiago S, Garzon IJ, Alaminos M, Campos A (2011) A novel histochemical method for a simultaneous staining of melanin and collagen fibers. J Histochem Cytochem 59:270–277. doi:10.1369/0022155410398001
Clasen B et al (2012) Effects of the commercial formulation containing fipronil on the non-target organism Cyprinus carpio: implications for rice-fish cultivation. Ecotoxicol Environ Saf 77:45–51. doi:10.1016/j.ecoenv.2011.10.001
Daci JV, Lewis SM (1996) Practical hematology, 7th ed. Churchill Livingstone, London
Das BK, Pattnaik P, Debnath C, Swain DK, Pradhan J (2013) Effect of beta-glucan on the immune response of early stage of Anabas testudineus (Bloch) challenged with fungus Saprolegnia parasitica. SpringerPlus 2:197. doi:10.1186/2193-1801-2-197
Dobsikova R et al (2012) Effect of beta-1.3/1.6-D-glucan derived from oyster mushroom Pleurotus ostreatus on biometrical, haematological, biochemical, and immunological indices in rainbow trout (Oncorhynchus mykiss). Neuro Endocrinol Lett 33(Suppl 3):96–106
Dugmonits K, Ferencz Á, Jancsó Z, Juhász R, Hermesz E (2013) Major distinctions in the antioxidant responses in liver and kidney of Cd 2+-treated common carp (Cyprinus carpio). Comp Biochem Physiol C Toxicol Pharmacol 158:225–230
El-Nekeety AA, El-Kady AA, Soliman MS, Hassan NS, Abdel-Wahhab MA (2009) Protective effect of Aquilegia vulgaris (L.) against lead acetate-induced oxidative stress in rats. Food Chem Toxicol 47:2209–2215. doi:10.1016/j.fct.2009.06.019
Firat O, Cogun HY, Yuzereroglu TA, Gok G, Firat O, Kargin F, Kotemen Y (2011) A comparative study on the effects of a pesticide (cypermethrin) and two metals (copper, lead) to serum biochemistry of Nile tilapia, Oreochromis niloticus. Fish physiol Biochem 37:657–666. doi:10.1007/s10695-011-9466-3
Garcia-Leston J et al (2012) Assessment of immunotoxicity parameters in individuals occupationally exposed to lead. J Toxicol Environ Health A 75:807–818. doi:10.1080/15287394.2012.690327
Gibbons D, Morrissey C, Mineau P (2015) A review of the direct and indirect effects of neonicotinoids and fipronil on vertebrate wildlife. Environ Sci Pollut Res Int 22:103–118. doi:10.1007/s11356-014-3180-5
Hashish EA, Elgaml SA, El-Murr A, Khalil R (2015) Nephroprotective and antioxidant significance of selenium and alpha-tocopherol on lead acetate-induced toxicity of Nile Tilapia (Oreochromis niloticus). Fish Physiol Biochem 41:651–660. doi:10.1007/s10695-015-0035-z
Hassanain MA, Abbas WT, Ibrahim TB (2012) Skeletal ossification impairment in Nile Tilapia (Oreochromis niloticus) after exposure to lead acetate. Pak J Biol Sci 15:729–735
Jennings KA, Canerdy TD, Keller RJ, Atieh BH, Doss RB, Gupta RC (2002) Human exposure to fipronil from dogs treated with frontline. Vet Hum Toxicol 44:301–303
Karaca M, Varışlı L, Korkmaz K, Özaydın O, Percin F, Orhan H (2014) Organochlorine pesticides and antioxidant enzymes are inversely correlated with liver enzyme gene expression in Cyprinus carpio. Tox Lett 230:198–207
Khidr BM, Mekkawy IA, Harabawy AS, Ohaida AS (2012) Effect of lead nitrate on the liver of the cichlid fish (Oreochromis niloticus): a light microscope study. Pak J Biol Sci 15:854–862
Kim YS, Ke F, Zhang QY (2009) Effect of beta-glucan on activity of antioxidant enzymes and Mx gene expression in virus infected grass carp. Fish Shellfish Immunol 27:336–340. doi:10.1016/j.fsi.2009.06.006
Konwick BJ, Fisk AT, Garrison AW, Avants JK, Black MC (2005) Acute enantioselective toxicity of fipronil and its desulfinyl photoproduct to Ceriodaphnia dubia. Environ Toxicol Chem 24:2350–2355
Luszczek-Trojnar E, Drag-Kozak E, Szczerbik P, Socha M, Popek W (2014) Effect of long-term dietary lead exposure on some maturation and reproductive parameters of a female Prussian carp (Carassius gibelio B.). Environ Sci Pollut Res Int 21:2465–2478. doi:10.1007/s11356-013-2184-x
Meadus WJ (2003) A semi-quantitative RT-PCR method to measure the in vivo effect of dietary conjugated linoleic acid on porcine muscle PPAR gene expression. Biol Procedures Online 5:20–28. doi:10.1251/bpo43
Natt MPHC (1952) A new blood diluent for counting the erythrocytes and leukocytes of the chicken. Poult Sci 31:4
Novotny K, Turzikova A, Komarek J (2000) Speciation of copper, lead and cadmium in aquatic systems by circulating dialysis combined with flame AAS. Fresenius J Anal Chem 366:209–212
Oliveira Ribeiro CA et al (2006) Hematological findings in neotropical fish Hoplias malabaricus exposed to subchronic and dietary doses of methylmercury, inorganic lead, and tributyltin chloride. Environ Res 101:74–80. doi:10.1016/j.envres.2005.11.005
Oropesa AL, Garcia-Cambero JP, Soler F (2009) Effect of a subchronic exposure to simazine on energetic metabolism of common carp (Cyprinus carpio). J Environ Sci Health 44:144–156. doi:10.1080/03601230802599068
Owens CW, Belcher RV (1965) A colorimetric micro-method for the determination of glutathione. Biochem J 94:705–711
Ozdemir S, Toplan S, Tanriverdi G, Sunamak O (2011) The effects of beta-glucan on iron levels and lipid peroxidation in intra-abdominal sepsis in rats. Gen Physiol Biophys 30:138–144. doi:10.4149/gpb_2011_02_138
Patrick L (2006) Lead toxicity part II: the role of free radical damage and the use of antioxidants in the pathology and treatment of lead toxicity. Altern Med Rev 11:114–127
Paul N, Chakraborty S, Sengupta M (2014) Lead toxicity on non-specific immune mechanisms of freshwater fish Channa punctatus. Aquat Toxicol 152:105–112. doi:10.1016/j.aquatox.2014.03.017
Ravindran R et al (2014) Determination of LC50 and LC95 values of fipronil against Haemaphysalis bispinosa based on adult immersion test. J Parasit Dis 38:132–134. doi:10.1007/s12639-012-0181-5
Rusia V, Sood SK (1992) Routine hematological tests. Med Lab Technol 1:252–258
Sahoo PK, Mukherjee SC (2001) Effect of dietary beta-1,3 glucan on immune responses and disease resistance of healthy and aflatoxin B1-induced immunocompromised rohu (Labeo rohita Hamilton). Fish Shellfish Immunol 11:683–695. doi:10.1006/fsim.2001.0345
Silva JP, Coutinho OP (2010) Free radicals in the regulation of damage and cell death—basic mechanisms and prevention. Drug Discov Therapeut 4:144–167
Stier H, Ebbeskotte V, Gruenwald J (2014) Immune-modulatory effects of dietary Yeast Beta-1,3/1,6-D-glucan. Nutr J 13:38. doi:10.1186/1475-2891-13-38
Tingle CC, Rother JA, Dewhurst CF, Lauer S, King WJ (2003) Fipronil: environmental fate, ecotoxicology, and human health concerns. Rev Environ Contam Toxicol 176:1–66
Topashka-Ancheva M, Metcheva R, Teodorova S (2003) Bioaccumulation and damaging action of polymetal industrial dust on laboratory mice Mus musculus alba. II. Genetic, cell, and metabolic disturbances. Environ Res 92:152–160
Valenzuela A (1991) The biological significance of malondialdehyde determination in the assessment of tissue oxidative stress. Life Sci 48:301–309
Vetvicka V, Vannucci L, Sima P (2013) The effects of beta-glucan on fish immunity. N Am J Med Sci 5:580–588. doi:10.4103/1947-2714.120792
Weydert CJ, Cullen JJ (2010) Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat Protoc 5:51–66. doi:10.1038/nprot.2009.197
Wintrobe MM, Greer JP (2009) Wintrobe’s clinical hematology, vol 1. Lippincott Williams & Wilkins, Philadelphia
Wu J, Lu J, Lu H, Lin Y, Wilson PC (2015) Occurrence and ecological risks from fipronil in aquatic environments located within residential landscapes. Sci Total Environ 518–519:139–147. doi:10.1016/j.scitotenv.2014.12.103
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The experiment complied with the guidelines of the Animal Welfare Act and supervised by a veterinarian at the Faculty of Veterinary Medicine, Zagazig University where all the national and institutional guideline was followed.
Rights and permissions
About this article
Cite this article
El-Murr, A., Ali, H.A., Elgaml, S.A. et al. The β-1,3-glucan alleviated the hepatotoxicity induced by combination of fipronil and lead in common carp (Cyprinus carpio) . Comp Clin Pathol 25, 689–697 (2016). https://doi.org/10.1007/s00580-016-2249-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-016-2249-6